The American Cancer Society and other national medical organizations emphasize the need for routine screening for prostate cancer in men over the age of 50. The serum prostate-specific antigen (PSA) assay is the test most commonly recommended for the purpose of screening. However, when PSA screening is examined critically from the standpoint of the principles of screening, evidence from prospective studies to support the routine use of PSA testing is lacking. Data suggest that screening often detects what may be indolent, nonaggressive prostate cancer. The treatment of such a cancer with radiation or radical prostatectomy can result in significant morbidity, including urinary incontinence and impotence, without a proven decrease in mortality. Evidence from randomized clinical trials in support of routine PSA screening is urgently needed.
The purpose of screening tests is to identify disease in asymptomatic persons at a stage when treatment will alter the natural history of the condition. There is now a widespread call to incorporate routine prostate-specific antigen (PSA) screening for prostate cancer into the care of men over age 50. However, little prospective evidence shows that the benefits of such testing outweigh the risks. The ideal way to assess the efficacy of screening is with prospective randomized clinical trials; unfortunately, no such trial of prostate cancer screening has been conducted.
Evidence Favoring Prostate Cancer Screening
Although screening for prostate cancer generally includes both a digital rectal examination and a serum PSA assay, use of the PSA level has thrust prostate cancer screening into the center of controversy. A number of descriptive studies of screening have shed much light on the question of what we can expect from prostate cancer screening by means of the PSA assay. One of the largest studies reported to date was supported by Hybritech, Inc., the manufacturer of one of the leading PSA assays.1 The study reported the results of screening with both PSA and digital rectal examination in 17,157 white men and 804 black men. Most of these men were 50 to 69 years of age. Table 1 shows the rates of positive results on screening tests and the incidence of prostate cancer in the study population. Clearly, screening detects prostate cancer in a significant number of asymptomatic men.
The prognosis of prostate cancer is related principally to the stage and grade of the disease at the time of diagnosis. Stage refers to the extent of spread of the disease (i.e., whether the cancer is confined to the gland, locally invasive or metastatic). Treatment decisions must be based on the clinical stage, with knowledge of the spread of the disease limited to the information available from the tests that can be performed at the time of diagnosis. In the Hybritech study,1 over 90 percent of the cancers detected in both blacks and whites were considered to be clinically localized at the time of diagnosis. This rate of localized disease is dramatically higher than the rate found when prostate cancer is detected in ways other than screening. Thus, with prostate cancer screening, we see what is called a “stage shift”: more cancers are detected at a clinically localized stage.
Grade refers to the histologic type, characterized by the Gleason score, with the prognosis worse for higher-grade cancers. Prostate cancers are often classified into three grades. Grade III includes those with a Gleason score of more than 7, grade II includes those with a Gleason score of 5 to 7, and grade I includes those with a Gleason score of less than 5. In the Hybritech study,1 the Gleason score was less than 7 in 60.0 percent of the black men and in 75.1 percent of the white men who were found to have prostate cancer. Patients with clinically localized cancer of a lower grade are considered to be the best candidates for curative treatment, especially radical prostatectomy. Thus, a substantial majority of cancers detected by screening in the Hybritech study were thought to be potentially curable at the time of diagnosis.
Clinical outcomes after surgery for localized prostate cancer are known to be good.2,3 A recent multi-institutional study that pooled the results of radical prostatectomy in men with clinically localized prostate cancer found disease-specific 10-year survival rates of 94 percent for grade I cancer, 80 percent for grade II cancer and 77 percent for grade III cancer.2 The high proportion of prostate cancers that are clinically localized when detected by screening, coupled with the high disease-specific survival rates after radical prostatectomy, forms the basis for the recommendation for mass screening for prostate cancer. However, when this evidence is subjected to scrutiny using the principles of screening, the argument in favor of prostate cancer screening is well short of convincing.
Argument Against Prostate Cancer Screening
Do Morbidity and Mortality from the Disease Justify Screening?
One must view the evidence favoring prostate cancer screening in the context of the general principles of screening.4 The first principle is that the morbidity and mortality from the disease must be sufficiently prevalent to justify the screening effort and expense. Prostate cancer is the second leading cause of cancer death in men in the United States.5 In addition, significant morbidity is associated with end-stage disease. A safe, cost-effective approach to reducing the number of deaths from prostate cancer would seem to be a welcome addition to our efforts to improve health.
The morbidity and mortality resulting from prostate cancer, however, are principally problems of the elderly. Figure 1 shows the age distribution of deaths caused by prostate cancer.5 Only 8.5 percent of the deaths occur in men younger than 65 years of age; 63.1 percent of the deaths occur in men 75 years of age and older. The popular press would lead one to believe that prostate cancer is often an aggressive, fatal disease in 50-year-old men, but the truth is quite different.
FIGURE 1.
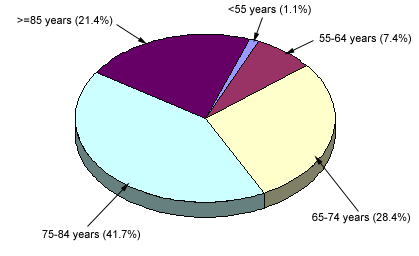
Age distribution of deaths from prostate cancer in the United States.
Although a reduction in the mortality rate from prostate cancer in the elderly may be worthwhile, competing causes of mortality are prevalent in the elderly population. A reduction in all-cause mortality, resulting in an actual increase in life expectancy, will be difficult to accomplish in this age group. Advocates of prostate cancer screening suggest that an alternate cause of death is preferable to death from prostate cancer. Yet nothing suggests that men spared death from prostate cancer by early detection and treatment experience a less difficult death from another cause.
Does the Natural History of the Disease Justify Screening?
The next principle of screening is that the natural history of the disease must be well known, and an asymptomatic phase of sufficient duration must exist to allow detection by screening at reasonable intervals. Autopsy studies and analyses of pathology specimens derived from prostate surgery for apparently benign disease, as well as case series of screening, certainly indicate that asymptomatic prostate cancer exists. However, prostate cancer is not biologically one disease: great variation exists in the aggressiveness of different tumors, with some cancers being quite aggressive and resulting in sickness and death soon after detection and others being indolent. The natural history from the development of prostate cancer to the occurrence of symptoms and, eventually, death is not well known for different types of prostate cancer.
Although prostate cancer likely has a spectrum of aggressiveness, the ends of this spectrum shed light on the unreliability of data suggesting benefits from earlier detection through screening. In the natural history of two biologically different prostate cancers, the time from the development of disease to the onset of symptoms is quite prolonged in indolent prostate cancer but short in aggressive disease. Thus, indolent cancer is likely to be detected by screening before the onset of symptoms, but aggressive cancer may not have an asymptomatic phase long enough to allow detection by screening before symptoms are manifested.
The stage shift in prostate cancers detected by screening is at least in part due to the bias toward detection of biologically indolent tumors, which are associated with a good prognosis, rather than detection of aggressive tumors with a poor prognosis, which generally are detected after the onset of symptoms. In the extreme, this detection bias means that prostate cancer screening detects tumors that would never have reached the symptomatic stage, much less result in mortality. A competing cause of mortality, such as heart disease, would have resulted in the patient's death before prostate cancer became symptomatic. Much evidence suggests that the current push for prostate cancer screening is leading to the diagnosis and treatment of many cases of prostate cancer that would otherwise have remained harmless.
The longitudinal Physician's Health Study6 included men whose age ranges were comparable to those in the Hybritech study. The participants in the Physician's Health Study were followed prospectively, with no specific efforts at prostate cancer screening. The cumulative incidence of prostate cancer four years after enrollment was 0.5 percent and reached only 2.5 percent after 10 years. This incidence is in stark contrast to the overall 3.3 percent incidence with a single screening examination in the Hybritech study. The difference in incidence rates between the two studies strongly suggests that asymptomatic prostate cancer was present at baseline in some of the men in the Physician's Health Study but still had not been detected 10 years later.
Figure 2 shows the incidence of prostate cancer reported by the Surveillance, Epidemiology, and End Results (SEER) Program from 1974 to 1994.5 The incidence has nearly doubled since the introduction of PSA screening in the late 1980s, consistent with what would be expected from a comparison of the incidence rates in the Physicians's Health Study and those in the Hybritech study. In all likelihood, many cancers that have been detected by PSA screening would never have become symptomatic in the patient's lifetime.
FIGURE 2.
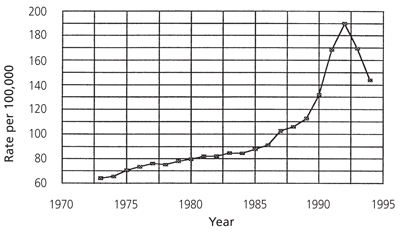
Prostate cancer incidence from 1973 to 1994, showing a dramatic increase in incidence since the introduction of prostate-specific antigen testing. The reason for the decline in incidence in 1994 is unknown. It may reflect a reversal in the upward trend in the number of men being screened for the first time or a low incidence of cancer in men receiving serial screening.
Do Sensitivity, Specificity and Cost-Effectiveness Justify Screening?
The next principle of screening is that a cost-effective, acceptable screening test with reasonable sensitivity and specificity must exist to allow detection in the asymptomatic phase. PSA screening does reasonably well in fulfilling this standard, although the percentage of cases detected (sensitivity) by PSA screening is not as high as one might expect. A true measure of sensitivity requires a gold standard: there must be another means of knowing whether cancer exists at the time the test is done. This is not possible with prostate cancer, since the definitive presence or absence of cancer can only be established by removal and pathologic examination of the prostate gland. Data from the Physician's Health Study6 provide an approximation of sensitivity by an analysis of the baseline serum PSA levels to determine how many persons who subsequently developed prostate cancer had an abnormal PSA level at baseline. Of those men with cancers diagnosed in the first four years after baseline, 75 percent had elevated PSA levels at baseline.
Does the Outcome with Early Intervention Justify Screening?
The last, and most troublesome, criterion for screening is that treatment in the asymptomatic phase must alter the natural history of the disease, and any morbidity and mortality caused by the detection and treatment of the disease must be clearly offset by reductions in morbidity and mortality from the disease. Unfortunately, reliable data about the effects of various treatments for prostate cancer are not available. Radical prostatectomy is the standard practice in the United States for the treatment of clinically localized prostate cancer.
The gold standard for the study of treatment of any disease is a randomized clinical trial; only one such trial has compared radical prostatectomy with conservative management.7 No difference in outcomes was demonstrated, but the sample size was inadequate as the basis for any firm conclusions. The best possible analysis is to examine the outcomes associated with different treatments selected in a case series fashion. However, the treatments selected would likely not be random and the comparison would likely include patients with different characteristics for which no data exist.
One specific bias will make results of radical prostatectomy appear more favorable than other treatments.8 It is well known that clinical staging is not accurate when compared with pathologic staging. About 50 percent of men whose disease is considered to be clinically localized prove to have extension beyond the prostate gland at the time of surgery and pathologic examination.9 A significant minority will in fact already have metastatic disease. Before radical prostatectomy is undertaken, a pelvic lymph node dissection is often performed and, if positive nodes are found, the prostatectomy is cancelled. Therefore, in case series reporting the outcomes after prostatectomy for clinically localized disease, many men with occult metastases are not included. Such is not the case with any other treatment, nor with conservative management, where those with occult metastases remain in the treated group.
A recent study sought to alleviate this problem by analyzing outcomes on the basis of intention to treat, thus including in the prostatectomy group those patients scheduled for prostatectomy but not undergoing the operation because positive nodes were found at the time of surgery.8 This study was also based on population, rather than on data gathered at one or a few special centers. Thus, it provides the best data we have for the expectations of conservative management, radiation therapy and prostatectomy.
Table 2 summarizes the results of this study.8 One of the most critical findings is that 10-year disease-specific survival in patients with clinically localized grade I or grade II prostate cancer is quite good with conservative management (i.e., no treatment unless the disease becomes symptomatic). This finding is supported by Swedish data showing good outcomes in men who chose conservative management.10 Although the data do not necessarily signify that survival cannot be improved with surgery or radiation therapy, they do suggest that there is little room for such improvement to occur. The data show that treatment offers no benefit for patients with grade I cancer and only marginal benefit for those with grade II or grade III cancer. If other factors that might influence survival were present but not reported, it is likely that a disproportionate number of men with adverse characteristics would be present in the conservatively managed group. Healthier men would be more likely to be selected for surgery.
In contrast to the limited data supporting the benefits of prostatectomy, the risks are well delineated. Analysis of Medicare claims data11 revealed a 30-day mortality rate of 0.5 percent, with no specific trend toward increasing mortality with increasing age. A significant number of men can be expected to die prematurely as a direct result of prostate cancer screening and prostatectomy.
Morbidity is also high after prostatectomy, largely because of urinary incontinence and sexual dysfunction. A national survey12 of 721 men who underwent radical prostatectomy revealed that 31 percent had incontinence that required the use of pads or clamps; a comparable percentage had some dripping but did not use any devices. Only 21 percent reported having an erection in the past month; an additional 16 percent had an erection postsurgery but not in the past month. Before surgery, 92 percent reported having erections. Current surgical techniques in specialized centers may improve these rates, but even a 50 percent reduction in morbidity would still leave significant surgical risks.
Clearly, the treatment of prostate cancer has the potential to cause harm. Assuming a 4 percent rate of detection and subsequent prostatectomy as a result of screening, one in 5,000 men screened can be expected to die, one in 81 will require the use of pads or clamps because of incontinence after the surgery, and one in 36 will develop new sexual dysfunction.
Final Comment
It is very likely that the current rush to screen for prostate cancer cannot be easily curtailed. The experience with lung cancer screening is remarkably analogous.13 A stage shift associated with the early detection of lung cancer by radiographic screening was noted. Outcomes were known to be better with treatment of early-stage disease and, as a result, a national campaign for lung cancer screening was undertaken. Randomized trials of screening were performed, but it took 30 years to sway the opinions of the experts who advocated screening before reliable evidence was available. Thus, one must hope for developments in reliable clinical staging of prostate cancer so that better selection of candidates for surgery is possible. Also needed are advances in treatment modalities that significantly reduce the harm often imposed by currently available approaches.
The official clinical guideline of the American Academy of Family Physicians14 recommends counseling men between the ages of 50 and 65 about the known risks and uncertain benefits of screening for prostate cancer. This certainly seems a prudent course to follow. Men deserve sufficient information to allow them to make an informed, personal decision.

