A number of pitfalls can be encountered in the interpretation of common blood liver function tests. These tests can be normal in patients with chronic hepatitis or cirrhosis. The normal range for aminotransferase levels is slightly higher in males, nonwhites and obese persons. Severe alcoholic hepatitis is sometimes confused with cholecystitis or cholangitis. Conversely, patients who present soon after passing common bile duct stones can be misdiagnosed with acute hepatitis because aminotransferase levels often rise immediately, but alkaline phosphatase and γ-glutamyltransferase levels do not become elevated for several days. Asymptomatic patients with isolated, mild elevation of either the unconjugated bilirubin or the γ-glutamyltransferase value usually do not have liver disease and generally do not require extensive evaluation. Overall hepatic function can be assessed by applying the values for albumin, bilirubin and prothrombin time in the modified Child-Turcotte grading system.
The commonly used liver function tests (LFTs) primarily assess liver injury rather than hepatic function. Indeed, these blood tests may reflect problems arising outside the liver, such as hemolysis (elevated bilirubin level) or bone disease (elevated alkaline phosphatase [AP] level).
Abnormal LFTs often, but not always, indicate that something is wrong with the liver, and they can provide clues to the nature of the problem. However, normal LFTs do not always mean that the liver is normal. Patients with cirrhosis and bleeding esophageal varices can have normal LFTs. Of the routine LFTs, only serum albumin, bilirubin and prothrombin time (PT) provide useful information on how well the liver is functioning.
The general subject of LFTs1,2 and the differential diagnosis of abnormal LFTs in asymptomatic patients3–5 have been well reviewed. This article discusses some common pitfalls in the interpretation of LFTs. Hints for interpreting these tests are presented in Table 1.
TABLE 1 Helpful Hints for Interpreting Liver Function Tests
| Situation | Comments |
|---|---|
| Mildly elevated ALT level (less than 1.5 times normal) | ALT value could be normal for gender, ethnicity or body mass index. |
| Consider muscle injury or myopathy. | |
| Alcoholic hepatitis | Laboratory values can appear cholestatic, and symptoms can mimic cholecystitis. |
| Minimal elevations of AST and ALT often occur. | |
| AST level greater than 500 U per L | The AST elevation is unlikely to result from alcohol intake alone. |
| In a heavy drinker, consider acetaminophen toxicity. | |
| Common bile duct stone | Condition can simulate acute hepatitis |
| AST and ALT become elevated immediately, but elevation of AP and GGT is delayed. | |
| Isolated elevation of GGT level | This situation may be induced by alcohol and aromatic medications, usually with no actual liver disease. |
| Isolated elevation of AP level (asymptomatic patient with normal GGT level) | Consider bone growth or injury, or primary biliary cirrhosis. |
| AP level rises in late pregnancy. | |
| Isolated elevation of unconjugated bilirubin level | Consider Gilbert syndrome or hemolysis. |
| Low albumin level | Low albumin is most often caused by acute or chronic inflammation, urinary loss, severe malnutrition or liver disease; it is sometimes caused by gastrointestinal loss (e.g., colitis or some uncommon small bowel disease). |
| Normal values are lower in pregnancy. | |
| Blood ammonia level | Blood ammonia values are not necessarily elevated in patients with hepatic encephalopathy. |
| Determination of blood ammonia levels is most useful in patients with altered mental status of new onset or unknown origin. |
ALT = alanine aminotransferase; AST = aspartate aminotransferase; AP = alkaline phosphatase; GGT = γ-glutamyltransferase.
Markers of Hepatocellular Injury
The most commonly used markers of hepatocyte injury are aspartate aminotransferase (AST, formerly serum glutamic-oxaloacetic transaminase [SGOT]) and alanine aminotransferase (ALT, formerly serum glutamate-pyruvate transaminase [SGPT]). While ALT is cytosolic, AST has both cytosolic and mitochondrial forms.
Hepatocyte necrosis in acute hepatitis, toxic injury or ischemic injury results in the leakage of enzymes into the circulation. However, in chronic liver diseases such as hepatitis C and cirrhosis, the serum ALT level correlates only moderately well with liver inflammation. In hepatitis C, liver cell death occurs by apoptosis (programmed cell death) as well as by necrosis. Hepatocytes dying by apoptosis presumably synthesize less AST and ALT as they wither away. This probably explains why at least one third of patients infected with hepatitis C virus have persistently normal serum ALT levels despite the presence of inflammation on liver biopsy.6,7 Patients with cirrhosis often have normal or only slightly elevated serum AST and ALT levels. Thus, AST and ALT lack some sensitivity in detecting chronic liver injury. Of course, AST and ALT levels tend to be higher in cirrhotic patients with continuing inflammation or necrosis than in those without continuing liver injury.
As markers of hepatocellular injury, AST and ALT also lack some specificity because they are found in skeletal muscle. Levels of these aminotransferases can rise to several times normal after severe muscular exertion or other muscle injury, as in polymyositis,8 or in the presence of hypothyroidism, which can cause mild muscle injury and the release of aminotransferases. In fact, AST and ALT were once used in the diagnosis of myocardial infarction.
Slight AST or ALT elevations (within 1.5 times the upper limits of normal) do not necessarily indicate liver disease. Part of this ambiguity has to do with the fact that unlike the values in many other biochemical tests, serum AST and ALT levels do not follow a normal bell-shaped distribution in the population.9 Instead, AST and ALT values have a skewed distribution characterized by a long “tail” at the high end of the scale (Figure 1).5 For example, the mean values for ALT are very similar from one population to another, but the degree to which the distribution is skewed varies by gender and ethnicity. The ALT distributions in males and nonwhites (i.e., blacks and Hispanics) tend to have a larger tail at the high end, so that more values fall above the upper limits of normal set for the average population.10,11
FIGURE 1. Typical ALT or AST Distribution
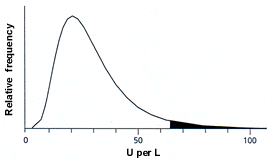
Typical population distribution of serum alanine aminotransferase (ALT) or aspartate aminotransferase (AST) levels. The population distributions for these aminotransferases do not follow a bell-shaped curve but are skewed, with a long “tail” at higher values. By convention, values above the 97.5th percentile are considered elevated (shaded area). Although average values for ALT and AST are similar in different populations, the distributions are more skewed in males and in nonwhite ethnic groups (i.e., blacks and Hispanics).
Figure 1 adapted with permission from Quinn PG, Johnston DE. Detection of chronic liver disease: costs and benefits. Gastroenterologist 1997;5:58–77.
AST and ALT values are higher in obese patients, probably because these persons commonly have fatty livers.12 ALT levels have been noted to decline with weight loss.13 Depending on the physician's point of view, the upper limits of normal for AST and ALT levels could be set higher for more obese persons.
Rare individuals have chronically elevated AST levels because of a defect in clearance of the enzyme from the circulation.14 For both AST and ALT, the average values and upper limits of normal in patients undergoing renal dialysis are about one half of those found in the general population.15 Mild elevations of ALT or AST in asymptomatic patients can be evaluated efficiently by considering alcohol abuse, hepatitis B, hepatitis C and several other possible diagnoses (Table 2).5
Various liver diseases are associated with typical ranges of AST and ALT levels (Figure 2). ALT levels often rise to several thousand units per liter in patients with acute viral hepatitis. The highest ALT levels—often more than 10,000 U per L—are usually found in patients with acute toxic injury subsequent to, for example, acetaminophen overdose or acute ischemic insult to the liver. AST and ALT levels usually fall rapidly after an acute insult.
FIGURE 2. Typical AST or ALT Values in Disease
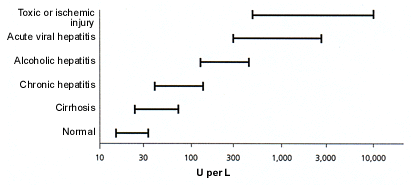
Typical serum aspartate aminotransferase (AST) or alanine aminotransferase (ALT) values for various diseases. Note that the horizontal axis is a log scale. As shown, the amino-trans-ferases are often normal in patients with cirrhosis. In patients with uncomplicated alcoholic hepatitis, the AST value is rarely greater than 500 U per L and is usually no more than 200 to 300 U per L. The highest peak aminotransferase values are found in patients with acute ischemic or toxic liver injury.
Lactate dehydrogenase (LDH) is less specific than AST and ALT as a marker of hepatocyte injury. However, it is worth noting that LDH is disproportionately elevated after an ischemic liver injury.16
It is especially important to remember that in patients with acute alcoholic hepatitis, the serum AST level is almost never greater than 500 U per L and the serum ALT value is almost never greater than 300 U per L. The reasons for these limits on AST and ALT elevations are not well understood. In typical viral or toxic liver injury, the serum ALT level rises more than the AST value, reflecting the relative amounts of these enzymes in hepatocytes. However, in alcoholic hepatitis, the ratio of AST to ALT is greater than 1 in 90 percent of patients and is usually greater than 2.17 The higher the AST-to-ALT ratio, the greater the likelihood that alcohol is contributing to the abnormal LFTs. In the absence of alcohol intake, an increased AST-to-ALT ratio is often found in patients with cirrhosis.
The elevated AST-to-ALT ratio in alcoholic liver disease results in part from the depletion of vitamin B6 (pyridoxine) in chronic alcoholics.18 ALT and AST both use pyridoxine as a coenzyme, but the synthesis of ALT is more strongly inhibited by pyridoxine deficiency than is the synthesis of AST. Alcohol also causes mitochondrial injury, which releases the mitochondrial isoenzyme of AST.
Patients with alcoholic hepatitis can present with jaundice, abdominal pain, fever and a minimally elevated AST value, thereby leading to a misdiagnosis of cholecystitis. This is a potentially fatal mistake given the high surgical mortality rate in patients with alcoholic hepatitis.19
Markers of Cholestasis
Cholestasis (lack of bile flow) results from the blockage of bile ducts or from a disease that impairs bile formation in the liver itself. AP and γ-glutamyltransferase (GGT) levels typically rise to several times the normal level after several days of bile duct obstruction or intrahepatic cholestasis. The highest liver AP elevations—often greater than 1,000 U per L, or more than six times the normal value—are found in diffuse infiltrative diseases of the liver such as infiltrating tumors and fungal infections.
Diagnostic confusion can occur when a patient presents within a few hours after acute bile duct obstruction from a gallstone. In this situation, AST and ALT levels often reach 500 U per L or more in the first hours and then decline, whereas AP and GGT levels can take several days to rise.
Both AP and GGT levels are elevated in about 90 percent of patients with cholestasis.20 The elevation of GGT alone, with no other LFT abnormalities, often results from enzyme induction by alcohol or aromatic medications in the absence of liver disease. The GGT level is often elevated in persons who take three or more alcoholic drinks (45 g of ethanol or more) per day.21 Thus, GGT is a useful marker for immoderate alcohol intake. Phenobarbital, phenytoin (Dilantin) and other aromatic drugs typically cause GGT elevations of about twice normal. A mildly elevated GGT level is a typical finding in patients taking anticonvulsants and by itself does not necessarily indicate liver disease.22,23
Serum AP originates mostly from liver and bone, which produce slightly different forms of the enzyme. The serum AP level rises during the third trimester of pregnancy because of a form of the enzyme produced in the placenta. When serum AP originates from bone, clues to bone disease are often present, such as recent fracture, bone pain or Paget's disease of the bone (often found in the elderly). Like the GGT value, the AP level can become mildly elevated in patients who are taking phenytoin.22,23
If the origin of an elevated serum AP level is in doubt, the isoenzymes of AP can be separated by electrophoresis. However, this process is expensive and usually unnecessary because an elevated liver AP value is usually accompanied by an elevated GGT level, an elevated 5′-nucleotidase level and other LFT abnormalities.
In one study,24 isolated AP elevations were evaluated in an unselected group of patients at a Veterans Affairs hospital. Most mild AP elevations (less than 1.5 times normal) resolved within six months, and almost all greater elevations had an evident cause that was found on routine clinical evaluation.
Persistently elevated liver AP values in asymptomatic patients, especially women, can be caused by primary biliary cirrhosis, which is a chronic inflammatory disorder of the small bile ducts. Serum antimitochondrial antibody is positive in almost all of these patients.
Indicators of How Well the Liver Functions
BILIRUBIN
Bilirubin results from the enzymatic breakdown of heme. Unconjugated bilirubin is conjugated with glucuronic acid in hepatocytes to increase its water solubility and is then rapidly transported into bile. The serum conjugated bilirubin level does not become elevated until the liver has lost at least one half of its excretory capacity. Thus, a patient could have obstruction of either the left or right hepatic duct without a rise in the bilirubin level.
Because the secretion of conjugated bilirubin into bile is very rapid in comparison with the conjugation step, healthy persons have almost no detectable conjugated bilirubin in their blood. Liver disease mainly impairs the secretion of conjugated bilirubin into bile. As a result, conjugated bilirubin is rapidly filtered into the urine, where it can be detected by a dipstick test. The finding of bilirubin in urine is a particularly sensitive indicator of the presence of an increased serum conjugated bilirubin level.
In many healthy persons, the serum unconjugated bilirubin is mildly elevated to a concentration of 2 to 3 mg per dL (34 to 51 μmol per L) or slightly higher, especially after a 24-hour fast. If this is the only LFT abnormality and the conjugated bilirubin level and complete blood count are normal, the diagnosis is usually assumed to be Gilbert syndrome, and no further evaluation is required. Gilbert syndrome was recently shown to be related to a variety of partial defects in uridine diphosphate-glucuronosyl transferase, the enzyme that conjugates bilirubin.25
Mild hemolysis, such as that caused by hereditary spherocytosis and other disorders, can also result in elevated unconjugated bilirubin values, but hemolysis is not usually present if the hematocrit and blood smear are normal. The presence of hemolysis can be confirmed by testing other markers, such as haptoglobin, or by measuring the reticulocyte count.
Severe defects in bilirubin transport and conjugation can lead to markedly elevated unconjugated bilirubin levels, which can cause serious neurologic damage (kernicterus) in infants. However, no serious form of liver disease in adults causes elevation of unconjugated bilirubin levels in the blood without also causing elevation of conjugated bilirubin values.
When a patient has prolonged, severe biliary obstruction followed by the restoration of bile flow, the serum bilirubin level often declines rapidly for several days and then slowly returns to normal over a period of weeks. The slow phase of bilirubin clearance results from the presence of delta-bilirubin, a form of bilirubin chemically attached to serum albumin.26 Because albumin has a half-life of three weeks, delta-bilirubin clears much more slowly than bilirubin-glucuronide. Clinical laboratories can measure delta-bilirubin concentrations, but such measurements are usually unnecessary if the physician is aware of the delta-bilirubin phenomenon.
ALBUMIN
Although the serum albumin level can serve as an index of liver synthetic capacity, several factors make albumin concentrations difficult to interpret.27 The liver can synthesize albumin at twice the healthy basal rate and thus partially compensate for decreased synthetic capacity or increased albumin losses. Albumin has a plasma half-life of three weeks; therefore, serum albumin concentrations change slowly in response to alterations in synthesis. Furthermore, because two thirds of the amount of body albumin is located in the extravascular, extracellular space, changes in distribution can alter the serum concentration.
In practice, patients with low serum albumin concentrations and no other LFT abnormalities are likely to have a nonhepatic cause for low albumin, such as proteinuria or an acute or chronic inflammatory state. Albumin synthesis is immediately and severely depressed in inflammatory states such as burns, trauma and sepsis, and it is commonly depressed in patients with active rheumatic disorders or severe end-stage malnutrition. In addition, normal albumin values are lower in pregnancy.
PROTHROMBIN TIME
The liver synthesizes blood clotting factors II, V, VII, IX and X. The prothrombin time (PT) does not become abnormal until more than 80 percent of liver synthetic capacity is lost. This makes PT a relatively insensitive marker of liver dysfunction. However, abnormal PT prolongation may be a sign of serious liver dysfunction. Because factor VII has a short half-life of only about six hours, it is sensitive to rapid changes in liver synthetic function. Thus, PT is very useful for following liver function in patients with acute liver failure.
An elevated PT can result from a vitamin K deficiency. This deficiency usually occurs in patients with chronic cholestasis or fat malabsorption from disease of the pancreas or small bowel. A trial of vitamin K injections (e.g., 5 mg per day administered subcutaneously for three days) is the most practical way to exclude vitamin K deficiency in such patients. The PT should improve within a few days.
BLOOD AMMONIA
Measurement of the blood ammonia concentration is not always useful in patients with known or suspected hepatic encephalopathy. Ammonia contributes to hepatic encephalopathy; however, ammonia concentrations are much higher in the brain than in the blood and therefore do not correlate well.28 Furthermore, ammonia is not the only waste product responsible for encephalopathy. Thus, blood ammonia concentrations show only a mediocre correlation with the level of mental status in patients with liver disease. It is not unusual for the blood ammonia concentration to be normal in a patient who is in a coma from hepatic encephalopathy.
Blood ammonia levels are best measured in arterial blood because venous concentrations can be elevated as a result of muscle metabolism of amino acids. Blood ammonia concentrations are most useful in evaluating patients with stupor or coma of unknown origin. It is not necessary to evaluate blood ammonia levels routinely in patients with known chronic liver disease who are responding to therapy as expected.
Grading Liver Function by Child-Turcotte Class
In communicating among themselves, many physicians use the Child-Turcotte class as modified by Pugh, often termed the “Child class,” to convey information about overall liver function and prognosis (Table 3).29 This grading system can be used to predict overall life expectancy and surgical mortality in patients with cirrhosis and other liver diseases.30
For elective general abdominal surgery, perioperative mortality is in the neighborhood of several percent for patients who fall into the Child class A, 10 to 20 percent for those in class B and approximately 50 percent for those in class C.31 These percentages must be balanced by prognostic considerations when transplantation becomes an option. The presence of cirrhosis by itself is not an indication for liver transplantation, and transplantation is rarely performed in patients who fall into Child class A. For example, the 10-year survival rate is as high as 80 percent in patients with hepatitis C and cirrhosis who have Child class A liver function and no variceal bleeding.32 However, once patients with any type of liver disease fall into the Child-Turcotte class B or class C category, survival is significantly reduced and transplantation should be considered.

