Dermabond is a cyanoacrylate tissue adhesive that forms a strong bond across apposed wound edges, allowing normal healing to occur below. It is marketed to replace sutures that are 5-0 or smaller in diameter for incisional or laceration repair. This adhesive has been shown to save time during wound repair, to provide a flexible water-resistant protective coating and to eliminate the need for suture removal. The long-term cosmetic outcome with Dermabond is comparable to that of traditional methods of repair. Best suited for small, superficial lacerations, it may also be used with confidence on larger wounds where subcutaneous sutures are needed. This adhesive is relatively easy to use following appropriate wound preparation. Patients, especially children, readily accept the idea of being “glued” over traditional methods of repair.
Physicians have long sought an efficient method of wound repair that requires little time and minimizes discomfort for their patients, yet produces a good cosmetic outcome. Dermabond, the newest tissue adhesive to be labeled by the U.S. Food and Drug Administration (FDA), may well meet those criteria. Cyanoacrylate tissue adhesives have been around for more than 20 years but have only recently become available for use in this country for incisional and laceration repair. They have been shown to have negligible histotoxicity, to form a strong bond to wound edges and to provide long-term cosmesis equivalent to, or better than, traditional methods of repair. In addition, they require less than one half the time for wound closure. Table 1 summarizes the benefits of tissue adhesive over sutures.1–11
TABLE 1. Advantages of Adhesive vs. Sutures
| Maximum bonding strength at two and one-half minutes |
| Equivalent in strength to healed tissue at seven days post repair |
| Can be applied using only a topical anesthetic, no needles |
| Faster repair time |
| Better acceptance by patients |
| Water-resistant covering |
| Does not require removal of sutures |
History and Characteristics of Cyanoacrylates
Cyanoacrylate tissue adhesives combine cyanoacetate and formaldehyde in a heat vacuum along with a base to form a liquid monomer.9 When the monomer comes into contact with moisture on the skin's surface, it chemically changes into a polymer that binds to the top epithelial layer. This polymer forms a cyanoacrylate bridge, binding the two wound edges together and allowing normal healing to occur below. The conversion from monomer to polymer occurs rapidly, preventing seepage of the adhesive below the wound margins as long as the edges are well apposed. Heat is often generated during the change from monomer to polymer, and this heat may be felt on occasion by patients during application to the skin. Cyanoacrylates have also been shown to have antimicrobial properties.10–14
Cyanoacrylates were first manufactured in 1949. The first adhesives were noted to have extreme inflammatory effects on tissues. N-butyl-2-cyanoacrylate, which was developed in the 1970s, was the first adhesive to have negligible tissue toxicity and good bonding strength, as well as acceptable wound cosmesis.
N-butyl-2-cyanoacrylate has been used in cartilage and bone grafting, coating of corneal ulcers in ophthalmology, repair of damaged ossicles in otolaryngology, coating of aphthous ulcers, embolization of gastrointestinal varices and embolization in neurovascular surgery.1,2,8,10,11 This adhesive is not labeled for this use by the FDA but has been used in Canada and numerous other countries for more than 20 years.
Dermabond (2-octylcyanoacrylate), the latest in cyanoacrylate technology, has less toxicity and almost four times the strength of N-butyl-2-cyanoacrylate.15 Special plasticizers have been added to the formula to provide flexibility. This adhesive reaches maximum bonding strength within two and one-half minutes and is equivalent in strength to healed tissue at seven days post repair.15
When Can Dermabond Be Used?
Dermabond is marketed as a replacement for sutures that are 5-0 or smaller in diameter. Properly selected wounds on the face, extremities and torso may be closed with the adhesive. The use of adhesive rather than sutures is solely up to the discretion of the physician and will reflect his or her level of comfort and experience. Extremity and torso wounds tend to heal better when subcutaneous sutures are placed first. If adhesive is chosen by the physician to be used on areas of high tension or mobility (such as joints), this area should be immobilized in a splint to prevent premature peeling of the adhesive.
Scalp wounds may be closed with adhesive using meticulous care so as not to allow excess adhesive to run through the hair. Dermabond must be kept dry in this area for at least five days for normal healing.
This tissue adhesive should not be used on animal bites, severely contaminated wounds, ulcers, puncture wounds, mucous membranes (including mucocutaneous junctions) or areas of high moisture content, such as the groin or axillae (Table 2). The adhesive may be used on selected hand, foot and joint wounds if these areas are kept dry and immobilized.4–6
TABLE 2. Contraindications to Use of Skin Adhesives
| Jagged or stellate lacerations |
| Bites, punctures or crush wounds |
| Contaminated wounds |
| Mucosal surfaces |
| Axillae and perineum (high-moisture areas) |
| Hands, feet and joints (unless kept dry and immobilized) |
Technique for Use in Wound Closure
The availability of a tissue adhesive by no means obviates the need for thorough wound irrigation and cleansing. Deeper wounds should undergo thorough wound preparation as with traditional methods of repair to reduce the risk of infection. This will often include the need for topical or local anesthesia. Good wound management should not be compromised for a quick repair with a tissue adhesive.
Wound closure with Dermabond is achieved in several steps (Table 3). Smaller lacerations can often be cleansed with an antibacterial compound and flushed with sterile saline solution before closure. Small lacerations on the face usually heal well with this preparation. One study6 showed that only one of five children needed local anesthesia for repair of minor facial lacerations with Dermabond. Because the adhesive peels off in five to 10 days, deeper lacerations to the torso and extremities should have subcutaneous sutures placed to strengthen the wound closure and optimize long-term cosmesis. Deep wounds without subcutaneous sutures seem to have a higher dehiscence rate.4
TABLE 3. Steps in Use of Dermabond
| 1. Apply topical anesthetic as needed. |
| 2. Prepare wound with antiseptic. |
| 3. Appose wound edges. |
| 4. Crush Dermabond vial and invert. |
| 5. Gently brush adhesive over laceration. |
| 6. Avoid pushing adhesive into wound. |
| 7. Apply three layers of adhesive. |
Dermabond comes in a single-use vial in sterile packaging. It consists of an outside plastic casing with an inner glass ampule containing 0.5 mL of adhesive that can be expressed through the applicator tip once the vial has been crushed. As the adhesive moves through the applicator tip, it mixes with an initiator and begins the chemical change from monomer to polymer. Moisture on the skin's surface adds the final catalyst to create the strong polymer bond that bridges the wound edges.
After cleansing, the wound should be positioned so that excess adhesive does not run off into areas not meant to be glued. If this should occur, the excess adhesive should quickly be wiped away with a dry gauze. Good hemostasis should be achieved using pressure on the wound or application of 1:1,000 topical epinephrine solution, if needed. On facial wounds, prior application of a topical anesthetic with epinephrine is usually sufficient. Excessive wound seepage before closure may prevent good bonding to the epithelial layer and may also result in excessive heat production during polymerization.
The edges of the wound must be approximated manually and evenly. If there is uncertainty about whether this can be done, the wound should probably be sutured instead. Forceps or manufactured skin approximation devices may also be used, if preferred. Lack of wound edge eversion does not seem to alter long-term cosmetic outcome.4–7
Once the edges have been approximated, the Dermabond vial is crushed between the thumb and index finger and inverted. The vial must be used in the next few minutes or polymerization in the applicator tip will prevent expression of the adhesive. The adhesive is expressed by gently squeezing the vial, which allows the adhesive to be seen at the applicator tip. If the vial is squeezed too hard, adhesive may drip from the end of the vial. To prevent this from happening, the vial must be squeezed gently and squeezing must stop when a drop begins to form at the tip, allowing the adhesive to be drawn back up into the vial with the vacuum thus created.
Once the adhesive is at the applicator tip, it is applied to the apposed wound edges with gentle brushing motions. At no time should the applicator tip be pressed into the wound; this may cause adhesive to enter the wound, which may lead to a foreign-body reaction and prevent normal wound healing or cause dehiscence.4 Adhesive will not enter the wound unless it is placed there by force. After applying adhesive across the wound edges and holding the edges together for at least 30 seconds before releasing, more adhesive should be applied in an oval pattern around the wound to encompass a greater surface area on the skin—this adds greater strength to the wound closure (Figure 1). At least three layers should be applied to ensure optimal strength to the wound closure. The first layer of adhesive reaches maximal strength within two and one-half minutes; the subsequent layers usually take longer to dry because less moisture is available for polymer formation. The wound should not be touched until the adhesive dries completely. Fanning or blowing on the wound will not speed up polymerization.
FIGURE 1.
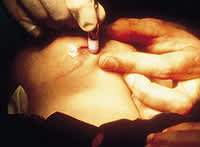
Application of adhesive to chin.
The adhesive acts as its own water-resistant bandage, and no added coverings are needed. Patients may shower normally and pat the area dry. The adhesive will spontaneously peel off in five to 10 days. No topical antibiotics should be applied to the closed wound because this would break down the adhesive and cause early peeling. In active children, a bandage may be recommended to prevent them from picking at their wound or reinjuring themselves in the same location. Children should not take baths, because excessive exposure to water may loosen the top epithelial layer of skin and cause premature peeling or wound dehiscence. Examples of facial wounds repaired with Dermabond are shown in Figures 2a, 2b, 2c, 3a and 3b.
FIGURE 2A.
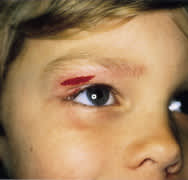
Laceration to lower eyebrow.
FIGURE 2B.
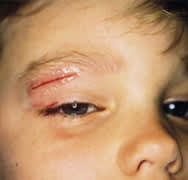
Closed wound with adhesive.
FIGURE 2C.
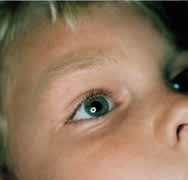
Three months after treatment with adhesive.
FIGURE 3A.
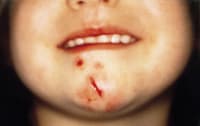
Laceration to chin.
FIGURE 3B.
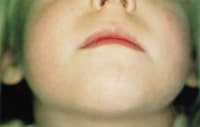
Three months after treatment with adhesive.
Precautions
Randomized controlled clinical trials4–8 have shown that infection rates are not significantly different between wounds that have been sutured and wounds that have been closed with Dermabond. However, if adequate wound cleansing and preparation are compromised because of the ease of use of a tissue adhesive, an increase in infection rates could occur.
Suspected infection below the adhesive may be treated with oral antibiotics. Purulence from a true infection generally pushes the dried polymer away from the skin. In these rare cases, the adhesive should be removed and standard wound care measures should be initiated. Reapplication of adhesive in such cases is not recommended.
Application of adhesive to a wound will sometimes result in run-off to areas not intended to be glued. Excess adhesive can simply be wiped away with a dry gauze if done immediately. If an object such as a finger or forceps becomes inadvertently adhered to the patient during repair, place pressure on the patient's skin adjacent to the edge of the object and gently roll the object away. This action allows the object to be peeled away from the skin without pulling the edges of the wound apart.
If wound edges are not aligned after the first application of adhesive, wipe the glue away immediately with a dry gauze. A 10-second “grace period” exists before the adhesive becomes too polymerized to wipe away. If the adhesive has already dried, the application of antibiotic ointment or petroleum jelly for 30 minutes will loosen the polymer for removal.
Generous amounts of ophthalmic antibiotic ointment may be placed on eyelids that have been inadvertently glued shut. Lids should not be pried open or eyelashes removed, as the lids will open after one to two days with this method.
Cyanoacrylates are used for corneal perforations and are not harmful to the eye.13 The placement of gauze over the eyes when working in this area should prevent exposure to run-off.
Repeat trauma to a wound or excessive picking at the wound may result in dehiscence. Reported rates of wound dehiscence are extremely low, ranging from three out of 68 patients in one study to no cases of dehiscence in two studies with 96 patients combined.4–8 Depending on the time of presentation, these wounds could be closed again with adhesive or sutures.
Cost
Dermabond is the only FDA-labeled and commercially available adhesive in this country and costs approximately $24 a vial (12 vials per box), with a shelf-life of two years. Sutures commonly used in the ambulatory care setting generally cost about $5 per package. In most cases when Dermabond is used, a suture tray need not be opened for minor facial lacerations, because only gauze, antiseptic solution, sterile saline and tissue adhesive are needed for closure. Patients can be quickly treated with this method. All other closures will require suture equipment and local anesthesia to ensure painless wound preparation and placement of deep sutures. Dermabond saves time even when used after placement of subcutaneous sutures and requires no suture removal or follow-up visit. Unless a complication develops, wounds closed with tissue adhesive need not be seen again.

