Infections caused by human parvovirus B19 can result in a wide spectrum of manifestations, which are usually influenced by the patient's immunologic and hematologic status. In the normal host, parvovirus infection can be asymptomatic or can result in erythema infectiosum or arthropathy. Patients with underlying hematologic and immunologic disorders who become infected with this virus are at risk for aplastic anemia. Hydrops fetalis and fetal death are complications of intrauterine parvovirus B19 infection.
Parvovirus B19 was discovered fortuitously in 1975 by Cossart and colleagues,1 who unexpectedly found viral particles in the sera of asymptomatic patients being screened for hepatitis B infection. Biochemical and molecular characteristics subsequently demonstrated that these particles were parvoviruses and, because specimen 19 of panel B contained the unexpected virus, parvovirus B19 was so designated.
The development of a parvovirus B19–specific serologic test led to the first report of symptomatic infection in humans. This report, published in 1980, described two soldiers who had a brief febrile illness.2 Subsequently, the simultaneous reports of parvovirus B19 as the etiologic agent for transient aplastic crisis among patients with sickle cell disease and for erythema infectiosum among schoolchildren established the association between parvovirus infection and these two disorders.3
Epidemiologic and serologic data not only have confirmed the role of parvovirus B19 in erythema infectiosum and transient aplastic crisis but have also established the importance of this virus as a cause of asymptomatic infection, chronic anemia in the immunocompromised host, acute and chronic arthritis and hydrops fetalis (Table 1). Although recent reports have implicated parvovirus B19 as a factor in the pathogenesis of vasculitis and neurologic disease, the precise role, if any, of this virus in these entities awaits further elucidation.4–6
TABLE 1 Clinical Entities Strongly Associated with Human Parvovirus B19 Infection
| Erythema infectiosum |
| Nonspecific febrile illness |
| Arthropathy |
| Erythrocyte aplasia |
| Transient aplastic crisis |
| Chronic anemia |
| Nonimmune hydrops fetalis |
Pathogenesis
Human parvovirus B19 is a single-stranded DNA virus with a predilection for infecting rapidly dividing cell lines, such as bone marrow erythroid progenitor cells.7 Experimental studies in which healthy adult volunteers were inoculated intranasally with the virus have contributed significantly to our understanding of the pathogenesis and clinical features of infection.8,9 Viremia occurs during the first week of infection, accompanied by constitutional symptoms of fever and malaise, and by erythroid progenitor cell depletion in the bone marrow. At the height of the viremia, a precipitous drop in the reticulocyte count occurs and is followed by anemia, which is rarely clinically apparent in healthy patients but can cause serious anemia if the red blood cell count is already low. The reduction in the reticulocyte count is occasionally accompanied by leukopenia and thrombocytopenia.
The appearance of parvovirus B19–specific IgM antibodies in the serum in the second week after inoculation corresponds with clearance of the viremia. In the third week after inoculation, specific IgG antibodies appear in the serum, and the rash of erythema infectiosum and arthropathy develop. Because the appearance of the rash corresponds with the development of IgG antibodies and occurs after the viremia has cleared, the rash of erythema infectiosum signifies that the virus can no longer be transmitted.
Epidemiology
Seroepidemiologic studies from several countries show that parvovirus B19 infection is common. Approximately 50 percent of children have detectable parvovirus B19–specific IgG antibodies by the age of 15, and 50 to 80 percent of adults are seropositive.10,11 Women of childbearing age have an annual seroconversion rate of 1.5 percent.12
Infection appears to be most common in late winter and spring.13 Because parvovirus DNA has been found in respiratory secretions at the time of viremia, respiratory spread appears to be the most common route of transmission. The virus appears to be readily transmitted with close contact; the transmission rate with household contact approaches 50 percent, while the transmission rate following school and daycare exposure has been reported to be 10 to 60 percent.3,14 Because the virus is present in high titers in serum and is resistant to conventional heat treatments, transmission from blood products has occurred.15 Nosocomial transmission of the virus also has been documented.16
Clinical Manifestations
ASYMPTOMATIC INFECTION
Most persons with parvovirus B19 infection remain asymptomatic. Most persons who are seropositive for the virus have no recollection of previous symptoms. In one study, 32 percent of household contacts of patients with acute parvovirus B19 infection reported no symptoms at the time that they had parvovirus-specific IgM antibodies.3
ERYTHEMA INFECTIOSUM
Erythema infectiosum, also known as fifth disease and “slapped-cheek” disease, most commonly affects children between the ages of four and 10 years and is the most recognizable illness associated with parvovirus B19 infection. Although the clinical features of erythema infectiosum have been recognized for almost two centuries, not until the early 1980s was the link between this exanthem and parvovirus B19 established. It is now known that parvovirus B19 is the only etiologic agent of erythema infectiosum.17
The classic course of erythema infectiosum can be divided into three distinct stages (Table 2). The first stage, occurring after an incubation period of four to 14 days, consists of a mild prodromal illness characterized by low-grade fever, headache and gastrointestinal symptoms. This stage, which often is unrecognized, corresponds with the period of viremia and the period of contagion.
TABLE 2 Classic Course of Parvovirus B19 Infection and Erythema Infectiosum
| Stage 1 |
| Period of transmissability |
| Mild prodromal illness |
| Viremia |
| Erythroid progenitor cell depletion |
| Development of parvovirus B19–specific IgM antibodies |
| Stage 2 |
| Facial exanthem, or ”slapped cheek” appearance |
| Clearance of viremia |
| Development of parvovirus B19–specific IgG antibodies |
| Stage 3 |
| Lacy, erythematous maculopapular exanthem on trunk and extremities |
| Evanescent course of exanthem over 1 to 3 weeks |
| Arthropathy |
The second stage of the illness, occurring three to seven days after the prodrome, is characterized by the appearance of a bright erythematous facial exanthem (Figure 1). Because this exanthem most commonly involves the malar eminences and spares the nasal bridge and perioral areas, the characteristic “slapped-cheek” appearance becomes evident. This stage is seen more commonly in children than in adults, and the exanthem may become more marked with exposure to sunlight.
FIGURE 1.

Typical facial exanthem of erythema infectiosum. Note the sparing of the perioral areas, resulting in the typical “slapped cheek” appearance.
The third stage of the illness occurs one to four days after the appearance of the facial exanthem and is characterized by the appearance of a lacy, erythematous, maculopapular exanthem on the trunk and extremities (Figure 2). This eruption may be pruritic and often is evanescent, recurring over one to three weeks. Because the appearance of the exanthem corresponds with the development of antibody, patients with the rash of erythema infectiosum are no longer contagious.
FIGURE 2.
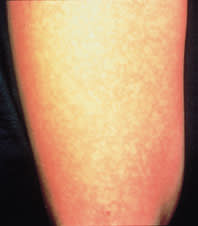
Lacy, reticular exanthem of the extremity in a child with erythema infectiosum.
Although it is helpful to classify the stages of erythema infectiosum, the distinct features may be variable. For example, the facial exanthem may be pronounced in some patients but not in others. Similarly, the third stage of the illness may range from a very faint erythema to a florid confluent eruption.
ARTHROPATHY
It has become increasingly clear over the past several years that parvovirus B19 causes arthritis and arthralgias in adults and children. Although parvovirus infections in adults are most commonly asymptomatic, an estimated 60 percent of women with symptomatic disease manifest arthropathy.18,19 Men appear to be affected much less frequently.
The most common presentation of parvovirus-related arthropathy in adults is the acute onset of arthralgias or frank arthritis involving the hands, knees, wrists and ankles. The symptoms usually subside within one to three weeks, although about 20 percent of affected women have persistent or recurring arthropathy for months and years.18 Concurrent constitutional symptoms such as fever are rare, but one half of patients have an associated generalized rash, and about 15 percent have the typical facial exanthem.19
The incidence of parvovirus-related arthropathy is lower in children than in adults, and girls are more likely than boys to have joint symptoms. Reports from a series of 22 children presenting to a pediatric rheumatology clinic with serologic or clinical evidence of an acute parvovirus B19 infection have shed light on the clinical features of arthropathy in children.20 Unlike in adults, the arthropathy in children most commonly affects the large joints such as the knees, ankles and wrists, mostly in an asymmetric pattern. In the series of 22 children, one half of the children had concurrent constitutional symptoms but, surprisingly, only one third had a concurrent exanthem. Although the joint symptoms promptly resolved in most of the children, eight of the 22 children had prolonged symptoms, and their disease would have met the criteria for juvenile rheumatoid arthritis if the diagnosis of parvovirus infection had not been made.
ERYTHROCYTE APLASIA
Because parvovirus B19 infects erythroid progenitor cells in the bone marrow and causes temporary cessation of red blood cell production, patients who have underlying hematologic abnormalities (and thus depend on a high rate of erythropoiesis) are prone to cessation of red blood cell production if they become infected. This can result in a transient aplastic crisis, which may occur in persons with chronic hemolytic anemia and conditions of bone marrow stress. Thus, patients with sickle cell anemia, thalassemia, acute hemorrhage and iron deficiency anemia are at risk.21,22 Typically, these patients have a viral prodrome followed by anemia, often with hemoglobin concentrations falling below 5.0 g per dL (50 g per L) and reticulocytosis. Although recovery is usually spontaneous and recurrence does not occur, severe disease with heart failure and death is possible. Therefore, such patients are best monitored carefully, usually in the hospital, for signs of congestive heart failure. Life-saving red blood cell transfusions may be required. These patients are contagious during the acute illness and thus need to be kept in respiratory isolation to prevent nosocomial transmission.
Chronic parvovirus B19 infection of the bone marrow has been described in immunocompromised hosts. Children and adults with hematologic and solid organ malignancies, transplant recipients and patients with human immunodeficiency virus infection are especially at risk of chronic bone marrow infection. This can result in severe, prolonged or recurrent anemia, which may require red blood cell transfusions.23 Administration of intravenous immune globulin may also be beneficial.24 However, its efficacy has not been proved in well-controlled trials.
INTRAUTERINE INFECTIONS
The likelihood of a healthy outcome is very high after parvovirus B19 infection in pregnancy. However, parvovirus infection can lead to fetal infection, possibly resulting in miscarriage or nonimmune hydrops fetalis. Because most pregnant women who become infected with this virus are asymptomatic, it has been difficult to determine the risk of fetal infection, fetal wastage and nonimmune hydrops. Estimates of parvovirus B19–associated fetal loss range from 2 to 10 percent.25,26 The overall risk of fetal loss as a result of parvovirus infection must take into account maternal susceptibility to infection and the likelihood of infection during pregnancy. Approximately 50 percent of women are seropositive for the virus before pregnancy, and the likelihood of infection ranges from 30 to 50 percent after a close exposure.26 The overall risk of parvovirus B19–associated fetal loss is estimated to be 1 to 2 percent. 26
Hydrops fetalis, which is manifested at birth by severe anemia, high-output cardiac failure and extramedullary hematopoiesis, is one possible consequence of congenital infection. Parvovirus B19 has been shown to cause a congenital infection syndrome, manifested by rash, anemia, hepatomegaly and cardiomegaly.27
Diagnosis and Management
The diagnosis of erythema infectiosum is made clinically, and laboratory studies are not needed under normal circumstances. Serologic tests are usually relied on for the diagnosis of parvovirus B19 infection in patients with transient aplastic crisis or arthropathy; a positive parvovirus B19–specific IgM antibody or a significant rise in parvovirus B19–specific IgG titer is indicative of an acute or recent infection.
The finding of pronormoblasts on bone marrow examination of patients with anemia is suggestive of parvovirus B19 infection. Because immunocompromised patients may not be able to mount an immune response, serologic tests in these patients may not be reliable. In such situations, viral DNA isolation from the blood or bone marrow by dot blot isolation or polymerase chain reaction may be helpful.
The management of parvovirus infections must take into account the severity of the infection and the patient's status. Because infection in healthy children and adults is self-limited, no specific therapy is warranted. Patients with arthropathy may be treated with nonsteroidal anti-inflammatory drugs for symptomatic relief. Patients with transient aplastic crisis may require blood transfusions to prevent congestive heart failure. Intravenous immune globulin has been used to treat immunocompromised patients who develop chronic anemia from parvovirus B19 infection. Finally, intrauterine fetal blood transfusions have been attempted in cases of parvovirus B19–related severe hydrops fetalis.28
Infection Control and Prevention
Children with erythema infectiosum are not infectious and can attend school and day care. Hospital isolation measures are not necessary if admission is required for another disorder.
Patients hospitalized with transient aplastic crisis from parvovirus B19 superimposed on chronic anemia should be kept in droplet isolation to prevent nosocomial spread and minimize health care worker exposure.29
Pregnant women who are exposed to children at home or at the workplace should be counseled about the risks of parvovirus B19 infection. Given the high prevalence of parvovirus B19 in the community, the high rate of silent infection and the low risk of adverse effects on the fetus, routine exclusion of pregnant women from the workplace where erythema infectiosum is present is not recommended.29 Health care workers who are pregnant should be informed of the preventive measures they can take to lower the risk of transmission, such as not caring for immunocompromised patients with acute or chronic parvovirus infection. Serologic testing, if available, may be offered to determine susceptibility in women at increased risk of exposure to parvovirus B19.

