Acute pancreatitis usually occurs as a result of alcohol abuse or bile duct obstruction. A careful review of the patient's history and appropriate laboratory studies can help the physician identify the etiology of the condition and guide management. Serum amylase and lipase levels are still used to confirm the diagnosis of acute pancreatitis. Although not routinely available, the serum trypsin level is the most accurate laboratory indicator for pancreatitis. Ultrasonography, computed tomography and endoscopic retrograde cholangiopancreatography are additional modalities that can help the family physician choose the best treatment approach. Prompt identification of patients who need intensive care referral or subspecialty consultation is crucial. The APACHE II and the multiple organ system failure scales provide prognostic information at the time of admission and may be repeated daily to monitor disease progression. Therapies such as nasogastric suctioning, anticholinergics and histamine H2-receptor blockers have not been shown to decrease symptoms or hospital stays in patients with acute pancreatitis. Systemic antibiotics have been found to improve outcome in patients with severe disease. With supportive care, most patients have a good clinical outcome.
The increased incidence of pancreatitis, coupled with new treatment options, poses a challenge for primary care physicians. Between 1960 and 1980, the incidence of acute pancreatitis increased 10-fold. Mortality secondary to pancreatitis ranges from 2 to 9 percent.1 Primary care physicians must make the diagnosis, determine etiology, provide supportive therapy and categorize the severity of pancreatitis to choose the best treatment approach. Although acute pancreatitis has numerous causes (Table 1),1 this article focuses on the two most common causes—alcohol abuse and biliary tract obstruction related to cholelithiasis (Figure 1). These two conditions account for 60 to 80 percent of all cases of acute pancreatitis.2 Diagnosis, risk assessment and formulation of a management plan are discussed.
FIGURE 1.
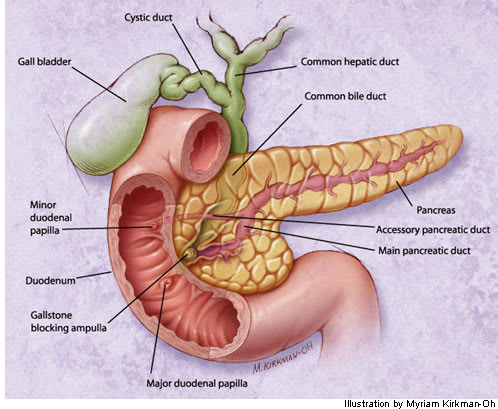
Biliary tract obstruction caused by a gallstone in the distal common bile duct.
Presentation
Patients with acute pancreatitis present with mild to severe epigastric pain, with radiation to the flank, the back, or both. Classically, the pain is characterized as constant, dull and boring, and is worse when the patient is supine. The discomfort may lessen when the patient assumes a sitting or fetal position. A heavy meal or drinking binge often triggers the pain. Nausea and nonfeculent vomiting are present in 75 to 90 percent of patients.3
Diagnosis
The clinical diagnosis of pancreatitis is difficult to make and is frequently missed. One review cited a misdiagnosis rate as high as 43 percent; in some cases, the diagnosis is made at autopsy.4 A history and physical examination suggestive of pancreatitis warrant further diagnostic work-up.
HISTORY
Patients as young as the early 30s can present with acute pancreatitis secondary to alcohol consumption. The estimated consumption of alcohol per day in patients with pancreatitis is about 150 g, but pancreatitis can occur in persons consuming only 50 g per day (approximately four 12-oz servings of beer of 3 to 5 percent alcohol content).5 On average, when alcohol-related pancreatitis occurs, it develops after four to seven years of drinking.6 In contrast, biliary pancreatitis usually occurs in older adults.7 These patients often have a history of cholelithiasis or intermittent, post-prandial right upper-quadrant pain.
PHYSICAL EXAMINATION
The spectrum of severity of acute pancreatitis is reflected on physical examination. Between 50 and 90 percent of patients have signs of abdominal distension or muscle spasms with epigastric pain and left upper-quadrant tenderness.3,6 Other signs are fever, tachycardia and jaundice. Often the patient is restless and dehydrated on presentation.3,4
LABORATORY DIAGNOSIS
Amylase. Serum amylase levels in patients with pancreatitis vary depending on the severity of the disease. On average, during uncomplicated cases, the serum amylase level starts increasing from two to 12 hours after the onset of symptoms and peaks at 12 to 72 hours. It usually returns to normal within one week.8,9 Although it lacks sensitivity (75 to 92 percent) and specificity (20 to 60 percent), measurement of the serum amylase level is the most widely used method of diagnosing pancreatitis. The advantages of amylase testing are that it is quickly performed, easily obtained and inexpensive.10 However, a variety of nonpancreatic conditions cause increased amylase levels4 (Table 2).11
Lipase. Lipase levels increase within four to eight hours of the onset of clinical symptoms and peak at about 24 hours. Levels decrease within eight to 14 days. The specificity (50 to 99 percent) and sensitivity (86 to 100 percent) of lipase measurements are better than those of amylase measurement, particularly in detecting alcoholic pancreatitis.10 The specificity of lipase measurement, as well as amylase measurement, may be improved by raising the threshold to at least three times the upper limit of the normal reference values.11
Trypsin/Elastase. Based on median sensitivities and specificities, an elevated trypsin level has a better likelihood ratio for detecting pancreatitis than the amylase level and is probably the most accurate serum indicator for acute pancreatitis.12 The elastase level has not proved to be better than trypsin or lipase levels in assisting the diagnosis of acute pancreatitis. However, a serum trypsin assay is not widely available and therefore is not routinely used.
Hepatic Function Studies. Hepatic transaminase levels may be elevated in patients with pancreatitis caused by alcohol abuse or cholelithiasis with obstruction. However, these tests are not sufficiently reliable for diagnosing acute biliary pancreatitis or determining its etiology.
Radiologic Studies
Plain Radiographs. Plain radiographs may support the diagnosis of acute pancreatitis when certain findings are present (Table 3).4 Of these findings, a gas-filled duodenum (sentinel loop) secondary to obstruction is the most specific for pancreatitis.4 However, none of the radiologic abnormalities on plain films can be used for specific diagnostic purposes.
Ultrasonography. Ultrasonography is an acceptable study for initial evaluation when biliary causes are suspected. Pancreatic ultrasonography has these advantages: it is noninvasive, relatively inexpensive and may be performed at the bedside. The sensitivity of this study in detecting pancreatitis is 62 to 95 percent.13,14 However, in 35 percent of cases, the pancreas is obscured secondary to bowel gas.10
Computed Tomography (CT). The contrast-enhanced CT scan provides the best imaging of the pancreas and surrounding structures. A CT study may be useful when other diagnostic studies are inconclusive, when the patient has severe symptoms, when fever is present or in the face of persistent leukocytosis that suggests secondary infection.10 In addition, CT scanning is especially helpful in assessing complications related to acute pancreatitis or as a follow-up study in patients who are clinically deteriorating. The CT findings in pancreatitis may show inflammation characterized by diffuse or segmental enlargement of the pancreas, with irregular contour and obliteration of peripancreatic fat, necrosis or a pseudocyst15 (Figures 2 through 4).
FIGURE 2.
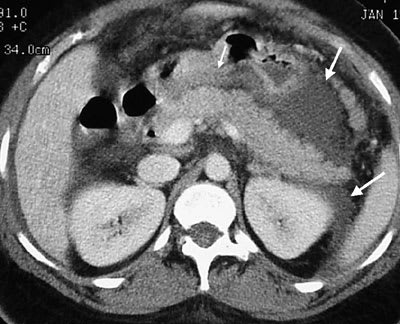
Contrast-enhanced axial computed tomographic section of the upper abdomen showing peripancreatic and retroperitoneal edema (large arrows) and stranding. The pancreas itself (small arrow) appears relatively normal.
FIGURE 3.
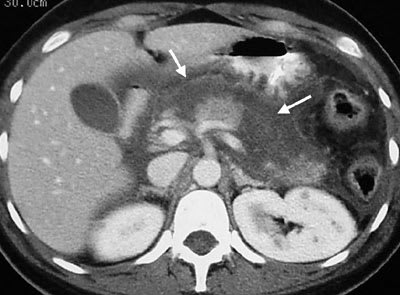
Contrast-enhanced axial computed tomographic section of the upper abdomen showing peripancreatic and retroperitoneal edema. Large non-enhancing areas of necrosis are visible in the body and neck of the pancreas (arrows).
FIGURE 4.
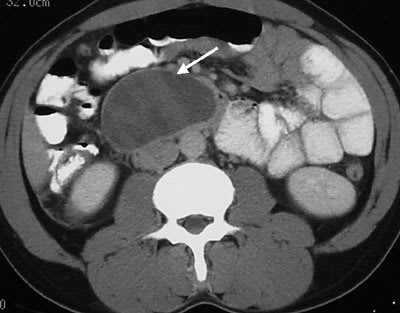
Contrast-enhanced axial computed tomographic section of the upper abdomen showing a well-defined fluid collection in the retroperitoneum (arrow) just below the level of the pancreas.
Endoscopic Retrograde Cholangiopancreatography (ERCP). ERCP has a limited role in management of acute pancreatitis. It is primarily indicated in patients with severe disease who are suspected of having biliary obstruction.16 This procedure is sometimes done to enable endoscopic sphincterotomy and remove impacted stones. The risks of performing ERCP with sphincterotomy include precipitating an acute episode of pancreatitis, introducing infection and causing hemorrhage and perforation. At least one study has shown that patients with severe biliary pancreatitis show a reduction in morbidity and mortality with early (less than 24 hours) ERCP.17
Determining Prognosis
About 20 to 30 percent of patients with acute pancreatitis develop complications of necrosis, organ failure, or both18–20 (Table 4).21 Unfortunately, the serum amylase level and the lipase level are not specific enough measures of disease activity to be used prognostically.
TABLE 4 Potential Complications of Acute Pancreatitis
| Hypovolemia |
| Pancreatic necrosis |
| Extrapancreatic necrosis |
| Acute respiratory distress syndrome |
| Acute renal failure |
| Adynamic ileus |
| Circulatory shock |
| Sepsis |
Information from Beger HG, Rau B, Mayer J, Pralle U. Natural course of acute pancreatitis. World J Surg 1997;21:130–5.
Clinical monitoring is inadequate for determining severity and predicting the course of pancreatitis because it only detects about 39 percent of severe cases.22 Thus, assessment of progress requires careful monitoring of clinical signs and symptoms, including pain and nausea, fever (greater than 38.6°C [101.5°F]), ascites, ecchymosis, laboratory assessments (specifically, C-reactive protein level) and the use of severity classifications systems.
Several systems have been developed in an attempt to provide reliable prognostic classification for patients with acute pancreatitis. However, two systems, the APACHE II scale (Table 5)21 and the multiple organ system failure (MOSF) scale (Table 6)23 have some advantages over the Ranson criteria (Table 7),4 the Imrie classification and clinical evaluation.23 The MOSF and APACHE II scales can usually be performed within a few hours after admission. In addition, they can be repeated daily to monitor disease progression.4,23,24 A direct association has been made between a five-point increase in APACHE II scores and intensive care unit death rates. One study23,24 evaluated mortality rates and APACHE II scores in ICU admissions from 13 hospitals from 1979 through 1981. A score of 30 to 34 was associated with 73 percent mortality, while patients with scores above 35 had an 84 percent mortality.
TABLE 5 APACHE II Scoring System
| Feature | Acute physiology score (APS) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Variable | + 4 | + 3 | + 2 | + 1 | 0 | + 1 | + 2 | + 3 | + 4 |
| Temperature | ≥41 | 30 to 40.9 | 38.5 to 38.9 | 36 to 38.4 | 34 to 35.9 | 32 to 33.9 | 30 to 31.9 | ≤29.9 | |
| Mean arterial BP | ≥160 | 130 to 159 | 110 to 129 | 70 to 109 | 50 to 69 | < 49 | |||
| Heart rate | ≥180 | 140 to 179 | 110 to 139 | 70 to 109 | 55 to 69 | 40 to 54 | ≤39 | ||
| Respiratory rate | ≥50 | 35 to 49 | 25 to 34 | 12 to 24 | 10 to 11 | 6 to 9 | < 5 | ||
| A-aPo2* | ≥500 | 350 to 499 | 200 to 349 | < 1 00 | 61 to 70 | 55 to 60 | < 55 | ||
| Pao2† | > 70 | ||||||||
| Arterial pH | ≥7.7 | 7.6 to 7.69 | 7.5 to 7.59 | 7.33 to 7.49 | 7.25 to 7.32 | 7.15 to 7.24 | < 7.15 | ||
| Serum bicarbonate‡ | ≥52 | 41 to 51.9 | 32 to 40.9 | 23 to 31.9 | 18 to 21.9 | 15 to 17.9 | < 15 | ||
| Serum sodium | ≥180 | 160 to 179 | 155 to 159 | 150 to 154 | 130 to 149 | 120 to 129 | 111 to 119 | ≤ 110 | |
| Serum potassium | ≥7 | 6 to 6.9 | 5.5 to 5.9 | 3.5 to 5.4 | 3 to 3.4 | 2.5 to 2.9 | < 2.5 | ||
| Serum creatinine | ≥3.5 | 2 to 3.4 | 1.5 to 1.9 | 0.6 to 1.4 | < 0.6 | ||||
| Hematocrit | ≥60 | 50 to 59.9 | 46 to 49.9 | 30 to 45.9 | 20 to 29.9 | < 20 | |||
| WBC count | ≥40 | 20 to 39.9 | 15 to 19.9 | 3 to 14.9 | 1 to 2.9 | < 1 | |||
BP = blood pressure; A-aPo2= alveolar-arterial oxygen pressure; Pao2= partial pressure of oxygen in arterial blood; WBC = white blood cell.
* —Use if percentage of inspired oxygen (Fio2) > 50 percent.
† —Use if Fio2< 50 percent.
‡ —Use only if no arterial blood gas measurements are available.
Age points (AP)
Chronic health problems (CHP)
Scoring
Age
Points
For patients with history of severe organ system insufficiency or immunocompromise, assign points as follows:
APS + AP + CHP = total score
≤44
0
45 to 54
2
Nonoperative or emergency postoperative: 5 points
Elective postoperative: 2 points
55 to 64
3
65 to 74
5
≥75
6
Scores indicating abnormal reading: on admission, > 9; after 24 hours, > 10; after 48 hours, > 9.
Of the two systems, MOSF has several advantages over APACHE II. The APACHE II system is more complex and necessitates the collection of a variety of laboratory data. In addition, the MOSF system has better clinical utility for evaluating patients at admission and at 48 hours.23
Complications
Most complications of acute pancreatitis and subsequent deaths occur within two weeks of onset of pain. Secondary pancreatic infection is the most common cause of death in acute pancreatitis, accounting for 70 to 80 percent of deaths.1,21 Complications frequently manifest as necrosis and organ failure, which often includes the cardiovascular, pulmonary and renal systems (Table 5).21 Cardiovascular complications may reflect bleeding into the retroperitoneal space and decreased vascular resistance. Pulmonary insufficiency may range from mild atelectasis to life-threatening adult respiratory distress syndrome. Acute renal failure defined as a twofold creatinine rise may ensue secondary to cardiovascular collapse and hypotension, resulting in acute tubular necrosis.23
CT scanning may detect late complications of pancreatitis. Complications that usually occur after three weeks include pseudocysts and abscess formation. Pseudocysts occur in about 1 to 8 percent of cases. Abscesses occur in 1 to 4 percent of patients.1
Management
The treatment of pancreatitis may be conservative or complex, depending on the severity of the presentation and the development of complications. A patient with a history and physical examination consistent with pancreatitis should have laboratory studies, including determination of amylase, lipase and, if available, serum trypsin levels to confirm the diagnosis. Additional tests that are helpful are a complete blood cell count and hepatic function tests. Most authorities recommend that radiographs of the chest and abdomen be obtained on presentation to rule out bowel perforation or early pulmonary complications. Ultrasonography is indicated only if the clinical presentation or laboratory assessment suggests biliary disease. Baseline CT scanning is indicated in the following situations: (1) the diagnosis is in doubt; (2) severe pancreatitis is suspected because of high fever (higher than 38.8° C [102°F]), distension and leukocytosis; or (3) the patient has an elevated severity score as determined by the MOSF or APACHE II criteria.15 Intravenous rehydration should usually be aggressive, with close attention to blood pressure, and cardiac and pulmonary status. In addressing the patient's pain, analgesia with meperidine (Demerol) along with an antiemetic is preferred over the use of morphine, because morphine may cause spasm of the sphincter of Oddi, which has the potential to worsen the condition.2 Admission to the intensive care unit should be considered in the presence of organ dysfunction or other poor prognostic signs.25
In the past, patients were routinely treated by withholding food and placing a nasogastric tube with suction for two to 10 days. This was done in an attempt to reduce pancreatic stimulation by food, hydrochloric acid, cholecystokinin and secretin.3 Withholding food by mouth does reduce pain, but use of a nasogastric tube with suction is no longer advocated as a routine therapeutic measure in acute pancreatitis because it has not been shown to decrease symptoms, mortality or hospital stay. However, a nasogastric tube may be used when the patient has protracted vomiting or if obstruction is seen on the abdominal radiograph.1,3,8,25 In mild pancreatitis, oral intake should be withheld until the nausea and vomiting subside. Total enteral feeding beyond the ligament of Treitz administered within 48 hours of onset of severe acute pancreatitis may reduce the incidence of total and infectious complications.26
If the patient's history is suggestive of biliary disease or in the presence of jaundice or elevated aspartate aminotransferase or alanine aminotransferase levels, a right upper-quadrant sonogram should be obtained. If cholelithiasis is present or there is a dilated common bile duct, the etiology may be an impacted stone in the common bowel duct or ampule. ERCP should be performed on a finding of dilated common bile duct or failure to improve within 48 hours after supportive care has been administered.
Anticholinergics have been used in an attempt to decrease gastric secretions and increase pH. As with the use of nasogastric tubes, anticholinergics do not decrease hospital stay or pain. Other treatments that have been studied but that show no clear benefit include atropine (Urised), glucagon, calcitonin (Calcimar) and protease inhibitors. The use of gabexate and somatostatin (Zecnil), which decrease pancreatic secretions, shows a trend toward favorable outcomes in small, uncontrolled studies.27,28 However, they still cannot be recommended for routine treatment of acute pancreatitis; furthermore, gabexate is not available in the United States.25 Essentially, no currently proven medical therapies for acute pancreatitis exist.
ANTIBIOTICS
The role of antibiotics in the management of early acute pancreatitis is controversial. Although some studies25 have shown decreased mortality with the use of antibiotics, the severity of complications makes it difficult to perform unblinded studies. Control patients who decompensate receive more surgical procedures, which may contribute to infection and bias the studies.25 However, intravenous or gut-sterilizing oral antibiotics are recommended for use in patients with acute necrotizing pancreatitis.29 Antibiotics that show promising results (Table 8)1,30–34 include imipenemcilastatin (Primaxin), cefuroxime (Zinacef) and ceftazidime (Ceptaz).
TABLE 8 Therapies for Acute Pancreatitis
| Treatment | Mechanism of action | Improved outcome/number of studies | |
|---|---|---|---|
| Nasogastric suction | Decreases pancreatic secretions | 0/3 | |
| Histamine H2-receptor blocker | Decreases pancreatic secretions | 0/3 | |
| Atropine (Urised) | Decreases pancreatic secretions | 0/1 | |
| Glucagon | Decreases pancreatic secretions | 0/4 | |
| Somatostatin (Zecnil) | Decreases pancreatic secretions | 3/5* | |
| Calcitonin (Calcimar) | Decreases pancreatic secretions | 0/1 | |
| Antibiotics | Prevent bacterial infections | 5/8 | |
| Ampicillin | 0/3 | ||
| Imipenem-cilastatin (Primaxin) | 2/2 | ||
| Cefuroxime (Zinacef) | 1/1 | ||
| Ceftazidime (Ceptaz) plus amikacin (Amikin) plus metronidazole (Flagyl) | 1/1 | ||
| Fresh frozen plasma | Inhibits proteases | 0/1 | |
| Total parenteral nutrition | Decreases pancreatic secretions | 0/1 | |
| Peritoneal lavage | Removes toxic factors | ||
| 2 to 4 days | 0/3 | ||
| 7 days | 1/1 | ||
*— Meta-analysis.
If necrosis is identified on CT scan, or if the patient continues to exhibit signs of infection (fever, tachycardia, abdominal pain, leukocytosis), percutaneous fine-needle aspiration should be performed to tap necrotic tissue and peripancreatic fluid. Patients with necrosis and aspirates positive for bacteria on Gram stain or culture should undergo surgical debridement. The necessity for intervention in patients with sterile necrosis is unclear. In the few studies35–37 that compare surgical debridement to medical management in sterile necrosis, surgical debridement has not been shown to improve outcome. This is true even in the presence of multisystem organ failure.21 However, aggressive surgical debridement is the standard of care for infected necrosis.38 If debridement is undertaken, it should be delayed as long as possible because survival improves with time.39
Final Comment
Acute pancreatitis may range from a mild to a life-threatening condition. Establishing a rapid diagnosis through laboratory testing and radiologic studies is important. In patients with signs of severe disease at onset or clinical deterioration after hospital admission, consultation with a gastroenterologist and possibly a surgeon is recommended. Even with aggressive and appropriate care, acute necrotizing pancreatitis has a mortality rate of 30 percent.

