
This is a corrected version of the article that appeared in print.
Am Fam Physician. 2000;62(5):1064-1070
A more recent article on degenerative cervical myelopathy is available.
See related patient information handout on cervical spondylotic myelopathy, written by the author of this article.
Cervical spondylotic myelopathy is the most common cause of spinal cord dysfunction in older persons. The aging process results in degenerative changes in the cervical spine that, in advanced stages, can cause compression of the spinal cord. Symptoms often develop insidiously and are characterized by neck stiffness, arm pain, numbness in the hands, and weakness of the hands and legs. The differential diagnosis includes any condition that can result in myelopathy, such as multiple sclerosis, amyotrophic lateral sclerosis and masses (such as metastatic tumors) that press on the spinal cord. The diagnosis is confirmed by magnetic resonance imaging that shows narrowing of the spinal canal caused by osteophytes, herniated discs and ligamentum flavum hypertrophy. Choice of treatment remains controversial, surgical procedures designed to decompress the spinal cord and, in some cases, stabilize the spine are successful in many patients.
Cervical spondylotic myelopathy (CSM) is the most common spinal cord disorder in persons more than 55 years of age in North America and perhaps in the world. As the number of older persons in the United States increases, the incidence of CSM will most likely increase. In a prospective study designed to more accurately define the incidence of CSM, 23.6 percent of 585 patients with tetraparesis or paraparesis admitted to a United Kingdom regional neuroscience center had CSM.1 The overall prevalence in this population is unknown.
Pathophysiology of CSM
Spondylosis refers to the degenerative changes that occur in the spine, including degeneration of the joints, intervertebral discs, ligaments and connective tissue of the cervical vertebrae. There are three important pathophysiologic factors in the development of CSM: (1) static mechanical; (2) dynamic mechanical; and (3) spinal cord ischemia.2 Static mechanical factors result in the reduction of spinal canal diameter and spinal cord compression. With aging, the intervertebral discs dry out resulting in loss of disc height.
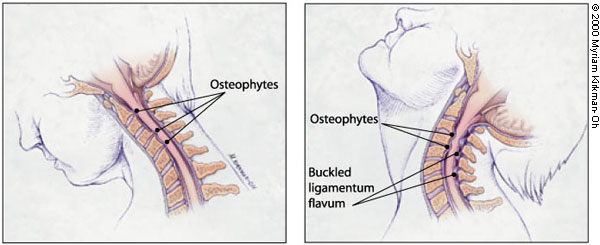
The disc also calcifies, further stabilizing the vertebrae. Osteophytes increase the weight-bearing surface of the end plates and, therefore, decrease the effective force being placed on them. In addition to osteophytic overgrowth, the ligamentum flavum may stiffen and buckle into the spinal cord dorsally. Osteophytic overgrowth ventrally and, in some cases, buckling of the ligamentum flavum dorsally can cause direct compression of the spinal cord resulting in myelopathy (clinically evident spinal cord dysfunction). Symptoms are believed to develop when the spinal cord has been reduced by at least 30 percent.4
Dynamic mechanical factors relate to the fact that the normal motion of the cervical spine may aggravate spinal cord damage precipitated by direct mechanical static compression. During flexion, the spinal cord lengthens, thus stretching over ventral osteophytic ridges. During extension, the ligamentum flavum may buckle into the spinal cord causing a reduction of available space for the spinal cord (Figure 2).
Spinal cord ischemia probably plays a role in the development of CSM, particularly in later stages.5,6 Histopathologic changes in the spinal cord consistent with ischemia have been observed in patients with CSM. However, the precise mechanism for spinal cord ischemia is not completely understood. Other factors associated with the development of spondylosis include heavy labor, posture and genetic predisposition.7,8 Also, 70 percent of patients with Down syndrome have an increased incidence of spondylosis by 50 years of age.9
Clinical History
Patients with CSM will generally have these symptoms: neck stiffness; unilateral or bilateral deep, aching neck, arm and shoulder pain; and possibly stiffness or clumsiness while walking (Table 1). CSM usually develops insidiously. In the early stages of CSM, complaints of neck stiffness are common because of the presence of advanced cervical spondylosis.10 Other common complaints include crepitus in the neck with movement; brachialgia, which is characterized as a stabbing pain in the pre- or postaxial border of the arm, elbow, wrist or fingers; a dull “achy” feeling in the arm; and numbness or tingling in the hands.
| Common symptoms |
| Clumsy or weak hands |
| Leg weakness or stiffness |
| Neck stiffness |
| Pain in shoulders or arms |
| Unsteady gait |
| Common signs |
| Atrophy of the hand musculature |
| Hyperreflexia |
| Lhermitte's sign (electric shock-like sensation down the center of the back following flexion of the neck) |
| Sensory loss |
Pain following a stereotypical dermatomal distribution is referred to as a radiculopathy rather than a myelopathy. For example, in patients with a disc herniation between the sixth and seventh vertebrae, pain radiates into the shoulder, upper arm, elbow, and index and middle fingers. It is typically unilateral. Numbness and weakness follow the same distribution. Some patients will exhibit signs and symptoms of radiculopathy and myelopathy.
The hallmark symptom of CSM is weakness or stiffness in the legs.10,11 Patients with CSM may also present with unsteadiness of gait. Weakness or clumsiness of the hands in conjunction with the legs is also characteristic of CSM. Symptoms may be asymmetric particularly in the legs. Loss of sphincter control or frank incontinence is rare; however, some patients may complain of slight hesitancy on urination.
Physical and Neurologic Examination
The physical and neurologic examination is used to confirm the presence of spinal cord dysfunction. Flexion of the neck may cause a generalized “electric shock-like” sensation down the center of the back,10 referred to as Lhermitte's sign (Table 1). Atrophy of the hands, particularly the intrinsic musculature, may be present.
Sensory abnormalities have a variable pattern on examination. Loss of vibratory sense or proprioception in the extremities (especially the feet) can occur. Superficial sensory loss may be asymmetric and persons are variably affected. The sensory examination may be confounded by the presence of diabetes mellitus and a concurrent peripheral neuropathy.
A characteristic physical finding of CSM is hyperreflexia. The biceps and supinator reflexes (C5 and C6) may be absent, with a brisk triceps reflex (C7). This pattern is almost pathognomonic of cord compression because of cervical spondylosis at the C5-C6 interspace.12 Ankle clonus and Babinski's sign (pathologic extension of the great toe elicited by stroking the foot) in the feet may also be revealed. Hoffmann's sign (a reflex contraction of the thumb and index finger after nipping the middle finger) is a subtle indicator of spinal cord dysfunction. A stiff or spastic gait is also characteristic of CSM in its later stages.
When cervical spondylosis is isolated to the C6-7, C7-T1 spinal levels, the arm reflexes may be normal. A hyperactive pectoralis muscle reflex elicited by tapping the pectoralis tendon in the deltopectoral groove causing adduction and internal rotation of the shoulder is a sign of cord compression in the upper cervical spine (C2-3, C3-4 spinal levels).13 The “dynamic” Hoffmann's sign (when a typical Hoffmann's sign is elicited after having the patient flex and extend the neck multiple times) may be an indicator of early CSM.14 Hyperreflexia may be absent in CSM patients who have concurrent diabetes, causing a peripheral neuropathy (Table 1).
Imaging and Diagnostic Studies
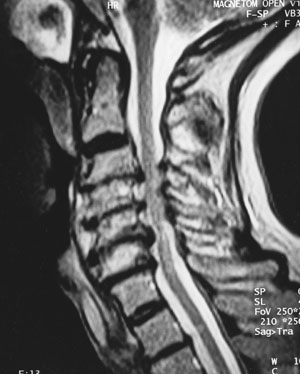
Magnetic resonance imaging (MRI) of the cervical spine is the procedure of choice during the initial screening process of patients with suspected CSM.15 MRI is noninvasive and provides images of the spine and spinal cord in several planes (Figure 3). In addition to giving an assessment of the degree of spinal canal stenosis, an MRI can identify intrinsic spinal cord lesions that can also present with myelopathy (e.g., tumors). High signal changes seen in the spinal cord of patients with CSM may indicate myelomalacia or permanent spinal cord damage.
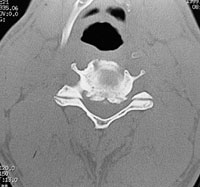
Computed tomography (CT) is complementary to MRI (Table 2). CT may give a more accurate assessment of the amount of canal compromise because it is superior to MRI in evaluating bone (osteophytes).16 Myelography or the intrathecal injection of a contrast agent is used in conjunction with CT. Since the advent of MRI, the use of myelography has decreased; however, it still provides useful information in some instances for surgical planning. Plain radiographs alone are of little use as an initial diagnostic procedure.
| Characteristic symptoms (leg stiffness, hand weakness) |
| Characteristic signs (hyperreflexia, atrophy of hands) |
| MRI or CT (showing spinal stenosis and cord compression as a result of osteophyte overgrowth, disc herniation, ligamentum hypertrophy) |
Electromyography is rarely useful in most patients with CSM; however, it may help in the exclusion of specific syndromes such as peripheral neuropathy. Somatosensory evoked potentials (SSEPs) provide a more direct assessment of spinal cord function (e.g., dorsal column function) than electromyography.17 However, SSEPs are nonspecific and therefore their use as a diagnostic tool is undetermined.
Differential Diagnosis
The presence of myelopathy on neurologic examination is not unique to CSM. Therefore, it is important to exclude other diagnoses that present in a similar fashion. In one study, it was found that 14.3 percent of patients who underwent surgery for CSM were later found to have other diagnoses.18 This finding could be an explanation for the lack of neurologic improvement after surgery in some cases.
Because cervical spondylosis is a universal finding in the elderly population, it is important to correlate cervical spondylotic changes with sensorimotor abnormalities identified on examination.10 If there is a lack of correlation, there may be a demyelinating process (e.g., multiple sclerosis). MRI is useful in this situation for identifying areas of demyelination in the spinal cord and cerebrum. In addition, a cerebrospinal fluid examination (e.g., oligoclonal bands) and visual evoked responses are important diagnostic adjuncts.
Amyotrophic lateral sclerosis (ALS) is another neurodegenerative disorder that can be confused with CSM. The absence of extremity sensory abnormalities on neurologic examination of a patient with myelopathy should alert the physician to the possibility of ALS. The presence of fasciculations on examination and a denervation pattern on electromyography serve as confirmatory evidence for ALS (Table 3).
| Feature | CSM | ALS |
|---|---|---|
| Age | Older than 55 | Older than 55 |
| MRI findings | Spondylosis | Spondylosis |
| Fasciculations | Absent | Present |
| Atrophy of arms | Present | Present |
| Atrophy of legs | Absent | Present |
| Denervation | Absent | Present |
Other conditions that can mimic CSM on presentation are primary spinal cord tumors, syringomyelia, extramedullary conditions (e.g., metastatic tumors), subacute combined degeneration of the spinal cord (vitamin B12 deficiency), hereditary spastic paraplegia, normal pressure hydrocephalus and spinal cord infarction (Table 4).10 Most of these conditions can easily be distinguished from CSM based on characteristic MRI findings.
| Amyotrophic lateral sclerosis |
| Extrinsic neoplasia (metastatic tumors) |
| Hereditary spastic paraplegia |
| Intrinsic neoplasia (tumors of spinal cord parenchyma) |
| Multiple sclerosis |
| Normal pressure hydrocephalus |
| Spinal cord infarction |
| Syringomyelia |
| Vitamin B12 deficiency |
Treatment
Evaluating the efficacy of any particular treatment strategy for CSM is difficult because reports show that as many as 18 percent of patients with CSM will improve spontaneously, 40 percent will stabilize and approximately 40 percent will deteriorate if no treatment is given.19 Unfortunately, the current understanding of CSM does not allow physicians to predict the course of a patient. Also, the literature regarding various treatment strategies (surgical and nonsurgical) for CSM is flawed because of a lack of prospective controlled studies.
NONSURGICAL TREATMENT
In patients who are mildly affected by CSM, a “careful watching” approach can be taken. A variety of nonsurgical strategies have been used with variable success for the treatment of CSM. These include cervical traction, cervical immobilization (collar or neck brace), skull traction and physical therapy. Cervical immobilization is the most commonly used treatment in the United States. Some studies demonstrate the benefits of wearing a brace, while other studies show that immobilization does not improve the patient's condition.20 It has also been reported that symptomatic patients may deteriorate neurologically during bracing, causing many to advocate earlier surgical intervention.20,21 A nonsurgical approach is usually inadvisable.
SURGICAL TREATMENT
Once frank myelopathy occurs, surgical intervention is necessary. The primary goal of surgery is to decompress the spinal cord, thus giving the neural elements more room. Traditionally, cervical laminectomy, a posterior approach, has been used for surgical treatment of CSM. However, over the past 20 years, it has been increasingly recognized that laminectomy is not appropriate for all patients. Further neurologic deterioration after laminectomy is attributed to a development of latent instability of the spine with development of kyphotic spinal deformities and to the inability of posterior approaches to directly address anterior vector compression secondary to osteophytic overgrowth.
For this reason, anterior approaches to the spine have been increasingly used.22 Through an anterior cervical approach, one can directly address and remove osteophytes and disc material for decompression of the spinal cord. Also, with the addition of interposition bone grafts and, in some cases, cervical plates (instrumentation) to promote spinal fusion, the development of instability of the neck can be prevented. A variety of factors must be considered when deciding whether to use an anterior or posterior approach, but the primary goal of both approaches is to provide adequate space for the spinal cord (Figure 4).
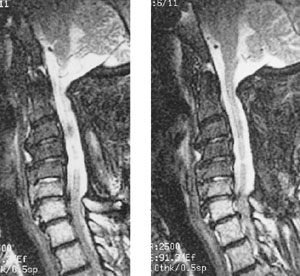
Many surgical series show “improvement,” or at least stabilization of symptoms with posterior and anterior approaches. After reviewing the surgical literature, one investigator found that the rate of successful outcome after surgery was at best 50 percent with the potential for significant postsurgical morbidity.22 The older surgical literature has been criticized because of the uncertainty of whether nonspondylotic myelopathy conditions (e.g., multiple sclerosis, ALS) had been sufficiently excluded before surgery.22
A variety of factors determine success after surgery. Factors that may portend a less than satisfactory surgical outcome include severe preoperative neurologic deficits, abnormal signal changes within the spinal cord and/or spinal cord atrophy seen on MRI, and severity of cord compression seen on radiographic studies.23–25
Final Comment
CSM is a common cause of disability in older persons. Because spondylosis is a universal finding as patients age, it is important to correlate clinical history and neurologic findings with radiographic studies. MRI is the most useful radiographic study for quantifying the degree of stenosis and excluding other pathologies. Current treatment remains controversial with regard to surgical and nonsurgical management. In the future, prospective randomized trials may be required to definitively establish treatment guidelines. Currently, surgical decompression is appropriate for many symptomatic patients.