Normal visual development is rapid during the first six months of life and continues through the first decade. Young children are uniquely sensitive to conditions that interfere with vision and visual development. Amblyopia, or functionally defective development of the central visual system, may be caused by common vision problems such as strabismus, uncorrected refractive errors and deprivation secondary to occlusion. Prematurity is especially associated with eye pathology, including retinopathy of prematurity, amblyopia, strabismus and refractive errors. When detected early, amblyopia and many other childhood vision abnormalities are treatable, but the potential for correction and normal visual development is inversely related to age. Since many affected children are asymptomatic, early detection of abnormal visual function requires effective screening throughout early childhood. Special considerations apply to screening examinations of children born prematurely.
The visual system at birth is functional but very limited. Visual fixation is present in full-term, alert newborns, but the ability to follow targets (tracking) is usually not observed until about two months of age. Visual acuity, when measured by visual evoked potential (VEP) using graded targets, is determined at birth to be about the equivalent of a Snellen visual acuity of 20/400. Color discrimination and contrast sensitivity (the ability to detect gradations in brightness) are present but poorly developed in newborns.1,2
During the first six months of life, rapid anatomic development in the eye and central visual pathways parallels a rapid improvement in visual acuity, contrast sensitivity and color discrimination. Maturation of the retina and retinal photoreceptor (rod and cone cells), myelination of the optic nerves and tracts, and increased synaptic density of the visual cortex are the anatomic substrates that allow visual acuity to reach the equivalent of Snellen 20/30 by six months of age.2
The visual system develops at a slower rate after six months of age. Myelination continues to increase in the central visual pathways until about four years of age, and development of the visual cortex continues throughout the first decade of life.
The eye movement system is incompletely functional at birth but develops rapidly in parallel with the development of vision. Binocularity (the ability to perceive vision from both eyes simultaneously) and stereopsis (integration of the images from the two eyes to produce a single image with depth perception) can develop only if the eyes are fairly precisely aligned. In addition, as the infant's interest in exploring the visual environment increases, the need for voluntary eye movements increases. An integrated system of voluntary and reflexive eye movements to allow normal alignment, fixation on objects of interest and smooth pursuit (tracking) movements develops rapidly, so that by three months of age infants are capable of following objects vertically and horizontally while the eyes remain aligned. Misalignment or abnormal eye movements are detectable by this age3 (Table 1).
TABLE 1 Normal Development of Vision and Eye Movements
| Age | Normal vision and eye movements |
|---|---|
| Birth (term) | Fixation |
| Poor following | |
| Intermittent strabismus frequently present | |
| Visual acuity 20/400 to 20/600 | |
| One month | Horizontal following to midline |
| Normal alignment | |
| Visual acuity 20/300 | |
| Two months | Vertical following begins |
| Normal alignment | |
| Visual acuity 20/200 | |
| Three months | Good horizontal and vertical following |
| Normal alignment | |
| Visual acuity 20/100 | |
| Accommodation begins | |
| Binocularity detectable | |
| Six months | Visual acuity 20/20 to 20/30 |
| Binocularity well developed | |
| Eight to 10 years | End of sensitive period for amblyopia |
Abnormalities in Visual Development
AMBLYOPIA
Amblyopia is a reduction in vision, usually in one but sometimes in both eyes, that is not attributable to anatomic eye or optic nerve pathology. Instead, the vision loss results from altered visual development within the central visual pathways. Amblyopia is the most common cause of vision loss in children and is found in as many as 5 to 7 percent of school-age children.4,5
Normal visual development requires that a focused image form in each eye that can be fused (superimposed and integrated) by the brain into a single image. Any problem that interferes with a focused, fusible image during the first eight to 10 years of life is capable of causing amblyopia. The most common causes of amblyopia include strabismus (misalignment of the eyes, images that are not fusible), anisometropia (unequal refractive components of eyes, images are not focused simultaneously), and deprivation (cataract, ptosis, corneal scar).
In the case of unilateral amblyopia, the visual pathway from the affected eye will develop poorly or even regress, relative to the opposite eye. Fewer cells will be responsive to the affected eye in the visual cortex and lateral geniculate, and the cells related to the involved side will be smaller and have fewer synapses. While amblyopia may under certain circumstances occur bilaterally, it appears that relative competitive effects between the eyes accelerate the development of amblyopia when the opposite eye is unaffected. For this reason, unilateral or asymmetric vision problems are the most likely to cause amblyopia.
The critical sensitive period in the development of amblyopia begins within the first weeks of life and lasts until about eight to 10 years of age3 and is also the period during which amblyopia may be reversed by treating the cause and stimulating visual development of the affected side. Clinically, it is apparent that reversal of amblyopia occurs most completely and rapidly in patients treated at the youngest ages. Thus, early detection and treatment of amblyopia are essential for the best possible outcome.
Therapy for amblyopia consists of treating the cause (strabismus, refractive error, etc.) and, in unilateral cases, forced use of the impaired eye. Because of the many primary causes of unfocused or unfusable images, initial treatment of the underlying cause must be individualized. Corrective lenses, strabismus surgery, cataract surgery, ptosis surgery or other therapies may be necessary to allow a fusible, focused image.
The most common mechanism of forcing use of the affected eye is occlusion of the opposite eye, usually achieved with an adhesive eye patch (Figure 1). Optical blurring, achieved with corrective lenses or pharmacologically with mydriatics (e.g., atropine [Atropisol]), is useful in selected cases. Frequent observation and vision testing are necessary in patients undergoing occlusion therapy to monitor progress and prevent amblyopia in the patched eye.4,6
FIGURE 1.
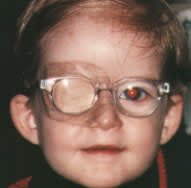
Occlusive patching for amblyopia therapy. Patching of the opposite eye is the most useful therapy.
Amblyopia therapy is generally continued until vision in the affected eye is normalized or until no further improvement is seen. Optimal visual potential and rate of reversal of amblyopia with treatment are age-dependant—the older the child, the slower the improvement in vision, and the less likely that there will be full recovery of normal vision. The most frequent reason for failure of amblyopia therapy is non-compliance. Other factors include age at time of treatment, severity of amblyopia and associated ocular conditions.
STRABISMUS AND EYE MOVEMENT DISORDERS
Strabismus, or misalignment of the eyes, includes an extremely heterogeneous group of eye movement problems ranging from constant to latent, and from congenital to those acquired late in life.
In addition to allowing the development of binocularity and stereopsis, eye movements allow visual fixation on objects of regard, as well as following moving objects. Complex coordination of visual, vestibular and proprioceptive inputs allow the symmetric control of the two eyes in a coordinated manner. Before the age of six weeks, coordination of eye movements is poor, and the eyes in normal infants may be misaligned. Therefore, it is difficult or impossible to diagnose strabismus before six weeks of age. By two to three months of age, alignment is stable, and abnormalities of alignment may be diagnosed accurately in cooperative, alert infants.
Strabismus in some form is found in approximately 4 percent of children under six years of age.7 Amblyopia is frequently associated, and restoration of normal eye alignment is essential to allow the normal development of binocularity and stereopsis. In humans, the precise age limit for restoration of alignment to allow development of normal binocularity and stereopsis is not well understood, but it appears that patients with congenital esotropia (convergent deviation) must be treated before two years of age for optimal visual outcome.6 Thus, early detection and treatment of strabismus are essential to maximize potential visual function.
Misalignment of the eyes is most easily detected in infants by observation of the corneal light reflex (Figure 2a). Displacement of the light reflex from the center of the pupil, or asymmetry, demonstrates abnormal alignment if the infant is fixating on the light and the observer is directly behind the light. The simultaneous red reflex test (Bruckner test) is also a rapid and simple screening tool for alignment, detecting asymmetry of the red reflex as abnormal when observing through the peephole of the ophthalmoscope with fixation on the light (Figure 2b). Photoscreening uses similar methods to detect misalignment with a camera. Other clinical methods used to measure ocular alignment in children include cover testing, prism-cover and prism-light reflex tests. In older children, secondary methods to detect suppression or amblyopia, including monocular vision tests and stereopsis tests, are useful in screening for strabismus. Strabismus can be classified according to age of onset.
FIGURE 2A
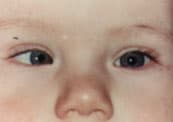
Demonstration of a right esotropia using the Hirshberg, or light reflex, method. The corneal light reflex is centered in the pupil in the left eye but displaced laterally in the right eye.
FIGURE 2B
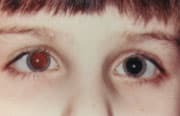
Demonstration of right esotropia using the Bruckner method. The color of the red (pupil) reflex is lighter and the reflex is brighter in the right eye when observed through the peephole of the direct ophthalmoscope. This method may also demonstrate asymmetric refractive error, fundus lesions, cataract and other types of strabismus.
Strabismus Beginning in Early Infancy. Infantile strabismus, defined as constant misalignment present before six months of age, includes infantile esotropia (convergent deviation) and exotropia (divergent deviation).
Infantile esotropia, sometimes called congenital esotropia, is not always observed at birth but is easily apparent by three months of age, usually with a large and constant convergent deviation of 15 degrees or more. There is frequently a family history of strabismus, and this syndrome is not usually associated with any other neurologic or developmental problems. Affected infants frequently show alternating fixation, but amblyopia may develop if one eye is preferred.
Treatment for infantile esotropia consists of therapy for amblyopia, if present, and strabismus surgery to realign the eyes. Corrective lenses may be necessary if significant refractive errors are present. Even with optimal treatment and surgical realignment, patients with infantile esotropia do not always develop a capacity for binocularity. Frequently, associated vertical deviations, recurrence of esotropia, and amblyopia require treatment later in life.8
Exotropia is much less common than esotropia during infancy. Infantile exotropia is rarely an isolated finding but is frequently seen in association with cerebral palsy, prematurity and other neurodevelopmental conditions. Treatment is similar to that used for infantile esotropia, but outcomes depend on associated conditions.9
Less common causes of congenital or infantile strabismus include Duane syndrome (congenital aberrant innervation of cranial nerves III and VI), Brown syndrome (congenital or acquired abnormality of the trochlea, causing vertical misalignment) and congenital cranial nerve palsies.
Strabismus Beginning after Six Months of Life. Accommodative esotropia is caused by abnormalities of the near reflex. Convergence of the eyes is controlled reflexively, along with focus for near vision (accommodation). This normal integration is necessary to maintain eye alignment and to focus with targets at various distances. Patients with refractive errors requiring a greater than normal amount of accommodation (hypermetropia), and patients with inherently excessive reflexive convergence will converge too much and cross their eyes when accommodating. Accommodative esotropia appears in children from six months to seven years of age, most commonly between two and three years of age. Frequently, the esotropia is present only when looking at near objects. Amblyopia is frequently associated.
Treatment for accommodative esotropia most often consists of lenses to correct refractive error and reduce the need for accommodation (Figures 3a and 3b). Bifocal lenses may be necessary to reduce accommodative needs for near fixation. Amblyopia therapy may be necessary in combination with corrective lenses. Occasionally, miotic eye drops (echothiophate iodide [Phospholine iodide], or pilocarpine [Pilopine HS]) are used to cause pharmacologic accommodation in place of or in combination with corrective lenses. Surgery may be necessary if other measures do not realign the eyes.8
FIGURE 3A
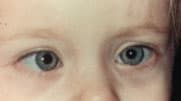
Esotropia demonstrated by the corneal light reflex method, without corrective lenses.
FIGURE 3B
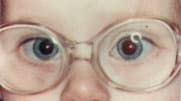
When corrective lenses are in place, the esotropia disappears, reducing the need for accommodation and eliminating the stimulus for excessive convergence.
The potential for recovery of normal stereopsis is greater in patients with accommodative esotropia than in patients with infantile esotropia.
In most cases, when exotropia is detected after six months of age, patients have intermittent exotropia. If extropia is intermittent, the eyes may be completely straight with normal stereopsis when the child looks at near objects (Figure 4a); deviation is most apparent when the child looks at distant objects (Figure 4b). Amblyopia rarely accompanies intermittent exotropia, but the latter is associated with myopia.
FIGURE 4A
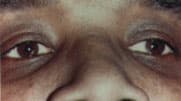
Intermittent exotropia. The eyes can be straight for near fixation, with good binocularity. The corneal light reflex in the left eye appears eccentric because the patient is not looking directly at the camera.
FIGURE 4B
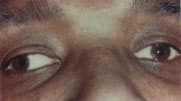
Intermittent exotropia. Fatigue, sedation or fixation on distant targets cause the right eye to deviate outward.
Intermittent exotropia does not always require treatment, since vision and stereopsis may be normal under usual circumstances. Some patients may benefit from orthoptic exercises to reinforce binocularity and prevent suppression. Correction of refractive error with corrective lenses may also help control the deviation. Patients with poorly controlled intermittent exotropia and those who regress to constant exotropia may require surgery to regain binocularity.9
Other Forms of Strabismus. Other, less common acquired forms of strabismus include paralytic strabismus related to palsy of cranial nerves III, IV or VI, and traumatic strabismus associated with orbital fractures (Figures 5a and 5b).
FIGURE 5A
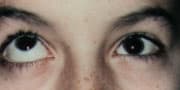
Patient with orbital fracture with strabismus. The left eye fails to elevate in up-gaze (hypotropia).
FIGURE 5B
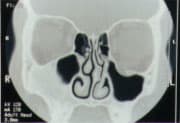
Coronal computed tomographic scan of the orbits of the patient in Figure 5a demonstrates an old left orbital floor fracture with entrapment of the inferior rectus muscle, with incidental fibrous dysplasia of the right maxilla.
Nystagmus. Nystagmus is involuntary, rhythmic oscillation of the eyes and may be caused by bilateral poor vision (sensory nystagmus) or abnormal motor input (motor defect nystagmus). Both types are frequently associated with strabismus. Sensory nystagmus may be caused by any abnormality of the anterior visual pathway causing bilateral poor vision early in life. Sensory nystagmus does not occur with unilateral vision problems or with defects of the visual pathway posterior to the optic chiasm. It is thought that in most infants, nystagmus has a sensory cause.
Ocular structural abnormalities, retinal diseases and optic nerve diseases are associated with sensory nystagmus. The evaluation of infants with nystagmus is therefore directed at detecting possible sensory causes and may require electrophysiologic tests, including electroretinogram (ERG) and VEP. Imaging studies of the brain are indicated only when nystagmus is associated with specific neurologic findings, including optic atrophy, papilledema and other focal neurologic abnormalities.
Motor defect nystagmus can be diagnosed only by excluding possible sensory abnormalities and may be familial. Motor defect nystagmus is thought to be caused by abnormal motor input signals from the oculomotor control centers. Patients with this disorder generally have better vision than those with sensory nystagmus.10
PREMATURITY AND THE EYES
Premature birth is associated with several eye and visual system problems. Retinopathy of prematurity (ROP) is a disorder of developing blood vessels that occurs in premature newborns. Normal retinal vascularization is usually complete around 40 weeks' postconceptional age in normal term pregnancies. ROP occurs when the normal pattern of progressive blood vessel growth within the retina is interrupted by premature birth. Vessel growth becomes poorly regulated and excessive. In advanced stages, abnormal blood vessels proliferate massively on the retina and into the normally avascular vitreous space in the center of the eye (Figure 6). These abnormal vessels can leak and bleed and, in subsequent late phases of ROP, may contract, causing distortion or detachment of the retina, leading to severe vision loss or blindness. Severe intravitreal vascularization, fibrosis and retinal detachment lead to the formation of membranes posterior to the lens (retrolental fibroplasia) in the most severe cases.
FIGURE 6.
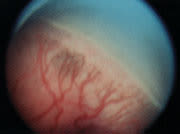
Retinopathy of prematurity. In this clinical fundus photograph, the margin of retinal vascularization is visible. To the lower left of the figure, regularly spaced vessels advance to the abnormal white ridge, beyond which no vessels are present. Proliferation of small vessels from the surface of the ridge into the vitreous space above the retina is consistent with stage III retinopathy of prematurity.
The incidence and severity of ROP are highly correlated with the degree of prematurity. The disorder occurs almost exclusively in infants with a birth weight of less than 1,500 g (3 lb, 5 oz) and a gestational age of less than 32 weeks. The incidence of ROP in infants with a birth weight less than 750 g (1 lb, 10 oz) is 90 percent, and drops to 47 percent in those from 1,000 to 1,250 g (2 lb, 3 oz to 2 lb, 11 oz).11 Other associations include the length and concentration of supplemental oxygen use, anemia and other conditions, such as intraventricular hemorrhage and necrotizing enterocolitis.
Treatment of ROP, consisting of laser photocoagulation or transscleral cryotherapy applied to the peripheral avascular retina, has been demonstrated to stimulate regression of the vascular proliferation and reduce the incidence of severe vision loss in advanced cases, but is not effective in all cases.12 Intraocular surgery to restore normal retinal anatomy after retinal detachment is unsuccessful in most cases.13
In infants with only mild involvement, spontaneous regression is the rule, and treatment is not necessary. The critical stage of ROP occurs when abnormal vessels begin to extend into the vitreous space.14 This “threshold” stage occurs most frequently at a postconceptional age of 32 to 46 weeks, or seven to 15 weeks postnatally11 (Figure 7). Only 6 percent of infants weighing less than 1,250 g will reach this stage of severity, but approximately two thirds of eyes reaching the “threshold” will develop severe vision loss or blindness. Treatment with cryotherapy or laser photocoagulation reduces this risk by at least one half.12
FIGURE 7.
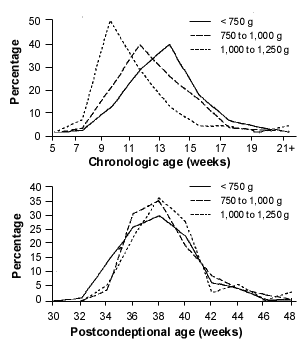
Distribution of onset of threshold retinopathy of prematurity (ROP). (Top) Onset of threshold ROP by chronologic postnatal age, in weeks. (Bottom) Onset of threshold ROP by postconceptional age, in weeks.
Adapted with permission from Palmer EA, Flynn JT, Hardy RJ, Phelps DL, Phillips CL, Schaffer DB, et al. Incidence and early course of retinopathy of prematurity. The Cryotherapy for Retinopathy of Prematurity Cooperative Group. Ophthalmology 1991;98:1628–40.
Premature infants born at less than 32 weeks of gestational age or less than 1,250 g must be monitored to detect the appearance of ROP and determine if treatment is indicated (Table 2). Infants with a birth weight of 1,251 to 1,500 g (2 lb, 12 oz to 3 lb, 5 oz), prolonged ventilation or other complications may be screened at the discretion of the neonatologist. Because the period of risk for retinopathy frequently extends past the date of hospital discharge, the primary care physician and the ophthalmologist must coordinate outpatient ophthalmic care.15
TABLE 2 Screening Retinal Examinations for Premature Infants
| 1. All infants with a birth weight of 1,250 g (2 lb, 11 oz) or less, or a postconceptional age of 32 weeks or less must be screened for ROP. In addition, premature infants with a birth weight of 1,251 to 1,500 g (2 lb, 12 oz to 3 lb, 5 oz) and prolonged artificial ventilation, and those requiring high concentrations of oxygen may be screened at the discretion of the referring neonatologist. |
| 2. The first retinal examination is performed at four to six weeks' chronologic age, or 32 weeks' postconceptional age, whichever is earliest. |
| 3. Retinal examination is repeated approximately every two weeks until treatment is required or retinal maturity is reached. Infants with rapidly progressive ROP or approaching the threshold for treatment may require more frequent examination. |
| 4. Children with premature birth (before 32 weeks of gestation) should have a follow-up eye examination, including refraction and motility evaluation, between six and 18 months of corrected age. |
ROP = retinopathy of prematurity.
Late ocular complications of prematurity include high refractive errors, strabismus and amblyopia. Children with significant prematurity should continue to have periodic complete eye examinations.16
Vision Screening in Primary Care of Children
Amblyopia is the leading cause of monocular vision loss in children and adults under 40 years of age and also causes impairment of depth perception, peripheral vision and contrast sensitivity.4 Amblyopia is potentially reversible if detected during the critical period for visual development and treated appropriately. Early detection and initiation of treatment are critical in preventing permanent visual impairment. Screening vision and eye examinations may also detect other common conditions such as strabismus, nystagmus and refractive problems, as well as rare diseases such as retinoblastoma, infantile glaucoma, cataract and retinal disease.
Age-appropriate eye and vision evaluation should be incorporated into the scheduled health evaluation routine for children.17,18 Physicians should be alert for and investigate any suspicion from parents, teachers and other adults who have reported observing visual difficulty or eye abnormality in a patient. Vision screening of infants and pre-verbal children consists of observation of their ability to visually fixate and follow objects or lights with each eye. Steadiness of fixation should be assessed to detect nystagmus. By three to four years of age, verbal children are usually capable of recognition acuity measurement using charts with pictures, figures or letters. Acuity must be evaluated with each eye separately, or unilateral defects will be missed. Adhesive eye patches prevent cheating and peeking, and allow reliable occlusion for monocular testing.
Eye alignment may be assessed most easily by observation of the corneal light reflex. With the child fixating on the light and the observer directly behind the light, the reflection on the cornea should be centered within the pupil and should be symmetric in each eye. Simultaneous observation of the red reflex of both eyes (Bruckner test), using the ophthalmoscope with a bright large illumination spot while the patient fixates on the light, allows assessment of the ocular media (cornea, lens, vitreous, retina) and may detect media opacification (corneal scar, cataract) or leukocoria (white reflex) resulting from retinoblastoma or coloboma. Misalignment of the eyes will produce an asymmetry of the red reflex between eyes when they are observed simultaneously (Figure 2b).
Cover-uncover testing can be performed in children capable of prolonged fixation. The patient is directed to look at a specific object. The eyes are observed for any shifting in alignment when one eye is occluded by the examiner, usually with a hand. The eyes are again observed for a shift in position when the occluder is removed, and the test is repeated on the opposite eye. A shift observed with the cover-uncover test demonstrates a manifest misalignment of the eyes, present under binocular conditions. Alternate cover testing is similar, but the occlusion is alternated between the eyes with one eye occluded at all times. A shift in alignment with the alternate cover test will occur with manifest strabismus as well as latent deviations, or phorias, which are only present under monocular conditions. Additional tests that may increase screening sensitivity to abnormality but should not replace other clinical tests include photo-screening and stereotesting. Timing of screening eye and vision evaluation should allow detection of abnormalities early enough to permit treatment (Table 3).
TABLE 3 Timing of Eye and Vision Screening Examinations
| Age | Screening test | Detectable abnormalities |
|---|---|---|
| Birth | Eye anatomic examination | Corneal opacity |
| Congenital glaucoma | ||
| Cataract | ||
| Iris coloboma | ||
| Microphthalmia | ||
| Red reflex (Bruckner test) | Optical opacity | |
| Corneal opacity | ||
| Cataract | ||
| Retinal coloboma | ||
| Retinoblastoma | ||
| Three to six months, repeat annually | Red reflex | Optical opacity |
| Corneal opacity | ||
| Cataract | ||
| Retinal coloboma | ||
| Retinoblastoma | ||
| Strabismus | ||
| Visual fixation and following | Reduced visual acuity | |
| Nystagmus | ||
| Corneal light reflex | Strabismus | |
| Three to four years, repeat annually | Monocular recognition visual acuity | Reduced visual acuity |
| Cover testing | Strabismus |
Children with abnormal anatomy, fixation, ocular misalignment, asymmetric or abnormal red reflexes, nystagmus or failure to achieve recognition acuity of 20/40 in each eye should be considered to have failed the screening evaluation, and undergo a more complete ocular and visual evaluation. Premature birth, family history of childhood vision problems or with other increased risks should prompt an early, complete evaluation. This evaluation must include a cycloplegic refraction (measurement of refractive status after pharmacologic paralysis of accommodation) to eliminate the possibility of latent refractive error. The primary care physician should select an appropriate ophthalmologic consultant who is comfortable and familiar with the examination of small children.

