The diagnosis of acute human immunodeficiency virus (HIV) syndrome requires a high index of suspicion and proper laboratory testing. Patients with the syndrome may have fever, fatigue, rash, pharyngitis or other symptoms. Primary HIV infection should be considered in any patient with possible HIV exposure who presents with fever of unknown cause. The diagnosis is based on a positive HIV-1 RNA level (more than 50,000 copies per mL) in the absence of a positive enzyme-linked immunosorbent antibody assay (ELISA) and confirmatory Western blot antibody test for HIV. Early diagnosis permits patient education as well as treatment that may delay disease progression. Triple-combination antiretroviral therapy should be started immediately and continued indefinitely. Compliance with medication regimens is essential to maximize benefit and discourage the development of viral resistance.
In the spring of 1986, a 24-year-old bisexual man presented with fever, markedly swollen cervical nodes and an exudative tonsillitis. He had dyspnea and cough with a normal chest radiograph. No pathogens were identified. It was thought that he might have the new disease called “acquired immunodeficiency syndrome” (AIDS), but a human immunodeficiency virus (HIV) antibody test was negative. He was treated for possible Pneumocystis carinii pneumonia, and his symptoms resolved in the ensuing two weeks. Three months later, he tested positive for HIV-1 antibody on enzyme-linked immunosorbent assay (ELISA), and the finding was confirmed by a Western blot test. He progressed to AIDS and died within 10 years.
Today this man's presentation would be recognized as acute HIV syndrome. It is now known that 50 to 90 percent of patients acutely infected with HIV experience at least some symptoms of the acute retroviral syndrome.1 The most common findings of the syndrome are fever (80 to 90 percent), fatigue (70 to 90 percent), rash (40 to 80 percent), headache (32 to 70 percent) and lymphadenopathy (40 to 70 percent). Additional findings include pharyngitis, myalgia, nausea, vomiting, diarrhea and other symptoms2 (Table 1).3 Symptoms usually develop within days to weeks after HIV exposure and last from a few days to several months, but usually less than 14 days.3
The differential diagnosis of the early findings of acute HIV infection can be confusing. Because of a low index of suspicion, the diagnosis is missed in as many as 75 percent of patients.3 Acute HIV infection may resemble infectious mononucleosis, influenza, severe streptococcal pharyngitis, viral hepatitis, toxoplasmosis or even secondary syphilis (Table 2). For example, a patient with symptoms suggestive of HIV infection could actually be an HIV-negative young adult manifesting for the first time the effects of a primary immunodeficiency disease involving B-cell and/or T-cell dysfunction. (Immunoglobulin A deficiency—by far the most common immunodeficient condition—often presents after the age of 21 years.4) On the other hand, a patient who returns from the tropics with an unexplained febrile illness is frequently assumed to have malaria, typhoid, schistosomiasis, filariasis or an intestinal helminthic infection but may actually have HIV infection. One study found that 3 percent of such travelers were infected with HIV.5 In areas where AIDS and malaria are endemic, 23 percent of fevers are ultimately attributable to HIV infection and only 15 percent to malaria.6 Age is also a factor, with the diagnosis of acute HIV infection often delayed in patients over 50 years old.7
TABLE 2 Differential Diagnosis of Acute HIV Infection
| Primary cytomegalovirus infection |
| Drug reaction |
| Epstein-Barr virus mononucleosis |
| Viral hepatitis |
| Primary herpes simplex virus infection |
| Influenza |
| Severe (streptococcal) pharyngitis |
| Secondary syphilis |
| Toxoplasmosis |
| Rubella |
| Brucellosis |
| Malaria |
HIV = human immunodeficiency virus.
Primary care physicians will be the first to encounter many of the new cases of HIV infection. Because the source person usually will not have appeared to be ill during contact, the newly presenting symptomatic patient will not recognize the possibility of having acquired HIV infection. However, when a patient presents with fever and fatigue, a sexually transmitted disease or a suggestive symptom complex, the possibility of HIV exposure or high-risk behavior should be reviewed.
Besides obviously risky behavior such as unprotected sex (vaginal, oral or anal) and intravenous illicit drug use (involving shared needles or a common “cooker” into which several syringes are dipped), less obvious sources of HIV infection should be investigated. Young athletes have been known to inject testosterone using a shared syringe. The shared, sharpened plastic “straw” used to sniff cocaine may transmit HIV by way of the nasal mucosa. Body piercing and tattooing may increase the risk of HIV exposure. Occupational exposure is a possible but rare source of HIV infection in health care workers.
Diagnosing Acute HIV Infection
Blood taken during the acute phase of HIV infection (days to weeks after exposure) may show lymphopenia and thrombocytopenia, but atypical lymphocytes are infrequent. The CD4 count usually remains normal. The HIV-1 antibody tests (ELISA and Western blot test), the only tests officially used to diagnose established HIV infection, do not become positive until three or four weeks (sometimes even months) after the infection is acquired.
On the other hand, the quantitative plasma HIV-1 RNA level (viral load) by polymerase chain reaction (PCR), which is 95 to 98 percent sensitive for HIV,8 becomes positive within 11 days of infection.9 During the symptomatic phase of acute HIV infection, the viral RNA shows in excess of 50,000 copies per mL.3 Three instances of false-positive HIV-1-RNA tests have been reported; in each instance, however, the person was not having symptoms, and the viral load was less than 2,000 copies per mL.10 The presence of high-titer HIV-I RNA (more than 50,000 copies per mL) in the absence of HIV antibodies establishes the diagnosis of acute HIV infection.3,11
HIV-1 antibody and viral load tests are readily available through commercial laboratories and should be performed whenever a patient presents with signs and symptoms of acute HIV syndrome and a history that is compatible with HIV infection. If viral RNA quantitation is not available, a serum or plasma p24 antigen test may be used to detect viral infection before the appearance of HIV antibodies.
If acute HIV infection is strongly suspected but the HIV-1 RNA PCR test is negative or shows a low titer, the initial high level of viral RNA may have already subsided. The patient should be followed with HIV-1 antibody tests at three months, six months and one year. If these tests remain negative, another diagnosis should be considered.
Benefits and Risks of Treatment
Even though treatment of acute primary HIV infection may seem difficult, compelling reports indicate that the administration of potent antiretroviral therapy can result in a rapid and sustained decline in the viral load to below the limit of detection within three months.12,13 Furthermore, studies of CD4 and CD8 lymphocyte dynamics show restoration of the normal ratio, reflecting recovery of the immune system.14 In one investigation of HIV-1–specific cell-mediated immune responsiveness, treatment of acute HIV infection resulted in the restoration of virus-specific immunity with control of viremia in six of six persons studied.15
Early institution of antiretroviral therapy has the advantage of keeping the viral load low by reducing replication and the appearance of resistant HIV phenotypes. It also may prevent immune depletion because of increased immune stimulation resulting from the strong antigenicity of HIV during primary infection.16 This allows for a more favorable initial immune response to HIV.
In isolated cases, treatment has been stopped after a period of time with no adverse effects. For example, the “Berlin patient” was treated aggressively for six months after acute HIV infection; the patient then stopped taking the medications but remained free of symptoms with barely detectable HIV-1 RNA for more than one year.17 However, other patients have rebounded after stopping treatment.18 The results of stopping antiretroviral medications at some point after the early initiation of therapy are currently being studied.
If acute HIV infection is not treated, the signs and symptoms disappear, along with the viremia. The person enters a prolonged stage of hidden viral replication during which the virus may not be culturable from the blood and HIV-1 RNA levels may be low or undetectable. During the next five to 10 years, lymph node architecture is destroyed, certain CD4 and CD8 cell lines are gradually depleted and progression to symptomatic disease ultimately occurs (Figure 1).19 Some subtle effects develop, reflecting early encephalopathy,20 but the liver, kidneys and lipid metabolism are spared.
FIGURE 1. HIV Infection Without Treatment
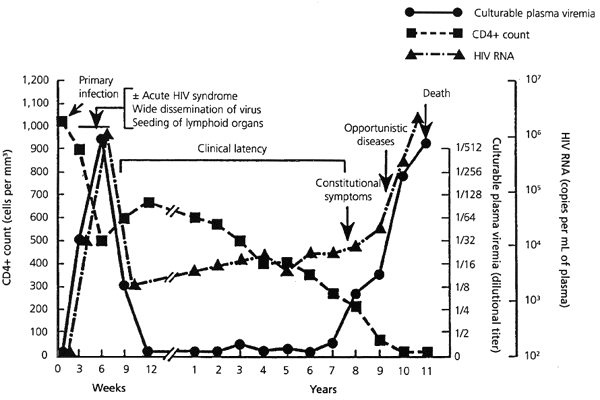
Adapted with permission from Fauci AS, Pantaleo G, Stanley S, Weissman D. Immunopathogenic mechanisms of HIV infection. Ann Intern Med 1996;124:654–63.
Some investigators suggest that persons who are not treated for acute HIV infection may be in a more favorable position when a cure is finally found,21 because they have been spared the toxicities of potent drugs, have avoided the development of drug-resistant HIV and have not suffered the stress of expensive, complex and potentially toxic medications. However, the potential benefits of early treatment appear to outweigh both the risks and negative effects of such treatment.22
Treatment of Acute HIV Infection
Patients identified during acute primary HIV infection should be treated with combination antiretroviral therapy to suppress viral replication to levels below the limit of detection on plasma HIV-1 RNA assays.1,23 Although opponents of this recommendation argue for conservative management,21 compelling arguments for early aggressive therapy have been presented.22
Patients who present with symptomatic acute HIV syndrome have a poorer prognosis than those with asymptomatic primary HIV infection.24 Early treatment may preserve immune system function by blunting the high level of HIV replication (decreasing the “set point,” or the plasma concentration of viral RNA) and reducing damage to the immune system during this period. Thus, early treatment has the potential to decrease the set point (thereby delaying disease progression), reduce the viral mutation rate and decrease the risk of HIV transmission.1 The current recommendation is to use, for an indefinite duration, combination antiretroviral therapy with at least three drugs to which the patient has never been exposed.
In most untreated patients, set-point plasma HIV-1 RNA levels stabilize between 1,000 and 10,000 copies per mL for a number of years. In the advanced stages of AIDS, HIV-1 RNA levels exceed 1 million copies per mL. In the absence of treatment, the apparent “nonprogessor” with an HIV-1 RNA level of less than 400 copies per mL is rare. Nonprogression is the state hopefully achievable with early aggressive therapy.1
The three-drug combinations can be the same as those used to treat established HIV infection (Table 3).1 We use the following:
- A nucleoside reverse transcriptase inhibitor (NRTI) with good central nervous system penetration, such as zidovudine (Retrovir; AZT), stavudine (Zerit; d4T) or abacavir (Ziagen).
- A nonthymidine NRTI such as didanosine (Videx; ddI), lamivudine (Epivir; 3TC) or zalcitabine (Hivid; ddC).
- A protease inhibitor such as indinavir (Crixivan), nelfinavir (Viracept), ritonavir (Norvir), saquinavir (Fortovase) or amprenavir (Agenerase). If a protease inhibitor is not tolerated or if the plan is to reserve protease inhibitors for future use should resistance develop, a nonnucleoside reverse transcriptase inhibitor, such as nevirapine (Viramune), delavirdine (Rescriptor) or efavirenz (Sustiva; EFV), should be tried.
Because treatment must continue indefinitely until new agents are introduced or a cure is found, several important issues need to be considered. Before this complex regimen is started, the patient should be counseled that interrupting therapy or reducing dosages can lead to viral resistance that is not reversible. Every effort should be made to choose a regimen that is durable and tolerable and that the patient can adhere to for a long time. The initial regimen needs to be the most efficacious available, so it should be one that is comfortable for the patient in terms of both side effects and lifestyle.
Virtually all antiretroviral agents have potentially serious side effects.1,23 Notable effects of protease inhibitors include the unmasking of latent diabetes mellitus, the development of hyperlipidemia or lipodystrophy (fatty deposits and the bloated abdomen known as “protease paunch”), the elevation of transaminase levels and the reactivation of previously quiescent viral hepatitis. Indinavir can cause nephrolithiasis and therefore should not be used in patients with preexisting renal disease. The nucleosides can cause pancreatitis, peripheral neuropathy, nausea and headache. Abacavir and amprenavir can cause serious hypersensitivity reactions. Efavirenz may cause dysphoria and nightmares.
If treatment needs to be stopped, all medications should be stopped to avoid the development of viral resistance. Abacavir should not be restarted because fatal hypersensitivity reactions have occurred on rechallenge.
Final Comment
Does aggressive treatment of acute HIV infection really decrease morbidity and mortality? Although few controlled studies have been conducted, the available data show that early and sustained therapy can result in the persistent suppression of HIV replication with reconstitution of certain virus-specific immune responses.15,16 We believe that treatment provides the best hope for patients with acute HIV infection. Some sources of updated AIDs information are provided in Table 4.
TABLE 4 Selected AIDS Information Resources
| National organizations | |
| American Foundation for AIDS Research | |
| 120 Wall St., 13th Floor | |
| New York, NY 10005 | |
| Telephone: 212-806-1600 | |
| Publishes a newsletter and an AIDS–HIV treatment directory | |
| CDC National Prevention Information Network | |
| P.O. Box 6003 | |
| Rockville, MD 20849-6003 | |
| Telephone: 800-458-5231 | |
| Reference and referral source on HIV, AIDS and STDs | |
| National AIDS Treatment Advocacy Project | |
| 580 Broadway, Suite 403 | |
| New York, NY 10012 | |
| Telephone: 212-219-0106 | |
| Treatment and education resource, publishes a newsletter | |
| San Francisco AIDS Foundation | |
| Telephone: 415-487-3000 | |
| Publishes a bulletin on experimental AIDS treatments | |
| International AIDS Society–USA | |
| 1001 B O'Reilly Ave. | |
| San Francisco, CA 94129-0916 | |
| Telephone: 415-561-6720 | |
| Educational resources for physicians on the management of HIV disease | |
| Newsletters and magazines | |
| GMHC Treatment Issues | |
| 119 W. 24th St. | |
| New York, NY 10011 | |
| Telephone: 212-367-1206 | |
| Provides education, advocacy, a telephone hotline and information on legal issues, treatment, meals, children, outreach and speakers | |
| PWA Newsletter | |
| Telephone: 212-255-0520 | |
| PWA Health Group provides information on drug research and treatments, and women's treatment issues. | |
| Treatment Review | |
| Telephone: 212-260-8868 | |
| AIDS Treatment Data Network publication that provides information on case management, treatment access and support | |
| National telephone hotlines | |
| AIDS Hotline | |
| Telephone: 800-342-AIDS | |
| CDC hotline that provides answers to questions, statistics, support, referrals and information about groups | |
| AIDS “Warm” Line | |
| Telephone: 800-933-3413 | |
| HIV telephone consultation service provided by the scientific division of the American Academy of Family Physicians | |
| Project Inform Hotline | |
| Telephone: 800-822-7422 | |
| Provides HIV treatment strategy information, resource guide and limited advocacy | |
| Internet sites | |
| AIDS Clinical Trials Information Services | |
| http://www.actis.org | |
| AIDS Treatment Data Network | |
| http://www.aidsnyc.org/network/ | |
| HIV/AIDS Information Center | |
| http://www.ama-assn.org/special/hiv/ | |
| Medscape HIV/AIDS | |
| http://hiv.medscape.com | |
| University of California at San Francisco | |
| http://hivinsite.ucsf.edu/ | |
| HIV/AIDS Treatment Information Service | |
| http://www.hivatis.org | |
| CDC | |
| http://www.cdc.gov | |
AIDS = acquired immunodeficiency syndrome; CDC = Centers for Disease Control and Prevention; HIV = human immunodeficiency virus; STDs = sexually transmitted diseases; GMHC = Gay Men's Health Crisis; PWA = People With Aids.

