
Am Fam Physician. 1999;60(2):558-564
A more recent article on heart murmurs in children is available.
See related patient information handout on heart murmurs in children, written by the authors of this article.
Many normal children have heart murmurs, but most children do not have heart disease. An appropriate history and a properly conducted physical examination can identify children at increased risk for significant heart disease. Pathologic causes of systolic murmurs include atrial and ventricular septal defects, pulmonary or aortic outflow tract abnormalities, and patent ductus arteriosus. An atrial septal defect is often confused with a functional murmur, but the conditions can usually be differentiated based on specific physical findings. Characteristics of pathologic murmurs include a sound level of grade 3 or louder, a diastolic murmur or an increase in intensity when the patient is standing. Most children with any of these findings should be referred to a pediatric cardiologist.
Primary care physicians frequently encounter children with heart murmurs.1 Most of these young patients do not have heart disease. One set of investigators2 found that physicians were generally accurate in determining whether a murmur was benign or pathologic. Nonetheless, 61 percent of the murmurs referred for subspecialist evaluation were found to be functional, or innocent, murmurs. The investigators hypothesized that increased education of health care providers and parents might be helpful in alleviating unnecessary anxiety and reducing the number of patients with innocent murmurs who are referred for further evaluation.2
Echocardiography is not always needed to diagnose pediatric murmurs. One study3 showed that direct referral for echocardiography was an expensive way to evaluate children with heart murmurs. Pediatric cardiology consultation was significantly less costly in that many innocent murmurs were diagnosed without echocardiography. A recent study4 found that pediatric echocardiograms performed in adult cardiology practices were unnecessary in 30 percent of patients, were of inadequate quality in 32 percent of patients and resulted in an erroneous impression of the nature or presence of pathologic disease in 32 percent of patients.
This article reviews the individual steps in the cardiac physical examination and the possible innocent or pathologic findings. The focus is on helping physicians become even more confident about their ability to diagnose innocent murmurs and to decide which patients might benefit from pediatric cardiology referral.
General Approach
Busy clinicians need an approach that allows them to appropriately identify and refer patients with pathologic murmurs to a pediatric cardiologist. This approach should also help them know when they can confidently reassure the parents of a child with a functional murmur that referral is unnecessary.
In a busy office practice, time constraints make it difficult to perform a complete cardiac physical examination on every patient. However, this examination must be performed on any child who has a heart murmur or historical features that indicate the presence of heart disease or abnormal cardiac function. Features of concern in infants include feeding intolerance, failure to thrive, respiratory symptoms or cyanosis. In older children, chest pain (especially with exercise), syncope, exercise intolerance or a family history of sudden death in young people should prompt a complete examination.
The examination is conducted in a quiet room. An infant may lie quietly on the examination table. However, it can be challenging to keep a one- to two-year-old child quiet enough for a good examination to be performed. Having the child sit in the lap of a parent or other caregiver may be helpful.
Precordial Palpation
The cardiac examination begins with palpation to assess precordial activity and femoral pulses. Increased precordial activity is commonly felt in patients with increased right or left ventricular stroke volume. Increased pre-cordial activity occurs in patients with an atrial septal defect, a moderate or large ventricular septal defect or significant patent ductus arteriosus. This increased activity should raise the possibility that the auscultatory findings may be pathologic. Other explanations for increased precordial activity include patient anxiety, anemia and hyperthyroidism.
Once both brachial pulses have been palpated, the right brachial pulse should then be palpated simultaneously with the femoral pulse. If the timing and intensity of the two pulses are equal and blood pressure in the right arm is normal, coarctation of the aorta is unlikely.
Precordial palpation is also necessary to feel “thrills,” which are the palpable consequence of blood flowing rapidly from high pressure to lower pressure. Some ventricular septal defects result in thrills at the lower left sternal border. Moderate to severe pulmonary valve stenosis may cause a thrill at the upper left sternal border. A thrill resulting from aortic stenosis is frequently palpable in the suprasternal notch.
Auscultation of First and Second Heart Sounds
Precordial palpation is followed by auscultation. This part of the examination entails separate evaluation of each heart sound and each phase of the cardiac cycle.1
FIRST HEART SOUND
Auscultation begins with listening for the first heart sound (S1) at the lower left border of the sternum. The S1 is caused by closure of the mitral and tricuspid valves and is normally a single sound. An inaudible S1 indicates that some sound is obscuring the closure sound of these valves. The differential diagnosis for murmurs that obscure S1 includes ventricular septal defects, some murmurs caused by atrioventricular valve regurgitation, patent ductus arteriosus and, occasionally, severe pulmonary valve stenosis in a young child. These S1-coincident murmurs are also known as “holosystolic” murmurs.
CLICKS
If S1 is audible but appears to have two components at some spots in the precordium, the patient has either a click or an asynchronous closure of the mitral and tricuspid valves. Clicks may originate from any valve in the heart. Depending on their origin, clicks have different identifying characteristics.
Ejection clicks originating from the pulmonic valve begin shortly after the atrioventricular valves close, vary with respiration and are best heard at the upper to middle area of the left sternal border. Aortic valve ejection clicks begin shortly after S1 and are best heard at the apex. They do not vary with respiration.
Systolic clicks originating from the mitral valve are best heard at the apical area when the patient is standing. Occasionally, the tissue closing a ventricular septal defect can pop or click early in systole (Figure 1).
SECOND HEART SOUND
After auscultation for clicks throughout the four listening areas, the next step is to return to the upper left sternal border and listen to the second heart sound (S2). This sound is caused by closure of the aortic and pulmonic valves.
The S2 should split into two components when the patient inspires. The first component, aortic second sound (A2), is closure of the aortic valve. The second component, pulmonic second sound (P2), is caused by closure of the pulmonary valve. The splitting of S2 occurs because inspiration brings more blood into the right ventricle. Right ventricular ejection is prolonged, and the pulmonary valve closes later. An awareness of this phenomenon is helpful in understanding the physical examination features of the patient with an atrial septal defect. A loud, single S2 indicates either pulmonary hypertension or congenital heart disease involving one of the semilunar valves.
Murmurs
Systolic murmurs have only a few possible causes: blood flow across an outflow tract (pulmonary or aortic), a ventricular septal defect; atrioventricular valve regurgitation, or persistent patency of the arterial duct (ductus arteriosus). Systolic murmurs can also be functional (benign).
GRADES
Systolic murmurs are graded on a six-point scale. A grade 1 murmur is barely audible, a grade 2 murmur is louder and a grade 3 murmur is loud but not accompanied by a thrill. A grade 4 murmur is loud and associated with a palpable thrill. A grade 5 murmur is associated with a thrill, and the murmur can be heard with the stethoscope partially off the chest. Finally, the grade 6 murmur is audible without a stethoscope. All murmurs louder than grade 3 are pathologic.
TIMING
Systolic murmurs may be timed as early, middle or late systolic. They can also be timed as holosystolic.
VENOUS HUMS
Many children with functional murmurs have venous hums. These sounds are caused by the flow of venous blood from the head and neck into the thorax. They are heard continuously when the child is sitting. The sounds should disappear when light pressure is applied over the jugular vein, when the child's head is turned or when the child is lying supine. Venous hums are common and are not pathologic. Patients with venous hums do not require pediatric cardiology referral. All other diastolic murmurs are pathologic and therefore warrant referral.
DESCRIPTION OF CHARACTER
The character, or tone, of a murmur may aid in the diagnosis. Words such as “harsh,” “whooping,” “honking,” “blowing,” “musical” and “vibratory” may be useful, albeit somewhat subjective, in describing murmurs.
A “harsh” murmur is consistent with high-velocity blood flow from a higher pressure to a lower pressure. “Harsh” is often appropriate for describing the murmur in patients with significant semilunar valve stenosis or a ventricular septal defect.
“Whooping” or “blowing” murmurs at the apex occur with mitral valve regurgitation. The term “flow murmur” is often used to describe a crescendo/decrescendo murmur that is heard in patients with a functional murmur (Figure 2). However, similar systolic ejection murmurs may be heard in patients with atrial septal defect, mild semilunar valve stenosis, subaortic obstruction, coarctation of the aorta or some very large ventricular septal defects.
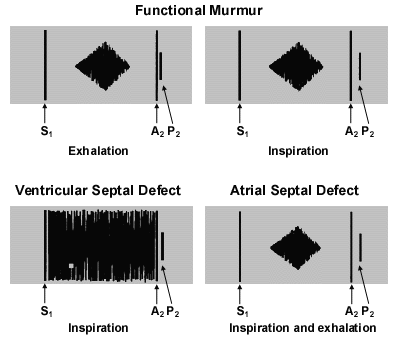
Many functional or innocent murmurs are “vibratory” or “musical” in quality. Still's murmur is the innocent murmur most frequently encountered in children. This murmur is usually vibratory or musical.5
LOCATION OF HIGHEST INTENSITY
The location of the highest intensity of a murmur is also important (Table 1). A murmur caused by aortic stenosis is often best heard at the upper sternal border, usually on the right side. A murmur resulting from pulmonary stenosis is heard best at the upper left sternal border. A murmur caused by a ventricular septal defect or tricuspid valve insufficiency is heard at the lower left sternal border. A murmur resulting from mitral valve regurgitation is best heard at the apex.
| Area | Murmur |
|---|---|
| Upper right sternal border | Aortic stenosis, venous hum |
| Upper left sternal border | Pulmonary stenosis, pulmonary flow murmurs, atrial septal defect, patent ductus arteriosus |
| Lower left sternal border | Still's murmur, ventricular septal defect, tricuspid valve regurgitation, hypertrophic cardiomyopathy, subaortic stenosis |
| Apex | Mitral valve regurgitation |
POSITION CHANGES IN THE DIFFERENTIATION OF MURMURS
Position changes are very helpful in differentiating functional and pathologic murmurs. The vibratory functional murmur heard in a young child (Still's murmur) decreases in intensity when the patient stands.
Most pathologic murmurs do not change significantly with standing. An important exception is the murmur of hypertrophic cardiomyopathy, a potentially life-threatening condition. This murmur increases in intensity when the patient stands. In the upright position, venous return to the heart is reduced, decreasing the left ventricular end diastolic volume. As left ventricular size decreases, the left ventricular outflow tract narrows, and the systolic outflow obstruction increases. This narrowing increases the intensity of the murmur. Overall, hypertrophic cardiomyopathy is rare; however, it is one of the leading causes of sudden death in athletes.6
Atrial Septal Defect
Perhaps the best way to decide whether a patient needs to be referred to a pediatric cardiologist is to know confidently the clinical findings of the atrial septal defect. The abnormal findings in patients with atrial septal defects are often quite subtle and thus are easily confused with the physical findings in patients with functional or innocent murmurs (Table 2). Loud murmurs from ventricular septal defects or significant aortic or pulmonic stenosis are not subtle and are not often confused with innocent murmurs.
| Physical finding | Innocent murmur | Atrial septal defect |
|---|---|---|
| Precordial activity | Normal | Increased |
| First heart sound (S1) | Normal | Normal |
| Second heart sound (S2) | Splits and moves with respiration | Widely split and fixed (i.e., does not move with inspiration) |
| Systolic murmur (supine) | Crescendo/decrescendo | Crescendo/decrescendo |
| Possibly vibratory at lower left sternal border | “Flow” at upper left sternal border | |
| Systolic murmur (standing) | Decreases in intensity | Does not change |
| Diastolic murmur | Venous hum | Inflow “rumble” across tricuspid valve area |
The first step in the examination is palpation for the precordial activity at the left sternal border, feeling for increased activity that may be present from right ventricular enlargement. The examination is performed with the patient in a supine position.
Auscultation at the lower left sternal border should reveal a normal, single and easily audible S1. Listening at the upper left sternal border for S2 should reveal variable splitting with respiration. A widely split S2 that does not change with respiration indicates either complete right bundle branch block or an atrial septal defect.
Because of the increased volume of blood in the right ventricle in the patient with an atrial septal defect, the S2 always sounds as if the patient has taken a deep breath. This means that the volume of blood in the right ventricle is increased and it takes longer for the ventricle to eject its contents, thus leading to a fixed, widely split S2.
Murmurs heard in patients with atrial septal defects are often of low pitch and intensity. However, the intensity of the murmur is not a reliable basis for the diagnosis. The systolic murmur is caused by an increased volume of blood coursing across the right ventricular outflow tract. If the murmur is audible, it can be heard at the upper left sternal border.
Patients with atrial septal defects may also have a diastolic murmur. In this situation, increased diastolic blood flow across the tricuspid valve may cause a soft, often barely audible low-frequency “diastolic rumble.” This sound is heard best with the bell of the stethoscope placed at the lower left sternal border. Pushing down with the bell causes the skin beneath the stethoscope to act as a diaphragm. Consequently, only the high-frequency sounds are heard. Releasing the tension on the bell allows the low-frequency rumbling sound to become audible at the lower left sternal border.
After the supine examination, the examination should be repeated with the patient standing. If the patient has an atrial septal defect, the features of increased precordial activity, a widely split S2, a systolic murmur at the upper left sternal border and a diastolic rumble should still be present when the patient is standing. If the patient has an innocent or functional murmur, the precordial activity should be normal, the S2 should split and move with respiration, the systolic murmur should decrease in intensity and no diastolic murmur should be present in the tricuspid valve area.
If no symptoms are present, the S1 is normal, the S2 splits and is of normal intensity, and no clicks are heard, the differential diagnosis of a grade 1 to grade 2 systolic murmur is frequently a choice between an atrial septal defect and a functional murmur.
In young children, it can be difficult to confidently distinguish the respiratory variation of S2 splitting in the patient with a normal murmur from the fixed S2 splitting in the patient with an atrial septal defect. Because the patient with an asymptomatic atrial septal defect will not develop pulmonary hypertension or other permanent sequelae early in life, careful yearly follow-up examination by the primary care physician can be recommended. By the time a child reaches preschool age, the normal or abnormal splitting of the S2 usually becomes clear (Table 3).
| Symptoms such as chest pain |
| Family history of Marfan syndrome or sudden death in young family members |
| Malformation syndrome (e.g., Down syndrome) |
| Increased precordial activity |
| Decreased femoral pulses |
| Abnormal second heart sound |
| Clicks |
| Loud or harsh murmur |
| Increased intensity of murmur when patient stands |
Follow-up or Referral
A patient who has a pathologic cardiac examination or who has cardiac symptoms and questionable findings on the cardiac examination should be referred to a pediatric cardiologist. A child with a malformation syndrome associated with congenital heart disease should also be referred for additional evaluation. Conversely, an asymptomatic patient whose physical findings on a conscientiously performed cardiac examination indicate a low probability of cardiac pathology should be followed by the primary care physician. Referral is indicated if possibly pathologic findings emerge on serial examinations.1
Final Comment
Many normal children have heart murmurs, but all of these children do not require referral to a pediatric cardiologist. Atrial septal defect is the pathologic diagnosis that is frequently missed or easily confused with a functional murmur. It is important for family physicians to remember the physical examination features that differentiate a functional murmur from a pathologic murmur. Before referring asymptomatic patients to a pediatric cardiologist, family physicians should attempt to rule out atrial septal defects by performing a complete, organized cardiac examination. In this way, they can reduce the number of referrals for evaluations of functional murmurs and at the same time increase their ability to confidently reassure families when referral is unnecessary.