Effective treatment of acne vulgaris can prevent emotional and physical scarring. Therapy varies according to the severity of the disease. Topical medication is generally adequate in clearing comedonal acne, while inflammatory acne usually requires the addition of oral medication. Systemic antibiotics are used most frequently and can be highly effective. Newer formulations of combined oral contraceptives are also helpful in modulating sebum production in the female patient. Severe nodulocystic acne that does not respond to topical retinoids and systemic antibiotics may be treated with isotretinoin. However, the side effect profile of this medication is extensive, and physicians should be well-versed in its potential adverse effects. (Am Fam Physician 2000;62:1823–30,1835–6.)
Acne vulgaris, or common acne, is a skin disorder of the pilosebaceous unit that generally develops in adolescence and improves in adulthood. Adolescent males, in particular, can be severely affected by this disease, which, if left untreated, can cause scarring.
Pathophysiology of Acne
At least four factors are important in the development of acne: plugging of the hair follicle with abnormally cohesive desquamated cells, sebaceous gland hyperactivity, proliferation of bacteria (especially Propionibacterium acnes) within sebum and inflammation (Figure 1).
FIGURE 1.
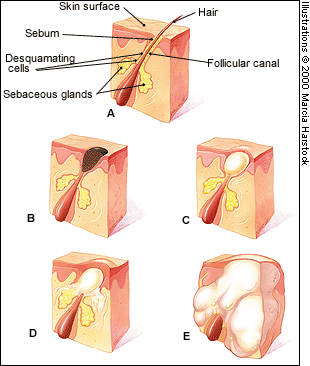
Stages of acne. (A) Normal follicle; (B) open comedo (blackhead); (C) closed comedo (whitehead); (D) papule; (E) pustule.
Earliest changes in the hair follicle occur when the follicular canal becomes blocked with abnormally keratinized desquamating cells. This plug starts above the opening of the sebaceous gland into the follicular canal and causes gradual expansion of cells and sebum within the canal. The plug becomes visible at the skin surface as a white papule (“white-head,” or closed comedo). If the opening of the follicular canal dilates, this plug protrudes from the canal and turns a dark color (“blackhead,” or open comedo).
Although sebum production increases during adolescence (particularly in boys, because of androgen stimulation), increased sebum alone does not cause acne.
Bacteria, most importantly P. acnes, are present in increased numbers in persons who have acne. Much of the inflammation that eventually occurs arises from the action of enzymes produced by the bacteria. These enzymes hydrolyze sebum into free fatty acids, which stimulate the inflammatory process. Chemotactic factors are released by this reaction, attracting neutrophils. As the follicular wall becomes inflamed, an erythematous papule appears at the skin surface. With increased sebum production, obstruction and bacterial colonization, the follicular unit ruptures, spilling its contents into the dermis. The inflow of neutrophils causes the formation of pustules. Continuation of severe inflammation leads to formation of nodules and subsequent cysts.
Classification of Acne
Acne can be classified into three categories for the purposes of treatment: comedonal, inflammatory and nodulocystic (Table 1). Comedonal acne consists predominantly of open or closed comedones with little or no accompanying inflammation (Figure 2). This type of acne typically responds to topical keratolytic agents that decrease the cohesiveness of the follicular cells. Erythematous papules and pustules characterize inflammatory acne, but comedones may also be present (Figure 3). Topical agents alone may be insufficient to treat inflammatory acne, which may benefit from systemic antibiotics. Nodulocystic acne may consist of comedones and inflammatory lesions, as well as deeper nodules and cysts (Figure 4). Although a six-month course of systemic antibiotics may be effective, nodulocystic acne frequently requires treatment with isotretinoin (Accutane). Before the initiation of isotretinoin therapy, however, patients should be evaluated for other causes of antibiotic treatment failure. Unusual causes of recalcitrant acne include drug-induced acne, tropical acne, acne conglobata and acne fulminans.
TABLE 1 Treatment Options for Acne
| Treatment | Comedonal acne | Inflammatory acne | Nodulocystic acne |
|---|---|---|---|
| Topical therapy | |||
| Salicylic acid (Keralyt) | X | ||
| Tretinoin (Retin-A) | X | X | |
| Azelaic acid (Azelex) | X | X | |
| Benzoyl peroxide | X | X | |
| Adapalene (Differin) | X | X | |
| Tazarotene (Tazorac) | X | X | |
| Antibiotics | X* | X | |
| Systemic therapy | |||
| Oral contraceptives | X | X | X |
| Erythromycin | X | X | |
| Tetracycline | X | X | |
| Doxycycline (Vibramycin) | X | X | |
| Minocycline (Minocin) | X | ||
| Isotretinoin (Accutane) | X | ||
*—May be helpful if inflammatory lesions are present as well.
FIGURE 2.
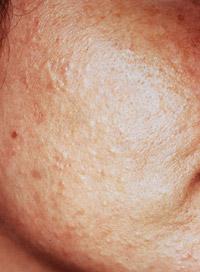
Comedonal acne. In closed comedones (whiteheads), a mass of desquamated cells plugs the follicular canal above the opening of the sebaceous gland. Sebum accumulates within the follicular canal and results in a white papule visible at the skin surface. In open comedones (blackheads), when the opening of the follicular canal dilates, the plug protrudes from the canal and turns a dark color.
FIGURE 3.
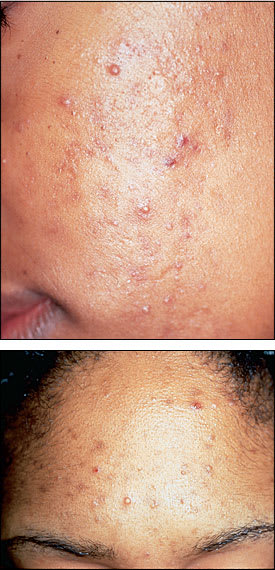
(Top and bottom) Inflammatory acne. The follicular wall ruptures, releasing sebum, cells and bacteria into the surrounding tissue, causing inflammation and redness.
FIGURE 4.
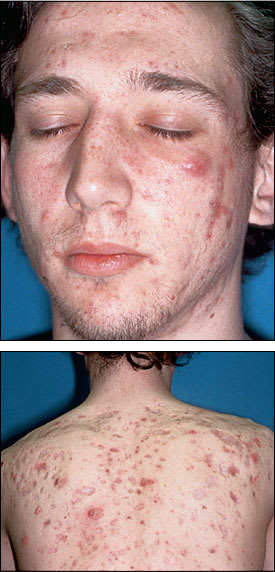
(Top and bottom) Nodulocystic acne. Extensive tissue inflammation results in the formation of nodules, cysts or abscesses.
Systemic Therapy for Acne
ORAL ANTIBIOTICS
Although topical therapy is generally adequate for comedonal acne, control of inflammatory acne usually requires systemic antibiotics. The antibiotics that have proved to be most effective include tetracycline, doxycycline (Vibramycin), minocycline (Minocin) and erythromycin. These drugs penetrate the follicle and sebaceous gland well and decrease colonization by P. acnes. They also have an anti-inflammatory effect independent of their antimicrobial properties. Although rare hypersensitivity reactions have been reported, the safety profile of these antibiotics is, in general, excellent, making routine laboratory monitoring in the asymptomatic, healthy patient unnecessary.1,2 Prescription guidelines for these antibiotics, as well as a detailed list of adverse reactions, are found in Tables 2 and 3.
TABLE 2 Prescription Guidelines for Systemic Antibiotics
| Antibiotic | Initial dosage* | Maintenance dosage | Comments |
|---|---|---|---|
| Tetracycline | 500 mg twice daily | 250 to 500 mg daily | Take on an empty stomach to avoid chelation with calcium, iron and other polyvalent cations. Take with a large glass of water to decrease dyspepsia. |
| Doxycycline (Vibramycin) | 100 mg twice daily | 50 to 100 mg daily | Take with meals. |
| Minocycline (Minocin) | 50 to 100 mg twice daily | 50 to 100 mg daily | Take with meals. |
| Erythromycin | 500 mg twice daily | 250 to 500 mg daily | Take with meals. |
*—Initial dosages should be continued until clear improvement in acne is seen, usually three to six weeks.
TABLE 3 Adverse Reactions Associated with Oral Antibiotic Use
| Adverse reaction | Tetracycline | Doxycycline (Vibramycin) | Minocycline (Minocin) | Erythromycin |
|---|---|---|---|---|
| Dyspepsia | X | X | X | X |
| Vaginal yeast infection | X | X | X | X |
| Photosensitivity | X | X | X* | X |
| Possible interference with oral contraceptives | X | X | X | |
| Tooth discoloration in children younger than 13 years or in developing fetuses | X | X | X | |
| Propionibacterium acnes antibiotic resistance | X | X | X | |
| Hyperpigmentation in scars | X | |||
| Pseudotumor cerebri | X | X | X | |
| Vestibular toxicity | X | |||
| Lupus-like reaction†2 | X | |||
| Single-organ dysfunction‡2 | X | X | X | |
| Hypersensitivity reaction§2 | X | X | X | |
| Serum sickness–like reaction∥2 | X | X | X |
*—Photosensitivity occurs less commonly with minocycline than with other tetracyclines.
†—Absence of idiopathic systemic lupus erythematosus (SLE) and the presence of antinuclear antibody and at least one clinical feature of SLE that resolves with discontinuation of drug. May occur late after initiation of therapy.
‡—Severe disease in a single organ (e.g., severe cutaneous reaction, pneumonitis, pancreatitis, hepatitis).
§—Fever, skin eruption and internal organ involvement, developing within eight weeks of initiation of therapy.
∥—Fever, skin eruption, arthralgia, ± lymphadenopathy, occurring within six weeks of initiation of treatment.
Tetracycline is extremely inexpensive but may cause adverse effects, including vaginal yeast infections and dyspepsia (rarely, esophagitis with esophageal ulcerations). Other rare adverse effects include photosensitivity and pseudotumor cerebri. It is unclear whether tetracycline interferes with oral contraceptive efficacy and, therefore, young women are often counseled to use a back-up method of contraception while taking tetracycline.3 Tetracycline may cause discoloration of forming teeth and should not be given to pregnant women or to children younger than 13 years unless all permanent teeth have erupted. Although the tetracyclines have a long track record of safety, instances of single-organ dysfunction (most commonly, severe cutaneous reaction) have been reported, as well as a few cases of hypersensitivity reaction and serum sickness–like reaction2 (Table 3). Tetracycline therapy should be avoided in patients with renal or hepatic disease.
Doxycycline is a tetracycline derivative that exhibits excellent penetration into follicles and sebaceous glands. It is better tolerated than tetracycline and may be taken with food. Of the tetracycline derivatives, it is the one most likely to cause photosensitivity. Other adverse effects are similar to those of tetracycline.
Minocycline is considered the most effective of the tetracycline derivatives, possibly because preexisting minocycline resistance is rare.4 It can be taken with food and, unlike the other tetracyclines, only infrequently causes photosensitivity. Most adverse effects of minocycline are similar to those of the other tetracyclines (Table 3). However, minocycline may cause cutaneous hyperpigmentation in scars, vertigo and, in rare instances, the development of a lupus-like syndrome. Minocycline is also associated with a higher frequency of hypersensitivity reactions than the other tetracyclines. Although hyperpigmentation is slow to fade, the other reactions usually resolve promptly with discontinuation of therapy.
Erythromycin is inexpensive but often causes dyspepsia or abdominal discomfort even when taken with meals. It is considered safe for use during pregnancy. However, P. acnes resistance to erythromycin develops more frequently (in as many as 60 percent of isolates) than with the other systemic antibiotics.
If treatment fails despite compliance with systemic antibiotic therapy, two clinical entities must be considered: antibiotic resistance and folliculitis related to overgrowth of gram-negative Enterobacteriaceae, Staphylococci or Malassezia yeasts.4 Aerobic and anaerobic cultures and sensitivity determinations should be used to decide on appropriate antibiotics. Gram-negative folliculitis is frequently treated with ampicillin, less commonly with trimethoprim-sulfamethoxazole (Bactrim, Septra) and, occasionally, with isotretinoin.5,6 The addition of topical benzoyl peroxide, a broad-spectrum antimicrobial agent, may also be beneficial in many patients who have folliculitis related to either bacteria or yeast.4
HORMONE THERAPY
Oral contraceptives may be a useful adjunctive therapy for all types of acne in women and adolescent girls. Sebum production is controlled by androgens, and oral contraceptives are known to reduce androgen levels by increasing sex hormone–binding globulin levels, thus reducing the availability of biologically active free testosterone.
The third-generation progestin norgestimate has lower intrinsic androgenicity than other currently available progestins7 and is effective in treating moderate inflammatory acne.8,9 Ortho Tri-Cyclen is a triphasic combination of norgestimate and ethinyl estradiol that has been labeled by the U.S. Food and Drug Administration (FDA) for the treatment of acne vulgaris in women and adolescent girls. Other contraceptive agents that contain norgestimate (Ortho-Cyclen) or desogestrel (Desogen) are also reasonable choices. Two to four months of therapy may be required before improvement is seen, and relapses are common if medication is discontinued.
Adult women who present with acne in conjunction with hirsutism, alopecia or menstrual disturbances should be examined for the possibility of ovarian or adrenal hyperandrogenism.10,11 Basic screening tests include measurement of free testosterone and dehydroepiandrosterone sulfate (DHEA-S) levels and the luteinizing hormone/follicle-stimulating hormone (LH/FSH) ratio. An elevated serum free testosterone level indicates a hyperandrogenous state of adrenal or ovarian origin; an increased DHEA-S level suggests adrenal hyperandrogenism and an LH/FSH ratio of greater than 3 suggests polycystic ovary syndrome. Consultation with an endocrinologist may be helpful in treating these conditions. Recalcitrant acne may respond to treatment with isotretinoin, low androgenic progestin oral contraceptives or other antiandrogens, such as spironolactone (Aldactone).11–13
Although not labeled by the FDA for treatment of acne, spironolactone, at dosages of 100 to 200 mg daily, may be effective in treating acne vulgaris in most women and adolescent girls; frequent adverse effects include menstrual irregularities, breast tenderness and fatigue.14 Hyperkalemia is rarely a problem in adolescents taking spironolactone for acne.14
ISOTRETINOIN
Nodulocystic acne, if left untreated, may cause physical and emotional scarring. This form of acne is unlikely to respond to topical therapy. Initially, patients should be prescribed an oral antibiotic. If the acne fails to respond after six months of conventional therapy, treatment with isotretinoin should be considered.6
Isotretinoin is an oral retinoid preparation that decreases the size and secretion of the sebaceous glands, normalizes follicular keratinization, inhibits P. acnes growth and exerts an anti-inflammatory effect.15 Isotretinoin is labeled for use in patients with nodulocystic acne and can markedly improve this condition in most persons. There is a growing international consensus that, although the primary indication for isotretinoin is nodulocystic acne, patients who have an inadequate response to appropriate conventional therapy for less severe acne may also benefit from this drug.16 Patients who have scarring inflammatory acne and those with acne that causes severe psychologic distress may also be candidates.6,16 However, isotretinoin is FDA–labeled only for treatment of severe recalcitrant nodular acne.17
The typical dosage of isotretinoin is 0.5 to 1 mg per kg daily in two divided doses, with a standard cumulative maximum of 120 to 150 mg per kg per treatment course.6,15,16,18,19 Because the intensity of the side effects of isotretinoin is dose-related, the lower dosage may be chosen. However, dosages of 0.5 mg per kg daily or less are more frequently associated with treatment failure.16,20 Initiation of isotretinoin therapy may cause a marked flare-up of the patient's acne. It is, therefore, common practice to introduce the medication slowly, beginning at 0.1 to 0.5 mg per kg daily, and increasing to the desired dosage of 1 mg per kg daily by the end of the first month of treatment.6,15 The average duration of therapy is five months, at which time most patients will have reached the desired goal of 120 to 150 mg per kg. However, a longer course of therapy is necessary in patients taking lower initial or daily dosages.
Adjuvant therapy with other agents may be considered during isotretinoin treatment. Topical antibiotics may be beneficial, but use of topical keratolytics and drying agents should be discontinued because concomitant use may lead to extensive dryness. Occasionally, oral erythromycin or prednisone is used at the beginning of isotretinoin therapy to control the initial acne flare-up. None of the tetracyclines should be used for this purpose because the combination of a tetracycline and isotretinoin increases the likelihood of pseudotumor cerebri development.20
Pustules generally clear more rapidly than papules or nodules. Lesions on the face, upper arms and legs tend to respond more quickly than those on the trunk. After reaching the goal dosage of 120 to 150 mg per kg, isotretinoin therapy should be discontinued even if the acne is not completely clear, because improvement continues for one to two months following cessation of treatment.
In a study of patients who were observed for 10 years, a single course of isotretinoin therapy completely cleared acne in more than 60 percent.16 If relapse occurred, it usually developed within the first three years after isotretinoin therapy; 78 percent of relapses were reported to occur within the first 18 months.16 Of the 39 percent of patients who experienced a relapse, 16 percent required reinitiation of oral antibiotics, and 23 percent required additional isotretinoin therapy.16 In those who relapse, repeated full courses of isotretinoin may be required. Of the 23 percent of patients who required repeated courses in one study, 17 percent had two courses, 5 percent had three courses and 1 percent had four to five courses, with predictably successful results and without additional adverse reactions.21
The side effect profile of isotretinoin is extensive, and physicians prescribing this medication should be well-versed in its potentially dangerous consequences. Patients should be evaluated every four weeks for adverse effects and to ensure compliance with therapy.18 A reduction in dosage or cessation of therapy usually causes fairly rapid resolution of clinical and laboratory side effects.
Mucocutaneous reactions are the most common adverse effects.19 Drying of the mucosal surfaces occurs in nearly all users, with cheilitis being the most common finding. Frequent application of moisturizing agents is necessary. Many patients who wear contact lenses are forced to switch to eyeglasses during the course of therapy because of conjunctival dryness. Reddening of the skin and increased photosensitivity vary among different populations.19
More serious adverse effects are rare, and most involve the musculoskeletal system.19 Arthralgias and muscle stiffness occur more frequently in those who participate in vigorous exercise. Long-term retinoid therapy may be complicated by skeletal changes, including osteoporosis and osteophyte formation. However, no studies have reported notable bony changes associated with short-term isotretinoin therapy for the treatment of acne. Mild to moderate headaches are fairly common; if the headache is severe or associated with visual changes, the patient should be evaluated for the presence of pseudotumor cerebri, a rare consequence of therapy. Patients may complain of fatigue or mild mood alterations and, rarely, isotretinoin may precipitate a depression. There have been reports of patients committing suicide while taking isotretinoin, and patients at risk should be monitored carefully.22
Laboratory monitoring is required to assess adverse metabolic events (Table 4). Disturbances in lipid metabolism resulting in hypertriglyceridemia and hypercholesterolemia occur in as many as 25 percent of patients. Liver enzyme levels should also be monitored periodically, although elevations beyond the reference range are rare.19 Although opinions vary, most physicians routinely measure liver enzyme and lipid levels at the start of therapy and again two to four weeks later.18,19 If no abnormal results are found at this point, some recommend discontinuing periodic testing,19 while others continue to test every four to eight weeks.18 If elevations occur, reducing the dosage or, in rare instances, discontinuing isotretinoin therapy should be considered.6
TABLE 4 Recommended Laboratory Monitoring for Patients Using Isotretinoin
| Test | Frequency | Comments |
|---|---|---|
| Qualitative hCG (urine pregnancy tests are adequate if they meet the required sensitivity of 50 IU per L)* | One week before starting treatment, then monthly* | Two forms of birth control (e.g., oral contraceptive plus barrier method) required; isotretinoin treatment should begin on the second or third day after the onset of a normal menstrual period; pregnancy should not be attempted until one month after discontinuation of therapy.* |
| Monthly pregnancy tests may be replaced by detailed questioning about contraceptive use, menses, etc., at monthly visits | ||
| Lipid levels* | Before starting treatment, then at weekly or biweekly intervals until the lipid response to isotretinoin is established, usually by four weeks* | Measure fasting triglyceride levels; if level rises to 350 mg per dL (3.95 mmol per L), repeat in two weeks. Isotretinoin therapy should be discontinued if levels exceed 700 mg per dL (7.9 mmol per L). |
| Liver function tests* | Before starting treatment, then at weekly or biweekly intervals until weekly or biweekly isotretinoin is established* | If elevations appear, reduce the dosage by 50 percent or interrupt treatment. |
hCG = human chorionic gonadotropin.
*—Official U.S. Food and Drug Administration labeling requirements.
The most devastating adverse effect of isotretinoin is teratogenicity. Major malformations may occur in 25 to 30 percent of fetuses exposed to isotretinoin.23 The most common developmental defects involve craniofacial, cardiac, thymic and central nervous system structures.23 Despite a pregnancy prevention program implemented by the manufacturer, pregnancy occurs in 3.4 per 1,000 courses of treatment with isotretinoin.24 Physicians are encouraged to use the “Patient Information and Consent to Treatment” form, which is available from the manufacturer, when counseling patients. Before therapy begins, the patient must be proved not to be pregnant, and two concurrent, effective forms of birth control should be used for one month before beginning isotretinoin therapy, throughout the entire course of treatment and for one month after cessation of therapy.25

