Although cancer has an annual incidence of only about 150 new cases per 1 million U.S. children, it is the second leading cause of childhood deaths. Early detection and prompt therapy have the potential to reduce mortality. Leukemias, lymphomas and central nervous system tumors account for more than one half of new cancer cases in children. Early in the disease, leukemia may cause nonspecific symptoms similar to those of a viral infection. Leukemia should be suspected if persistent vague symptoms are accompanied by evidence of abnormal bleeding, bone pain, lymphadenopathy or hepatosplenomegaly. The presenting symptoms of a brain tumor may include elevated intracranial pressure, nerve abnormalities and seizures. A spinal tumor often presents with signs and symptoms of spinal cord compression. In children, lymphoma may present as one or more painless masses, often in the neck, accompanied by signs and symptoms resulting from local compression, as well as signs and symptoms of systemic disturbances, such as fever and weight loss. A neuroblastoma may arise from sympathetic nervous tissue anywhere in the body, but this tumor most often develops in the abdomen. The presentation depends on the local effects of the solid tumor and any metastases. An abdominal mass in a child may also be due to Wilms' tumor. This neoplasm may present with renal signs and symptoms, such as hypertension, hematuria and abdominal pain. A tumor of the musculoskeletal system is often first detected when trauma appears to cause pain and dysfunction out of proportion to the injury. Primary care physicians should be alert for possible presenting signs and symptoms of childhood malignancy, particularly in patients with Down syndrome or other congenital and familial conditions associated with an increased risk of cancer.
Only about 2 percent of all cancers in western industrialized nations occur in children. Hence, childhood cancer is relatively uncommon, with an annual incidence of 150 new cases per 1 million children in the United States. However, cancer accounts for 10 percent of childhood deaths and is second only to accidents as a cause of death in U.S. children.1
Although the incidence of childhood cancers increased slightly in the early 1980s, there has been no substantial change in the incidence of major pediatric cancers since the mid-1980s.2 Mortality rates for these malignancies have declined significantly as a result of improvements in supportive care and therapy.3
The most common childhood malignancies are acute lymphoblastic leukemia, central nervous system (CNS) tumors and lymphomas. Together, these cancers account for 63 percent of cases (Table 1).2 Childhood cancers occur at a slightly higher rate in males and at a significantly higher rate in whites.1,3
Although cancer can develop in children of any age, certain malignancies have a predilection for specific age groups. For example, neuroblastoma, retinoblastoma and Wilms' tumor most commonly occur in children between birth and four years of age, whereas osteosarcoma, Ewing's sarcoma and Hodgkin's disease tend to occur in children more than 10 years of age.
This article reviews the typical presenting features of the more common childhood cancers. Some genetic syndromes requiring increased vigilance for cancer are also discussed.
Difficulties in Diagnosis
Childhood cancer can be difficult to diagnose in the primary care setting. The index of suspicion tends to be low because of the relative rarity of malignancies in children. Furthermore, as shown in Table 2, the presenting signs and symptoms are often nonspecific and mimic those of common childhood conditions such as viral infections. Signs and symptoms that warrant evaluation are summarized in Table 3.
TABLE 2 Signs and Symptoms of Childhood Cancers and Conditions That Can Mimic These Cancers
| Sign or symptom | Type of cancer | Common conditions in the differential diagnosis |
|---|---|---|
| Fever | Leukemia, lymphoma | Infection |
| Vomiting | Abdominal mass, brain tumor | Infection, gastroesophageal reflux |
| Constipation | Abdominal mass | Poor diet |
| Cough | Mediastinal mass | Upper respiratory infection, reactive airway disease, pneumonia |
| Bone or muscle pain | Leukemia, bone tumor, neuroblastoma | Musculoskeletal injury, viral infection |
| Headache | Brain tumor | Tension headache, migraine, infection |
| Lymphadenopathy (> 2 cm) | Leukemia, lymphoma, metastatic disease | Lymphadenitis, systemic infection, collagen vascular disease |
| Hematuria | Wilms' tumor | Urinary tract infection, glomerulonephritis |
| Voiding difficulty | Rhabdomyosarcoma | Congenital urinary tract abnormalities |
TABLE 3 When and How to Evaluate Signs and Symptoms for Cancer
| Symptom | When to evaluate | Test |
|---|---|---|
| Fever | Fever lasts longer than 14 days with no identifiable cause. | CBC with differential |
| Vomiting | Vomiting lasts longer than 7 days with no identifiable cause. | Abdominal and head CT scans |
| Vomiting is associated with headache during sleep. | Head CT scan | |
| Constipation | Constipation is prolonged (> 1 month) and does not respond to conventional measures. | Abdominal and pelvic CT scans |
| Cough | Cough is prolonged (> 2 weeks) and has no identifiable cause. | Chest radiograph |
| Bone or muscle pain | Pain is prolonged (> 2 weeks) and has no identifiable cause. | Plain-film radiograph, bone and CT scans, CBC |
| Headache | Headache occurs during sleep, is associated with neurologic signs and vomiting, or occurs in the absence of a family history of migraine. | Head CT scan |
| Hematuria | Evaluate immediately if hematuria has no identifiable cause. | Abdominal ultrasound examination |
| Voiding difficulty | Evaluate immediately if voiding difficulty has no identifiable cause. | Abdominal ultrasound examination |
| Lymphadenopathy (> 2 cm) | Evaluate if lymphadenopathy does not respond to a 7-day course of antibiotic. | CBC with differential, lactate dehydrogenase level |
CBC = complete blood count; CT = computed tomography.
The presenting manifestations of leukemia are related to bone marrow infiltration and resultant pancytopenia. These symptoms include the following: fatigue and pallor secondary to anemia; petechiae, ecchymoses and mucus membrane bleeding secondary to thrombocytopenia; signs of infection secondary to neutropenia; and bone pain secondary to marrow hyperplasia. Constitutional symptoms, including fever, anorexia and weight loss, are also quite common.
The signs and symptoms of solid tumors are related to the presence of a mass lesion. CNS tumors can present with headache alone. (Clearly, however, most childhood headaches are not caused by brain tumors.) Headaches associated with neurologic abnormalities, such as ataxia, gait disturbances, seizures or cranial nerve palsies, merit further investigation.
Abdominal masses can present with pain, vomiting, constipation or, less commonly, intestinal obstruction. It is not unusual for a child with an abdominal mass to present because a family member notices a protuberant mass. For masses in the chest, the presenting symptoms are usually persistent cough or dyspnea.
Extremity lesions usually present with pain subsequent to trauma. It is important to realize that the traumatic event merely leads to careful examination of the extremity (including diagnostic imaging) and may not be causing the pain.
Leukemia
Leukemia is the most common malignancy of childhood, and acute lymphoblastic leukemia is the most common type of leukemia in children. Acute lymphoblastic leukemia typically develops in children between one and 10 years of age, although it can occur at any age. This leukemia is more common in males and in whites.1
Diagnosing acute lymphoblastic leukemia can be difficult. Frequently, the diagnosis is delayed because early symptoms are nonspecific and may mimic those of viral infections. Most children who have this cancer present with generalized malaise, loss of appetite and a low-grade fever. Additional symptoms that should prompt concern include pallor, petechiae or ecchymoses, bone pain and significant weight loss.
The physical examination may reveal no abnormalities, but the presence of significant lymphadenopathy or any hepatosplenomegaly should raise suspicion for leukemia. Compared with adults, children normally have more lymphoid tissue in their tonsils, adenoids and cervical regions; however, hepatosplenomegaly is always an abnormal finding.
A prudent approach to the child with any suspicious findings is to obtain a complete blood count (CBC) with a white blood cell differential and a reticulocyte count. The presence of blast cells on the peripheral smear is indicative of leukemia. However, many patients with leukemia only have blast cells in their bone marrow. The finding of anemia, especially if accompanied by reticulocytopenia or a high mean corpuscular volume, thrombocytopenia, leukopenia or leukocytosis, should prompt consultation with a pediatric hematologist or oncologist, because the likelihood of leukemia is high.
The presenting signs and symptoms of childhood leukemia are listed in Table 4.4 Specific illnesses that often mimic leukemia include infectious mononucleosis caused by Epstein-Barr virus or, less frequently, cytomegalovirus infection, collagen vascular disease and aplastic anemia. The differential diagnosis for an abnormal CBC is given in Table 5.
TABLE 5 Differential Diagnosis of Abnormal Complete Blood Count
| Infections |
| Nonspecific viral syndrome |
| Epstein-Barr virus infection |
| Cytomegalovirus infection |
| Human immunodeficiency virus infection |
| Autoimmune disorders |
| Systemic lupus erythematosus |
| Systemic-onset juvenile rheumatoid arthritis |
| Hematologic disorders |
| Aplastic anemia |
| Leukemia |
Central Nervous System Tumors
CNS tumors are the second most common childhood malignancy. Many brain tumors are histologically benign but are considered “malignant” because they cause severe symptoms and require treatment secondary to their anatomic location. CNS tumors are classified as intracranial or spinal. In adults, the majority of intracranial tumors are supratentorial. However, infratentorial lesions account for 60 percent of CNS tumors in children.5
Children who have infratentorial lesions usually present with ataxia and other gait disturbances, frequently have hydrocephalus as a result of aqueduct compression and may also have cranial nerve abnormalities from brainstem compression. Figure 1 shows a typical posterior fossa mass (ependymoma) in a six-year-old child.
FIGURE 1.
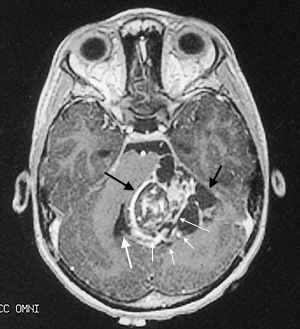
Ependymoma. This axial T1-weighted gadolinium-enhanced magnetic resonance image of the brain demonstrates an eccentric cerebellar mass (black arrows) with heterogeneous enhancement. The mass appears centered in the left cerebellar hemisphere and is compressing the fourth ventricle (large white arrow). Focal areas of signal void correspond to calcifications (small white arrows).
Supratentorial tumors occur at any age. These lesions can present with signs of elevated intracranial pressure (headache and vomiting, or an enlarging head in infants) and focal neurologic deficits. Less common findings include seizures, endocrine abnormalities and personality changes.
Spinal tumors are more common in older children. Malignancies of the spine frequently present as back pain and signs of spinal cord compression, such as weakness and loss of bladder and bowel function.
Headaches are not uncommon in children, and most headaches are not caused by brain tumors. Sleep-related headaches and the absence of a family history of migraine are the strongest predictors that a headache is caused by a space-occupying lesion. Other predictors include vomiting, confusion, neurologic signs, absence of visual symptoms and a duration of less than six months.6 Diagnostic imaging of the brain is recommended if headaches are awakening a child from sleep, are associated with neurologic signs (including seizures) or occur with vomiting in the absence of a family history of migraine.
Back pain is also a common complaint in children. The sudden onset of back pain of short duration most likely represents a muscle injury. However, examination of the spine, preferably with magnetic resonance imaging (MRI), is critical when a child has back pain that is persistent or worsens when the child is supine, or when back pain is accompanied by signs of spinal cord compression or peripheral neuropathy. Standard radiographs of the spine can easily miss a spinal tumor.
Lymphoma
Both Hodgkin's disease and non-Hodgkin's lymphoma occur in children. These malignant conditions present at different ages and with different symptoms.
HODGKIN'S DISEASE
The incidence of Hodgkin's disease increases throughout childhood and peaks in the late teenage years.3 This form of malignant lymphoma most often presents with a painless mass in the neck. Other presentations include a persistent cough secondary to a mediastinal mass (Figures 2 and 3) or, less commonly, splenomegaly or enlarged axillary or inguinal lymph nodes.
FIGURE 2.
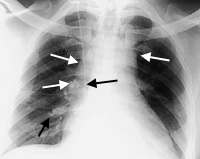
Hodgkin's disease. The chest radiograph demonstrates a lobular configuration of the hilar regions (white arrows) secondary to partially calcified lymph nodes (black arrows).
FIGURE 3.
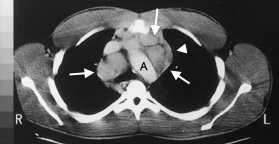
Hodgkin's disease. This contrast-enhanced computed tomographic scan of the patient in Figure 2 better delineates the extensive anterior mediastinal adenopathy (large arrows) from the thymus (arrowhead) and aorta (A).
Approximately 30 percent of children with Hodgkin's disease present with constitutional symptoms, the so-called “B” symptoms. These symptoms may include intermittent fever, drenching night sweats and loss of greater than 10 percent of total body weight.7,8 Other symptoms include anorexia, fatigue and pruritus.
Any persistent painless mass (especially a neck mass) that does not respond to antibiotics should be investigated. This investigation should include a lymph node biopsy. Because of sampling errors and difficulties in obtaining an accurate diagnosis, excisional biopsy (rather than needle biopsy) of enlarged lymph nodes should be performed.
A persistent cough, especially in the presence of any “B” symptoms, should be investigated. As part of this investigation, a chest radiograph should be obtained.
It is also important to investigate “B” symptoms associated with any lymphadenopathy or splenomegaly. Laboratory tests can often be helpful in ambiguous cases. Although nonspecific, elevations in the erythrocyte sedimentation rate, lactate dehydrogenase level and ferritin level are suspicious findings in children with other signs or symptoms of Hodgkin's disease. Infrequently, the CBC reveals abnormalities, including anemia and eosinophilia.
NON-HODGKIN'S LYMPHOMA
Non-Hodgkin's lymphoma is more common in older children, but it is less common than Hodgkin's disease. In general, non-Hodgkin's lymphoma is more aggressive (and more responsive to therapy) in children than in adults. The most frequent sites of this malignancy are the abdomen, mediastinum and head and neck.
When the abdomen is affected, the most common presenting features are abdominal pain with vomiting or diarrhea, a palpable mass and intussusception.9 In the absence of a palpable mass, persistent abdominal pain with vomiting or diarrhea, especially when accompanied by significant weight loss, should prompt a thorough investigation, including diagnostic imaging. Unless proved otherwise, lymphoma should be considered the cause of intussusception in a child more than six years of age. Intussusception requires surgical intervention.
Non-Hodgkin's lymphoma involving the mediastinum generally progresses more rapidly than Hodgkin's lymphoma. A child with this malignancy can present acutely with severe dyspnea and, occasionally, superior vena cava syndrome. These are unusual presentations for Hodgkin's disease.
In children with head and neck masses, the presenting sign can be a palpable mass, cranial nerve palsies or nasal obstruction.
Blood laboratory studies can be helpful in diagnosing non-Hodgkin's lymphoma. A child with a large mass frequently has an extremely elevated serum lactate dehydrogenase level.
IMAGING
The same imaging modalities are used for Hodgkin's disease and non-Hodgkin's lymphoma. In the child with a persistent cough, a chest radiograph may reveal a mediastinal mass or mediastinal widening. Abdominal masses are best imaged with computed tomographic (CT) scanning, and head and neck lesions can be imaged with MRI or CT studies.
Scanning performed with gallium-67 citrate can be helpful in evaluating a child with systemic symptoms of lymphoma but no signs of a mass. However, this radiopharmaceutical agent cannot differentiate a malignant lesion from an inflammatory one, and it is not 100 percent sensitive.
Neuroblastoma
Neuroblastoma, a tumor of the sympathetic nervous system, is the most common extracranial solid tumor of childhood. It accounts for 8.2 percent of all cancers diagnosed in children less than 15 years of age and for 15 percent of all cancers in children less than five years of age.1,3 Approximately 850 new cases of neuroblastoma occur annually in the United States.10
The signs and symptoms of neuroblastoma are a function of the location of the primary tumor (which may occur anywhere along the peripheral sympathetic nervous system) and the sites of metastatic disease. Up to 65 percent of primary tumors arise in the abdomen11 (Figure 4). Signs and symptoms include anorexia and abdominal pain and distention.
FIGURE 4.
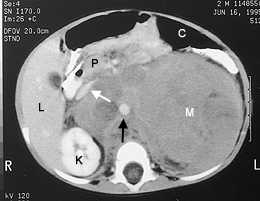
Neuroblastoma. A contrast-enhanced CT scan of the abdomen demonstrates a large, heterogeneously enhancing upper retroperitoneal mass (M) that displaces the normal left kidney caudally (not seen); the abdominal aorta (black arrow) and pancreas (P) are displaced anteriorly. Although calcifications are commonly detected on CT scans of neuroblastomas, this scan shows several low-density areas that suggest the presence of necrosis. The inferior vena cava is also visualized (white arrow). (L = liver; C = transverse colon; K = normal right kidney)
Primary thoracic tumors account for 20 percent of neuroblastomas in children.11 These tumors are often diagnosed incidentally during the evaluation of trauma, infection or respiratory symptoms. Large masses often cause respiratory distress. Children with high thoracic and cervical tumors may present with Horner's syndrome. Paraspinal tumors in the cervical, thoracic or abdominal regions may extend into the neural foramina and cause signs and symptoms of nerve root or spinal cord compression.
Bone and bone marrow metastases are common. These metastases cause bone pain and, possibly, signs of bone marrow failure (i.e., anemia and purpura). The skull is a frequent site of bone metastases, which commonly present as proptosis and periorbital ecchymoses.
Paraneoplastic phenomena, including hypertension, secretory diarrhea and opsoclonusmyoclonus, are occasional presenting signs and symptoms of neuroblastomas.
The prognosis is excellent for infants at all stages of neuroblastoma and for children with limited-stage disease. Despite aggressive multimodality therapy, including bone marrow transplantation, the outcome is poor for children with advanced-stage disease.12 Newer biologic therapies may improve the outlook for these patients.13
Wilms' Tumor
Wilms' tumor should be considered in the differential diagnosis of an abdominal mass in a child. The mean age at diagnosis is 41.5 months for males and 46.9 months for females.1 Wilms' tumor can present (albeit rarely) as an abdominal mass in newborns, and it occasionally develops in adolescents. This malignancy can also present with abdominal pain, gross hematuria, fever and hypertension.
When an abdominal mass is detected in a child, ultrasound examination of the abdomen should be performed within 24 hours by a radiologist with pediatric experience. If the ultrasound examination does not clearly identify the origin of the mass, abdominal CT scanning should be performed. If the mass is confirmed to be of renal origin, immediate referral to a pediatric oncologist and a pediatric surgeon is indicated.
Wilms' tumor occurs with increased frequency in patients with aniridia, hemihypertrophy, cryptorchidism, hypospadias and other genitourinary abnormalities. The risk of Wilms' tumor is markedly increased in children with Beckwith-Wiedemann syndrome, Drash syndrome, Bloom syndrome and WAGR syndrome (Wilms' tumor, aniridia, genitourinary malformation and mental retardation).14,15 Annual renal ultrasound examination is recommended for these patients.14,15
A Wilms' tumor can rupture, putting the patient at risk for significant abdominal hemorrhage and peritoneal seeding of the tumor. If rupture occurs, expedient surgical intervention by a surgeon with specific expertise in removal of this fragile tumor is necessary to ensure an optimal outcome.
Malignancies of the Musculoskeletal System
A tumor that arises in the musculoskeletal system often presents as a mass, a painful extremity or, occasionally, a pathologic fracture. Because musculoskeletal pain is a frequent symptom in children, it can be difficult to determine when a more comprehensive evaluation is warranted.
A malignancy should be suspected if pain awakens a child at night, causes significant extremity dysfunction (when trauma is not involved) or is accompanied by a nontraumatic mass. Any fracture that presents with a soft tissue mass or injury out of proportion to the traumatic event should be considered a possible malignancy. Malignancy, including leukemia, should also be suspected if extremity pain is present for two weeks without a clear cause.
The initial evaluation of these signs and symptoms includes standard radiography (Figure 5), bone scanning and CT scanning. When the presence of a bone or soft tissue malignancy is considered in a child or an adolescent, input from a pediatric oncologist and an orthopedic oncologist is critical before a biopsy is performed. Limb-saving surgery depends on an appropriate biopsy decision.
FIGURE 5.
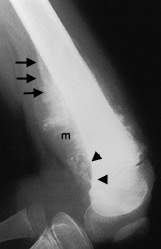
Osteosarcoma. This lateral radiograph of the distal femur demonstrates a large, heterogeneously ossified mass (m) centered about the metaphysis and extending into the overlying soft tissues. Angular periosteal elevation (black arrows) and cortical destruction (arrowheads) of the distal metaphysis are noted. The fluffy ossification of the mass is typical of osteosarcoma.
The three most common musculoskeletal tumors in children and adolescents are rhabdomyosarcoma, osteosarcoma and the Ewing's sarcoma family of tumors.3 Rhabdomyosarcoma affects the broadest age range of patients. In young children, this tumor often occurs as an orbital, paranasal sinus or genitourinary lesion. In adolescents, rhabdomyosarcoma more often occurs as a tumor of skeletal muscle in an extremity.
Osteosarcoma is the most common bone tumor. It generally develops during puberty and occurs primarily in the distal femur and proximal tibia.
The age distribution of Ewing's sarcomas is similar to that of osteosarcomas. However, Ewing's sarcomas have been known to occur in children as young as three years of age and in adults well beyond 30 years of age. In addition to occurring in the extremities, these tumors often develop in the bone and soft tissue of the central axis, including the skull, vertebral bodies, rib cage, skull, abdomen and pelvis. Some patients with Ewing's sarcoma may present with an isolated soft tissue mass without evidence of bone involvement.
Recent advances in the molecular genetics of rhabdomyosarcoma and Ewing's sarcomas have allowed more accurate diagnosis of these tumors and better determination of the prognosis for affected patients. Samples of frozen tissue are secured at the time of surgery and then analyzed for specific molecular abnormalities.
Genetic Syndromes Associated with Cancer
Although the cause of cancer remains unknown in most children, a number of chromosomal disorders or constitutional syndromes are associated with an increased risk of malignancy.16 Some of the more common genetic disorders, such as Down syndrome and neurofibromatosis, are associated with an increased risk of leukemia, particularly myeloid leukemia. Other disorders carrying an increased risk of leukemia include a variety of immunodeficiency disorders, such as Wiskott-Aldrich syndrome and common variable immunodeficiency, as well as DNA repair disorders such as ataxiatelangiectasia syndrome, Bloom syndrome and Fanconi's syndrome.
The neurocutaneous syndromes, including neurofibromatosis, tuberous sclerosis and von Hippel-Landau disease, are associated with an increased risk of brain tumors. Gastrointestinal syndromes, such as familial polyposis coli and Gardner's syndrome, carry a greater risk of colon cancer.
If a child with one of these syndromes or disorders presents with signs or symptoms even remotely suggestive of a malignant process, the primary care physician should have a high index of suspicion and initiate appropriate diagnostic studies.
A recent comprehensive review of familial cancer syndromes is available,17 and a number of tertiary care medical centers have interdisciplinary genetic cancer centers. Further information can also be obtained on the Internet from the National Cancer Institute Familial Search Database (Web site: http://cancernet.nci.nih.gov/wwwprot/genetic/genesrch.html).
Final Comment
The presentations of cancers in children frequently mimic those of common childhood conditions. Therefore, deciding whether a child requires further investigation can be challenging. If the diagnosis of cancer is suspected, consultation with a pediatric hematologist or oncologist is indicated. Because childhood cancers are significantly different from adult cancers, referral to an adult oncologist can lead to inaccurate diagnosis and result in inappropriate treatment.
The combined medical expertise of surgeons, oncologists, pathologists, radiologists and radiation therapists is necessary for the diagnosis and treatment of childhood cancers. The primary care physician has the task of coordinating referrals to the appropriate consultants.
Pediatric oncologists routinely provide educational materials and referrals to social workers and psychologists who specialize in childhood cancer. The Candlelighters Childhood Cancer Foundation (7910 Woodmont Ave., Suite 460, Bethesda, MD 20814; telephone number: 800-366-2223; Web site: www.candle.org) is a charitable organization that provides educational materials to families, support groups for parents and patients, and funding for special needs. Parents can also find educational material and information on support groups from the Pediatric Oncology Resource Center (http://www.ped-onc.org/) and OncoLink (http://oncolink.upenn.edu/). Families who live far from cancer centers can usually obtain temporary housing at Ronald McDonald houses.
Children with cancer and their families benefit from early referral and a close working relationship between the oncology team and the primary care physician. In addition to providing or arranging psychosocial support, the primary care physician can assist in numerous aspects of care, ranging from the evaluation of complications such as infections to the administration of outpatient chemotherapy in the form of intramuscular and subcutaneous injections.
Finally, it is important to consider referral to clinical research consortia that provide care for children with cancer, such as the Pediatric Oncology Group and the Children's Cancer Group. Pediatric oncology centers belonging to these groups can provide the most up-to-date therapies. Of note, these groups will merge in the year 2000 to form the Children's Oncology Group.
The outcome for children with cancer is significantly better than it was 25 years ago (Table 6).3 At that time, the pediatric oncology consortia began an era of collaboration on the treatment and research of pediatric cancer. As a result of this collaborative approach, the overall survival rate for children with cancer has increased from 28 percent in the early 1960s to more than 70 percent in the 1990s.3,18

