
A more recent article on premenstrual syndrome and premenstrual dysphoric disorder is available.
Am Fam Physician. 1998;58(1):183-192
See related patient information handout on premenstrual syndrome, written by the author of this article, is provided on page 197.
Premenstrual syndrome (PMS) refers to a group of menstrually related disorders. Premenstrual symptoms sufficient to impair daily activity and relationships are estimated to occur in up to 40 percent of women of reproductive age, with 5 percent of these women having severe impairment. Although no single causative factor has been identified, PMS is related to ovulatory cycles and resolves at menopause. A specific diagnostic test for PMS does not exist. Treatment is based on the symptoms. Oral contraceptive agents, diuretics and antidepressants have been used successfully to relieve symptoms. Cognitive therapy may help the patient develop appropriate ways to cope with the obstacles, frustrations and discomforts of daily life. Although dietary deficiencies may be difficult to demonstrate, nutritional supplementation may result in significant improvement. Various treatments, including progesterone supplementation, vitamin B6 in high doses, alprazolam and gonadotropin-releasing hormone agonists, are generally not recommended. A comprehensive treatment plan that encourages a healthy lifestyle may relieve symptoms in most women.
Premenstrual syndrome (PMS) refers to a group of menstrually related disorders and symptoms that includes premenstrual dysphoric disorder (PDD) as well as affective disturbances, alterations in appetite, cognitive disturbance, fluid retention and various types of pain. The metabolic and behavioral contexts of menstrually related disorders are being carefully studied.1
Premenstrual symptoms sufficient to impair daily life and relationships are estimated to affect up to 40 percent of women of reproductive age, with severe impairment occurring in approximately 5 percent.2 PMS may have an onset at any time during the reproductive years and, once symptoms are established, they tend to remain fairly constant until menopause. Successful treatment of PMS is possible through careful evaluation of the symptoms and the formulation of a patient-centered treatment plan based on evidence gleaned from well-designed research studies.
Spectrum of the Symptom Complex
| Affective |
| Depression or sadness |
| Irritability |
| Tension |
| Anxiety |
| Tearfulness or crying easily |
| Restlessness or jitteriness |
| Anger |
| Loneliness |
| Appetite change |
| Food cravings |
| Changes in sexual interest |
| Pain |
| Headache or migraine |
| Back pain |
| Breast pain |
| Abdominal cramps |
| General or muscular pain |
| Cognitive or performance |
| Mood instability or mood swings |
| Difficulty in concentrating |
| Decreased efficiency |
| Confusion |
| Forgetfulness |
| Accident-prone |
| Social avoidance |
| Temper outbursts |
| Energetic |
| Fluid retention |
| Breast tenderness or swelling |
| Weight gain |
| Abdominal bloating or swelling |
| Swelling of extremities |
| General somatic |
| Fatigue or tiredness |
| Dizziness or vertigo |
| Nausea |
| Insomnia |
Symptom Correlates
It is likely that a “vulnerability” exists for the development of premenstrual symptoms. Correlates of PMS, such as a strong association with depression, have been found, but no single causative factor has been identified. The vulnerability for development of PMS might consist of one or more genetic, environmental or behavioral factors, which would be consistent with the variety of treatments found to be successful.4 While the numerous symptoms in the perimenstruum have been grouped into symptom clusters, such groupings are not central to successful treatment. Treatment directed at one type of symptoms often yields improvement in other areas.
Affective Symptoms
In 1987, late luteal-phase dysphoric disorder (LLPDD) was included as a condition needing further study in the appendix of the Diagnostic and Statistical Manual of Mental Disorders (3d Ed Rev). In the fourth edition, which was published in 1994, the term “premenstrual dysphoric disorder” was included in the appendix and replaced LLPDD.5 While criteria for PDD focus on affective disturbances, studies of this disorder have also yielded useful information regarding physical symptoms.1
Affective symptoms of PMS strongly resemble major depression, except that PDD differs from major depression in that PDD occurs in the premenstrual phase alone. Abnormalities in central nervous system serotonergic responses have been observed in studies of women with PMS,6 and antidepressant drugs have been found useful in treatment. Selective serotonin reuptake inhibitors have been shown to be effective in the treatment of premenstrual dysphoria.7–10 Noradrenaline reuptake inhibitors have also been found to relieve depressed mood but not other symptoms associated with PDD. In addition, decreased plasma beta-endorphin levels, as well as abnormalities of central opioid modulation, have been found in PMS.7–10
Cognitive and Performance Aspects
Women who report a greater degree of symptoms around the time of menses have been found to have greater expectations of negative symptoms, to be more likely to have a mother or sister with expectations of perimenstrual distress, to have increased measures of depressed mood and to experience more stress in their lives than women with low premenstrual symptom scores.11,12 Suicidal ideas and death wishes and thoughts have been reported in as many as 10 percent of women with PMS.13 Women may mislabel their negative feelings as “PMS” because PMS may be a more socially acceptable diagnosis than depression or anxiety, or because they misunderstand the latter conditions. Some women may ascribe somatic symptoms to affective distress they cannot otherwise describe.14
Studies have shown that daily stressors are perceived by patients with PMS as more stressful premenstrually and less stressful postmenstrually than similar events experienced by women without symptoms of PMS.11
Although women frequently complain of decreased performance before or during menses, several studies have shown no decrease in task performance related to the menstrual cycle. Decreased mood and energy, however, have been documented premenstrually.14
Related Factors
Endocrine
As a major menstrual-cycle hormone, progesterone has long been thought to relate to premenstrual symptoms. Controlled studies of women with PMS, however, have failed to show consistent abnormalities in progesterone metabolism or consistent superiority of progesterone supplementation over placebo in single- and double-blind trials. Controlled trials of estrogen therapy in PDD have not shown positive effects.
Women with PMS have an increased variability in thyroid function measures, suggesting a disorder of the hypothalamic-pituitary-thyroid axis. However, no specific thyroid abnormality has been consistently documented in women with symptoms of PMS, but studies of this aspect are limited by small sample sizes.16
Nutrient Abnormalities
Deficiency in a number of vitamins, minerals and other nutrients may be associated with premenstrual distress. Deficiencies of magnesium, manganese, B vitamins, vitamin E and linoleic acid and its metabolites have been reported in women with PMS.19–23 In addition, dietary deficiencies including a low intake of calcium, magnesium and manganese have been described in women with menstrually related discomforts. Supplementation with these agents has been shown to improve several types of premenstrual symptoms.24 Dietary modification that raises the level of tryptophan, the essential amino acid precursor for serotonin, has been demonstrated to improve premenstrual affective symptoms. An excess in the dietary intake of simple carbohydrates has been associated with mood disturbances, edema and fatigue.25,26 A higher intake of dietary sodium is associated with edema, and women consuming more caffeine have been found to have more irritability than women with a limited caffeine intake.27 Some women, especially those with a family history of alcoholism, have been found to increase their alcohol consumption before menses.26,28
The essential fatty acid linoleic acid is required for the synthesis of certain prostaglandins.31 A defect in the conversion of linoleic acid to gammalinolenic acid has been suggested in women with PMS, and evening primrose oil, one of the few sources of gammalinolenic acid, has been reported to be effective in relieving symptoms.32
Perspective for Symptom Assessment and Successful Treatment
Patients with PMS may present with a wide variety of symptoms, which can seem overwhelming to the busy clinician. By narrowing the range of symptoms into symptom clusters (Table 1), an effective treatment approach can be developed.
The overall treatment goals for patients with PMS include amelioration of the patient's symptoms and functional impairment, and optimization of the patient's overall health. Successful treatment of PMS begins with a thorough assessment of the symptoms and their impact on daily life. PMS may be the label a woman has given to symptoms she does not understand. In one study, a majority of women who referred themselves to a PMS clinic met the diagnostic criteria for affective disorders, most commonly major depression or anxiety disorders.33
Applying research findings to the individual patient with PMS is problematic. First, entry criteria and outcome measures vary from trial to trial. Second, the placebo response in PMS studies has frequently been reported to be between 25 and 50 percent. Third, few trials have compared one treatment modality against another.1 However, most of the first-line treatments that are effective in reducing premenstrual symptoms also benefit general health and well-being, are inexpensive and have few significant side effects.34
The woman with PMS can be assured that improvement is virtually certain with a combination of several interventions. Consulting a knowledgeable and empathetic clinician who facilitates communication is the patient's first step in gaining relief. An explanation of the prevalence and treatability of premenstrual symptoms has itself been shown to be therapeutic. Women who adopt self-care measures that include dietary modification and moderate exercise have been reported to have a significant decrease in premenstrual symptoms, both short- and long-term.35,36
Women with premenstrual distress frequently consult practitioners in several disciplines. Alternative treatments are frequently used instead of, or along with, conventional therapies. A willingness to discuss and incorporate safe alternative treatments will help the physician establish a position of advocacy for the woman's total well-being.
A diagnostic test for PMS does not exist. Because thyroid disorders are common in women of reproductive age,37 measurement of serum thyroid-stimulating hormone may be useful. Other studies should be considered only as suggested by findings on the history and physical examination. Laboratory tests to detect specific nutrient deficiencies are not recommended because the usual tests do not accurately assess physiologic status.
Successful treatment begins by clarifying four areas in the patient's history: (1) what are the major presenting symptoms, (2) how does the timing of symptoms relate to the patient's menstrual cycle, (3) what symptoms are troubling to the patient and (4) what, if any, symptoms are unrelated to the menstrual cycle.
Major Presenting Symptoms
A checklist such as a premenstrual assessment form (Figure 1) has been shown to correlate well with more extensive research instruments.38 Significant affective symptoms should cue the clinician to perform an assessment of suicide risk. Having the patient repeat the symptom assessment during and after treatment can provide encouragement and documentation of improvement.
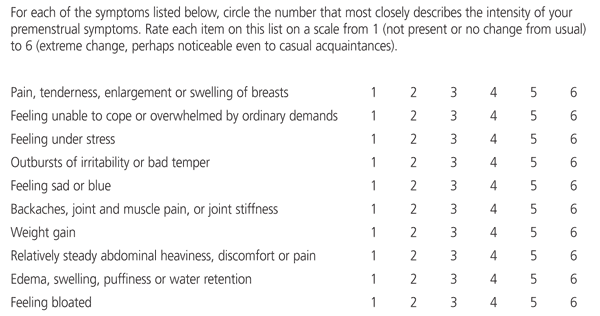
Timing of Symptoms
Determining whether the symptoms are confined to the luteal phase or are unrelated to the menstrual period is key to making an accurate diagnosis. If the patient has not definitely noted that her symptoms are exclusively confined to the luteal phase of her cycle, an association may be uncovered by having the patient complete a symptom calendar (Figure 2). If the symptoms are erratic or menstrual cycles are irregular, measurement of basal body temperature throughout the cycle may provide presumptive evidence of ovulation.

Level of Patient's Distress
The physician must clarify which symptoms are most distressing to the patient and then base the treatment plan on alleviation of her major distress. Some women may be more concerned that the pain represents infection, a risk of infertility or cancer than they are troubled by the pain itself. The patient may have concerns about the effect of her mood on family relationships and job performance, or on self-destructive impulses.39 Women with concerns about weight may engage in bingeing and purging behavior in response to food cravings.23
Symptoms Unrelated to PMS
Affective symptoms that are not confined to the luteal phase do not represent PDD and are more likely related to an affective disorder. A history of mood disorder, trauma, unresolved losses or ongoing threats of harm (e.g., violence, abandonment, incarceration, homelessness, hunger, job loss) raises the likelihood of an affective diagnosis.4,39 Some women experience premenstrual magnification of chronic emotional disorders.5 The Beck Depression Inventory can be useful in identifying women with symptoms more consistent with a diagnosis of depression.40
Primary Treatment Strategies
Providing Information
Along with supportive counseling by the clinician, excellent sources of patient information are available through the World Wide Web (Table 2). Patients also may be given advice about reliable self-help books that might be beneficial.
| Organization and its URL | Types of information |
|---|---|
| American Academy of Family Physicians https://www.aafp.org/healthinfo.html | Women's health and other patient education information |
| American Dietetic Association http://www.eatright.org/ | Nutrition resources, including “The Good News Guide for Healthy Women” |
| Food and Nutrition Information Center, U.S. Department of Agriculture http://www.fnic.nal.usda.gov/ | “Dietary Guidelines for Americans,” food guide pyramid and more |
| ShapeUp America! http://www.shapeup.org/ | Resources on exercise and weight management, body-mass index information |
| American Medical Association http://www.ama-assn.org/ama | Women's health and other patient education information |
| OBGYN.net http://www.obgyn.net./women/women.htm | Women's health and other patient education information |
Dietary Modification
The recommended dietary intake for the treatment of PMS conforms closely to dietary recommendations of the American Heart Association and the American Cancer Society. As advocates for women's health throughout the life span, clinicians are responsible for prescribing a general risk-reduction diet. Several evidence-based sources of diet information are widely available. Analysis of the woman's typical intake using a computerized dietary assessment program may help identify areas of excess or deficiency. Consultation with a registered dietitian may also be beneficial.
Exercise
Secondary Treatment Strategies
Secondary treatment modalities may be indicated if PMS symptoms do not sufficiently improve after two to three months of lifestyle modification, if clues to coexisting conditions are uncovered or if symptoms are moderate to severe. Because few direct comparative studies of drug versus nondrug therapies for PMS have been conducted, drug therapy should be reserved for use in patients who have insufficient improvement of PMS symptoms by other means.
Dietary Supplements
Numerous dietary supplements to relieve PMS have been tried, but very few have been subjected to rigorous scientific study. While this schema represents only part of the mechanism involved in the development of PMS, it is clear that many treatments can have beneficial effects. Interventions designed to ensure optimal intake of several nutrients are likely to be most successful. If dietary modification does not yield sufficient relief of symptoms, dietary supplementation may be tried. The amounts recommended in Table 3 are based on findings from existing clinical studies and on data regarding potential toxicity.24,29,30,41
| Nutrient | Recommended daily dosage |
|---|---|
| Calcium | 1,000 mg |
| Magnesium | 400 mg |
| Manganese | 6 mg |
| Vitamin E | 400 IU |
Nonsteroidal Anti-inflammatory Drugs
Several nonsteroidal anti-inflammatory drugs (NSAIDs) are effective for treatment of dysmenorrhea, and their use has been recommended for other perimenstrual discomforts.1 While NSAIDs may be effective for pain relief, nonselective inhibition of prostaglandin synthesis also blocks the beneficial effects of prostaglandins, including those on vascular function and gastric mucosal protection. NSAIDs should be prescribed for short-term use only and in the lowest effective dosages.
Antidepressants
Although several antidepressants have been shown to be effective for premenstrual symptoms, the lower side effect profile and efficacy data for the selective serotonin reuptake inhibitors support their use over other classes of antidepressants. Antidepressant therapy should be prescribed daily in the usual dosages for depression. Luteal-phase administration alone has been shown to improve only mood symptoms, not both affective and physical discomforts.7–9
Cognitive Behavioral Therapy
Patients with expectations of negative symptoms or of impaired performance around menses may respond well to cognitive therapy, with current knowledge regarding menstrually related disorders applied to the individual patient's situation.43 Women whose symptoms are severe or whose self-esteem is seriously impaired may require more extensive treatment from a therapist trained in cognitive behavioral methods.11,14 A reduction of symptoms for at least several months has been reported with therapy designed to reduce negative emotions through cognitive restructuring, enhanced problem-solving skills in personal and interpersonal negotiations and responsible assertiveness in managing obstacles, frustrations and discomfort in the context of difficult social situations.42 Relaxation therapy has also been successful.44
Hormonal Contraceptives
Hormonal contraceptive methods suppress ovulation; thus, combined oral contraceptive pills or a progestin-only contraceptive agent may provide relief of PMS. The low cost, effectiveness and reversibility of oral contraceptives favor their use. Efficacy of various contraceptive formulations for the treatment of PMS has not been compared.
Diuretics
Symptoms related to fluid retention can usually be eradicated through dietary measures, most specifically restriction of sodium and simple sugars. However, diuretics may be useful in patients with very troubling edema. Spironolactone has been demonstrated to be effective in a dosage of 100 mg per day.45
Treatments Not Generally Recommended
Progesterone. Multiple double-blind, placebo-controlled studies of progesterone have failed to show evidence of efficacy in PMS, and its use is not currently recommended.34
High-Dose Vitamin B6. Vitamin B6 has been used for the treatment of PMS for many years, but controlled trials have failed to document its effectiveness. Peripheral neuropathy has been reported with daily dosages of 200 mg or more.14,46 Alprazolam (Xanax). Several studies have examined the use of alprazolam for treatment of premenstrual symptoms.47 While some improvement has been reported with use of this drug, the studies were small and the magnitude of change was not greater than that reported for other therapies. Therefore, because of the potential for abuse of this drug, alprazolam should be avoided in the treatment of PMS.
Gonadotropin-Releasing Hormone Agonists or Antagonists. Several studies have reported success with the use of gonadotropin-releasing hormone agonists or antagonists. However, their expense and side effect profile, including hypoestrogenism and an increased risk of osteoporosis, along with the absence of data comparing their safety and efficacy with that of less drastic measures, would recommend against their use for PMS.
Hysterectomy or Endometrial Ablation. While uterine factors may have a role in the pathogenesis of PMS, insufficient data exist to recommend hysterectomy or endometrial ablation as treatments for this condition.
Final Comment
A metabolic predisposition for the development of severe premenstrual symptoms may exist in some women. Our understanding of PMS is increasing through ongoing research, and studies to date have confirmed the effectiveness of a healthy diet, moderate aerobic exercise and stress reduction through information and support (Table 4). If these behavioral modifications fail to relieve symptoms, pharmacologic treatment such as oral contraceptives may be considered.
| Information to help the patient understand what her symptoms most likely represent, and the goals and components of treatment |
| Assessment of the patient's current description and severity of symptoms for comparison at follow-up |
| Recommendations for dietary modification |
| Prescription of regular, moderate aerobic exercise within the context of her life and responsibilities |
| Counseling to enhance self-efficacy and self-management of distress |
| Clarifying mutual hopes and intentions, to facilitate reaching a therapeutic common ground |
Family physicians have an excellent opportunity to provide assistance in the context of a woman's multiple life roles and phases. As our understanding of this complex group of symptoms grows, we can assure each woman, with confidence, that her distress can be eased.