Peyronie's disease is an acquired inflammatory condition of the penis associated with penile curvature and, in some cases, pain. It primarily affects men between 45 and 60 years of age, although an age range of 18 to 80 years has been reported. If left untreated, Peyronie's disease may cause fibrotic, nonexpansile thickening of relatively discrete areas of the corpora tunica, typically resulting in focal bend, pain or other functional or structural abnormalities of the erect penis. Many cases resolve without treatment. Medical therapies, including antioxidants (such as vitamin E and potassium aminobenzoate) and corticosteroids injected directly into the plaque, lack adequate scientific support. Surgery remains a mainstay when conservative measures fail.
Peyronie's disease was first described in 1704. It is named for Francois de la Peyronie, who, in 1743, described a patient who had “rosary beads of scar tissue to cause an upward curvature of the penis during erection.” The penile curvature of Peyronie's disease is caused by an inelastic scar, or plaque, that shortens the involved aspect of the tunica albuginea of the corpora cavernosa during erection.1,2 In approximately one third of patients, the scarring involves the dorsal and ventral aspects of the shaft. Such offsetting plaques may cause the penis to be straight but shortened or to have a lateral bend (Figure 1). The circumference of the shaft may also be reduced, resulting in an erect penis that is flail at the site of the constriction, firm proximal to the constriction and soft distally.3
FIGURE 1.
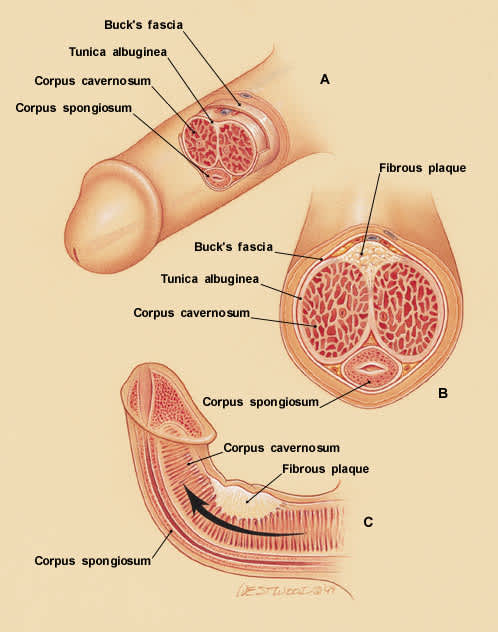
Penile curvature associated with Peyronie's disease. (A) anatomy of a normal erection. (B) Peyronie's disease. Penile cross-section showing plaque between the corpora. (C) Penile curvature. Fibrous plaque prevents uniform lengthening as erection occurs. As the rest of the corpus cavernosum and corpus spongiosum lengthen, the penis bends toward the involved area.
The first symptom of Peyronie's disease may be focal pain with erection, new curvature with erection or inability to penetrate as a result of curvature or distal flaccidity.3,4 Some patients who do not have pain with erection have tenderness on palpation of the indurated plaque.
Potential Etiologies
A number of authors believe that Peyronie's disease results, in part, from trauma.5–7 More than 75 percent of patients with Peyronie's disease are between 45 and 65 years of age, when elasticity of the collagen of the penis has diminished.5 Many patients recall an episode of penile trauma, such as an invasive procedure, blunt trauma or injury during intercourse, at the site of subsequent plaque formation. Up to 47 percent of patients with Peyronie's disease also had another condition associated with loss of elasticity, such as Dupuytren's contracture or Ledderhose's disease (fibrosis of the palmar and plantar fascias, respectively).8–11 Some authors5,12 suggest that either a single episode or recurrent episodes of flexion of the tunica albuginea may result in tears that bleed and form a clot, with subsequent fibrin deposition. Biopsy may demonstrate fibrin deposition and perivascular inflammation underlying the tunica albuginea and, occasionally, within and beneath Buck's fascia overlying the plaque.5
Presentation
Patients typically present with focal pain that occurs with erection, bent erection, presence of a hard mass and/or inability to have intercourse secondary to flail penis distal to the lesion.3 One half to two thirds of patients with Peyronie's disease describe pain as a symptom. Pain is associated with the inflammation generated by the active phase of the healing process, and it typically disappears as the inflammation resolves. It is believed to be the result of inflammation of the adjacent Buck's fascia, since the tunica albuginea itself has no nerve fibers.5
Clinical Course
During the first year or so after formation of the plaque, while the scar in the tunica is undergoing the process of remodeling, penile distortion may increase, remain static or, as is most often the case in younger men, resolve and disappear spontaneously.3–5 In most patients the curvature remains static as the scar matures although, in some patients, it becomes worse as fibrosis ensues and the scar contracts. In 25 percent of these patients the scarring process progresses to calcification, and in 25 percent of those it progresses to bone formation.3,5
After the scar has matured, the configuration of the tunica albuginea is unlikely to be changed by nonsurgical treatments.4 However, many patients with advanced disease who have not sought surgical correction have been able to continue mutually satisfactory sexual intercourse with a partner. Approximately one third of patients with end-stage disease have a disabling curvature that requires surgical correction.
Pain that occurs in conjunction with Peyronie's disease may also progress with the onset of new injuries to the corpora cavernosa occurring as a direct result of the patient's attempts to correct or compensate for the original defect during sexual intercourse.5 One of the more common reasons for seeking treatment involves discomfort of the patient's partner during intercourse, which is associated with penile curvature.
Diagnosis
Indurated plaques may be palpated on physical examination of the penis. Such palpation may elicit pain if the disease is still in the inflammatory stage. Corroboration of Peyronie's disease may be obtained by having the patient photograph the erect penis, demonstrating curvature, an hourglass shape or flail distal penis.
Radiographs of the penis may show calcification in 20 to 25 percent of patients with end-stage disease, and 25 percent of these patients have frank bone.3,5 Doppler flow studies and results of dynamic infusion cavernosometry and cavernosography are normal both proximal and distal to the plaque, demonstrating that disparity in the erection is not associated with lack of blood flow at or beyond the lesion.3–5
Treatment
Despite numerous treatment options, there is no generally accepted, standard nonsurgical treatment for Peyronie's disease. Moreover, the success of treatment may be difficult to assess because 20 to 50 percent of patients with Peyronie's disease experience spontaneous resolution.4 This potential for improvement probably warrants delay of surgical correction for at least six to 12 months after diagnosis unless the plaque is calcified or the patient is completely incapable of sexual activity.4
Oral agents, particularly those with antioxidant properties, have been tried with limited success. Such agents include vitamin E,10 potassium aminobenzoate (Potaba),13 and colchicine. Experimental intralesional treatments include corticosteroids, parathyroid hormone,11 collagenase and verapamil (Calan).8,14,15 Various modes of energy transfer, including ultrasound, radiation, laser therapy, short-wave diathermy and lithotripsy, have also been used.10,16,17 However, all current published reports of these treatments have been compromised by limited-sample patient populations, lack of control populations, poorly characterized outcome parameters, inadequate follow-up periods and inconclusive results. It has been difficult, therefore, to determine which, if any, of the nonsurgical treatments may be effective. Caution should be used when recommending any of these experimental treatments.
VITAMIN E AND VERAPAMIL
One possible medical regimen is 100 mg of vitamin E taken three times a day for a minimum of four months. Theoretically, this antioxidant will prevent further development of plaque, although studies have suggested that it is no more effective than placebo.
Injectable verapamil also has received some attention recently, although studies have either shown no statistical improvement over placebo or have been critically compromised by very small study size.14,15
POTASSIUM AMINOBENZOATE
Potassium aminobenzoate is considered a member of the vitamin B complex and is believed to promote antifibrotic activity through its mediation of increased oxygen uptake at the tissue level. Potassium aminobenzoate is rapidly excreted in the urine, so dosages are given at approximately three-hour intervals.13
The standard regimen of potassium aminobenzoate is 12 g daily, divided into six doses of four 500-mg tablets. The total of number of tablets per day is 24 (720 tablets per month). Since a minimum of six to 12 months of treatment is recommended, patients must take a huge number of pills during the course of treatment to achieve any benefit. In a study of 21 patients with Peyronie's disease who were treated with potassium aminobenzoate,13 morphologic deformity resolved completely in 18 percent of the study subjects and decreased in an additional 82 percent. The plaque resolved completely in 11 percent and decreased in size in an additional 67 percent, and pain resolved in every patient.13 A retrospective review18 noted that no well-controlled prospective, double-blind study containing adequate study subjects has been conducted to establish the efficacy of potassium aminobenzoate in the treatment of Peyronie's disease. Since the rate of spontaneous resolution is high, the results with potassium aminobenzoate may be no better than the results with placebo.
CORTICOSTEROID INJECTION
The corticosteroid preparation used in the treatment of Peyronie's disease varies, but two common regimens are dexamethasone (Decadron), in a dosage of 0.2 to 0.4 mg per plaque injected weekly for 10 weeks,8 and triamcinolone hexacetonide (Aristospan Intralesional), in a dosage of 2 mg administered once every six weeks for a total of six injections. Courses of treatment have been repeated in some instances. A small syringe and a fine needle are used to inject the medication into the plaque and the tissues immediately adjacent to it. Local anesthetic agents are not used routinely because of the risk of injection into the vascular corpora.
Using the dexamethasone regimen in 31 patients, one study13 reported an 81 percent benefit to a moderate or greater degree, with 42 percent of patients achieving what they described as marked improvement. In a study4 of 42 patients treated with triamcinolone, 33 percent of patients had complete recovery or marked improvement in symptoms and signs during the course of treatment.
Steroid injections are probably most effective during the initial formation of Peyronie's plaque, and success is limited with mature plaques. Patients are advised to abstain from sex during treatment to minimize further potential trauma to the penis.
SURGICAL MANAGEMENT
A number of surgical techniques are used for treatment of Peyronie's disease. The technique should be individually chosen for each patient. The optimal surgical approach considers penile rigidity, degree of curvature, shaft narrowing and erectile response.19 One commonly used surgical technique, the Nesbit procedure, involves excision of the plaque accompanied by “patch grafting” of the defect left by the excision. Graft material generally is taken from scrotal tunica vaginalis or non–hair-bearing skin from the forearm. Artificial graft material such as Gortex has also been used but with mixed results. These materials are generally less elastic and do not permit adequate stretch of the corpora during erections. Other techniques include penile prosthesis and plication of the tunica albuginea.
Excision of the plaque has been associated with complaints of diminished rigidity of erection and impotence following surgery. These problems have been attributed to damage of the erectile nerves during penile surgery. Thus, it is sometimes more practical to treat severe cases of Peyronie's disease with placement of an artificial penile prosthesis following incision and release of the plaque.

