Although beta-adrenergic blockers can significantly reduce mortality after a myocardial infarction, these agents are prescribed to only a minority of patients. Underutilization of beta blockers may be attributed, in part, to fear of adverse effects, especially in the elderly and in patients with concomitant disorders such as diabetes or heart failure. However, studies have shown that such patients are precisely the ones who derive the greatest benefit from beta blockade. Advancing age or the presence of potentially complicating disease states is usually not a justification for withholding beta-blocker therapy. With use of cardioselective agents and through careful dosing and monitoring, the benefits of beta blockers after myocardial infarction far outweigh the potential risks in most patients.
Although the benefits of beta-adrenergic blockers after acute myocardial infarction have been apparent since the early 1980s, there has been, in recent years, a tremendous resurgence of interest in the use of beta blockers in the short- and long-term management of myocardial infarction. Despite the benefits of these agents, data from health services research studies show that beta blockers are often not used in patients who have had a myocardial infarction.1–3 In addition, emerging data on the safety and benefits of beta blockers in patients with congestive heart failure have led to reconsideration of the use of beta blockers under circumstances in which these drugs were previously considered contraindicated.
The American Heart Association and the American College of Cardiology emphasize the importance of beta blockade in their current treatment guidelines for myocardial infarction.4 On the basis of these guidelines, the Health Care Financing Administration (HCFA) and the National Coalition for Quality Assurance (NCQA) have identified beta-blocker therapy after myocardial infarction as a critical marker for quality of care. HCFA and NCQA now include beta-blocker use after infarction in profiles of hospitals and managed-care organizations.
Benefits of Beta Blockade After Infarction
Beta blockers reduce mortality during both acute and long-term management of myocardial infarction. Administration of intravenous beta blockers within 12 to 24 hours of infarction, followed by oral therapy, has been found to reduce the mortality rate approximately 13 percent within the first week of infarction.5 Studies indicate that the most marked reduction (25 percent) occurs in the first two days after infarction.5
Initiation of beta-blocker therapy within days to weeks after infarction and continuation of therapy for periods ranging from several months to three years was found in randomized trials to reduce total mortality, nonfatal myocardial infarction and sudden death by approximately 20 to 30 percent.6 Benefit occurred regardless of the patient's age or sex, infarct location and initial heart rate, or the presence or absence of ventricular arrhythmias. The greatest benefit was seen, however, in high-risk patients, including the elderly and those with large anterior infarctions, arrhythmias or left ventricular dysfunction.7
Underuse of Beta Blockers
Findings from various studies indicate considerable underuse of beta blockers following myocardial infarction, with only 20 to 50 percent of eligible patients receiving these agents.1–3,8,9 One study identified numerous factors associated with the failure to prescribe beta blockers after an infarction (Table 1).2 In this study, a twofold variation in beta-blocker use was observed in different regions of the country. Use was also much lower in patients receiving calcium channel blockers and in patients who underwent percutaneous transluminal coronary angioplasty.2
This study also revealed that general and primary care physicians were less likely to prescribe beta blockers than were internists and cardiologists.2 Different pattern of use by physician specialty was not supported, however, by another study in which no difference in beta-blocker use was found between cardiologists and generalists, including internists, and family or general practitioners.8 In both of these studies, information on the physician's primary field of practice was based on self-reported data from state licensure or Medicare records.
TABLE 1 Factors Associated with Failure to Prescribe Beta Blockers After Infarction
| Patient characteristics |
| Older age |
| Diabetes |
| Heart failure |
| Peripheral vascular disease |
| Prior myocardial infarction |
| Reduced left ventricular function |
| Renal dysfunction |
| Smoker |
| Other factors |
| Percutaneous transluminal coronary angioplasty |
| Physician specialty |
| Geographic differences |
| Use of calcium antagonists |
Information from Krumholz HM, Radford MJ, Wang Y, Chen J, Heiat A, Marciniak TA. National use and effectiveness of beta-blockers for the treatment of elderly patients after acute myocardial infarction: National Cooperative Cardiovascular Project. JAMA 1998;280:623–9 [Published erratum appears in JAMA 1999;281:37].
Lower beta-blocker use may be associated with certain characteristics that might be considered relative contraindications to beta blockade. In one study of Medicare patients with myocardial infarction, 80 percent of the patients had one or more comorbid conditions, including diabetes, congestive heart failure, chronic lung disease, conduction disorders and hypotension, which could theoretically explain the failure to prescribe beta blockers.1 Studies have shown, however, that the risk reduction from beta-blocker use in patients with such comorbid conditions is often equal to or greater than the risk reduction in patients without these conditions, in part because the baseline mortality rate is greater in high-risk patients.9 Thus, the number of patients who must be treated to prevent one adverse outcome is lower in high-risk patients, such as the elderly and patients with heart failure or diabetes, than in other patients.
Tolerability of Beta Blockers
Although a common belief is that patients may feel worse or experience a decreased quality of life when they are taking beta blockers compared with other cardiac agents, this effect is not supported by clinical studies. In the Beta Blocker Heart Attack Trial (BHAT)10 of nearly 4,000 patients, the incidence of side effects such as tiredness, sleep disturbance and impotence was not significantly different in patients taking propranolol compared with those taking placebo. In a meta-analysis of 90 comparative studies of angina, beta blockers were found to be associated with a lower incidence of adverse effects than calcium antagonists.11 A comprehensive evaluation of quality-of-life measures in hypertension trials found that beta-blocker therapy was associated with levels of well-being that were similar to those associated with calcium antagonists or angiotensin-converting enzyme inhibitors.12
Studies have suggested that some patients tolerate hydrophilic and beta1-selective blockers, such as atenolol (Tenormin) and metoprolol (Lopressor), better than lipophilic or nonselective agents, such as propranolol (Inderal). However, the data are not definitive.13
While few absolute contraindications exist for beta-blocker use after infarction, the risks are likely to outweigh the benefits in certain situations (Table 2).
TABLE 2 Absolute Contraindications to Beta-Blocker Therapy
| Asthma (active or recent, or concurrent use of bronchodilator therapy) |
| Cardiogenic shock |
| Chronic obstructive pulmonary disease (moderate or severe with reversible obstruction) |
| Decompensated heart failure (even after optimizing medical therapy) |
| Heart block (second- or third-degree block without pacemaker) |
| Severe peripheral arterial disease |
| Sick sinus syndrome |
| Symptomatic bradycardia |
| Symptomatic hypotension |
Benefits of Beta Blockers in Specific Populations
ELDERLY
Although most patients in the clinical trials of beta blockers after myocardial infarction were relatively young, substantial evidence indicates that beta blockers are as effective, if not more effective, in the elderly. Subgroup analyses from randomized trials, including patients up to 75 years of age, show a greater relative risk reduction in elderly patients than in young patients (Figure 1).14 Pooled data from these trials demonstrate that beta-blocker therapy is associated with a 40 percent reduction in mortality in older patients (more than 60 years of age).14 In contrast, there is a 28 percent reduction in mortality rate among younger patients.14 Subsequent observational studies of Medicare patients demonstrate a 14 to 43 percent reduction in mortality in patients 65 years or older who are treated with beta blockers.2,3 These studies show that beta blockers can be safely used in the elderly if therapy is initiated at a low dosage and titrated slowly and if patients are monitored for potential complications.
FIGURE 1. Beta-Blocker Therapy and Age
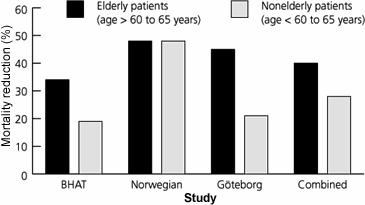
Percentage of mortality reduction in elderly and younger patients who received long-term beta-blocker therapy after myocardial infarction. (BHAT = Beta Blocker Heart Attack Trial; Norwegian = Norwegian Multicenter Timolol Study; Göteborg = Göteborg Metoprolol Trial)
Information from Forman DE, Bernal JL, Wei JY. Management of acute myocardial infarction in the very elderly. Am J Med 1992;93:315–26.
DIABETES
Diabetes is associated with a high risk of early and late mortality following myocardial infarction.15 Clinical trials of beta blocker use after infarction have demonstrated mortality reductions that are 10 to 30 percent greater in diabetic patients than in nondiabetic patients (Figure 2).15 In addition, the reduction in the incidence of nonfatal infarctions and sudden death has been found to be greater in patients with diabetes than in patients without diabetes.16,17
FIGURE 2. Beta-Blocker Therapy and Diabetes
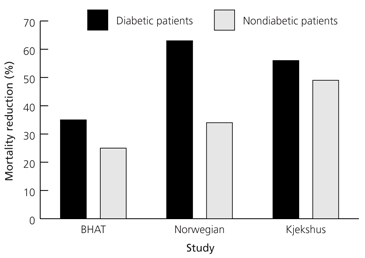
Percentage of mortality reduction in patients with and without diabetes who received long-term beta-blocker therapy after myocardial infarction. (BHAT = Beta Blocker Heart Attack Trial; Norwegian = Norwegian Multicenter Timolol Study; Kjekshus = study by Kjekshus J, et al.15)
Information from Kjekshus J, Gilpin E, Cali G, Blackey AR, Henning H, Ross J Jr. Diabetic patients and beta-blockers after acute myocardial infarction. Eur Heart J 1990;11:43–50.
These benefits, however, are not entirely without risk in patients with diabetes. Beta blockers can impair glucose tolerance, block the symptoms of hypoglycemia and delay recovery from a hypoglycemic episode.18 In most patients with type 2 diabetes (formerly known as non–insulin-dependent diabetes), however, hypoglycemia is relatively uncommon. In most patients with diabetes, the benefits of beta blockers outweigh the risks, and a trial of beta-blocker therapy is warranted. When patients with diabetes are given beta blockers, they should be educated about monitoring for hypoglycemia and the potential need to adjust the dosage of the hypoglycemic agents.
NON–Q-WAVE MYOCARDIAL INFARCTION
Non–Q-wave infarction is associated with a substantial risk of death, especially in the elderly. Although subgroup analysis of data from the BHAT suggests no benefit in non–Q-wave infarction, this finding has been criticized on the basis of methodologic concerns.19 Subsequently, data from the Norwegian timolol trial and observational studies suggest that beta blockers probably improve survival and prevent repeat infarction in patients with non–Q-wave infarction.20,21 Current management guidelines for myocardial infarction do not distinguish between Q-wave and non–Q-wave infarctions with respect to the recommendations for use of beta blockers.4
HEART FAILURE
Beta blockers can precipitate or exacerbate heart failure. Despite this potential, subgroup analysis of randomized controlled trials has shown that patients with heart failure derive greater benefit from beta blockers following myocardial infarction than patients without heart failure.22 In the BHAT,22 propranolol reduced mortality equally, by approximately 25 percent, in patients with and without heart failure. However, beta-blocker therapy was associated with a 47 percent reduction in sudden death in patients with heart failure, compared with a 13 percent reduction in patients without heart failure.
Recent studies have shown that beta blockers are often beneficial in the treatment of chronic heart failure. Two meta-analyses of randomized trials reported a reduction in mortality of approximately 30 percent in patients with heart failure who received beta blockers.23,24 Carvedilol (Coreg) was labeled by the U.S. Food and Drug Administration for the treatment of mild to moderate heart failure after publication of a large randomized trial that demonstrated a 65 percent mortality reduction in patients who received this agent.25 Subsequently, studies of the beta blockers bisoprolol26 and metoprolol27 in the treatment of heart failure were terminated early when reductions of 34 percent in all-cause mortality and 40 to 45 percent in sudden death were demonstrated. It should be kept in mind that beta blockers were added in these studies after treatment had been optimized with diuretics, angiotensin-converting enzyme (ACE) inhibitors and, in some cases, digoxin. Furthermore, despite promising preliminary data, the efficacy and safety of beta blockers in patients with New York Heart Association (NYHA) class IV heart failure remains uncertain.28
Clinical evidence clearly shows that beta blockers are beneficial after myocardial infarction in most patients, including those with reduced left ventricular function or heart failure. In patients with heart failure, beta-blocker therapy should be started at the lowest dosage and slowly titrated upward. A transient worsening of the symptoms of heart failure may occur before significant improvement is seen. Beta blockers should be withheld in patients with severe or decompensated heart failure, until the patient's condition is stabilized with diuretic and ACE inhibitor therapy.
LIPID DISORDERS
Beta blockers, particularly nonselective agents, have been shown to increase the serum triglyceride level and lower the high-density lipoprotein (HDL) cholesterol level.29 Other pharmacologic effects of beta blockers, however, may actually impede the development of atherosclerosis and thrombogenesis.29 The overall clinical importance of the lipid-modifying effects of beta blockers remains unclear. Lipid abnormalities should be treated but are not a justification for withholding beta-blocker therapy after myocardial infarction.
CHRONIC PULMONARY DISEASE
Although beta blockers can produce life-threatening complications in patients with asthma, some patients taking beta2 agonists can tolerate up to 100 mg daily of the beta1 blocker metoprolol.30 The use of beta blockers in patients with nonreversible chronic obstructive pulmonary disease (COPD) is less clear. While some COPD patients can tolerate beta blockers, others, particularly those who require beta2 agonists, may have significant deterioration in pulmonary function.31 Findings from a recent observational trial9 suggest that beta blockers can be used safely in patients with COPD. In another study,32 esmolol did not adversely affect pulmonary function in patients with nonreversible obstruction.
Although these results are encouraging, further study is needed in this area. In the meantime, it is wise to avoid the use of beta blockers in patients with asthma or COPD with a reversible component and in patients requiring bronchodilator therapy. In patients with mild, fixed obstruction, beta-blocker use may be a consideration, provided there is close monitoring of symptoms and pulmonary function tests.
PERIPHERAL VASCULAR DISEASE
Peripheral vascular disease has generally been considered a contraindication to the use of beta blockers. Worsening of the symptoms of peripheral vascular disease or intermittent claudication has been noted in small uncontrolled reports of beta-blocker use in patients with peripheral vascular disease.33,34 Nonselective agents that produce beta2 blockade have been associated with the greatest risk. Conversely, a meta-analysis of 11 randomized, controlled trials found that beta blockers did not adversely affect symptoms, walking distance or blood flow in the calf muscles of patients with mild to moderate peripheral arterial disease.35
Therefore, a trial of beta-blocker therapy can be initiated in patients with mild to moderate peripheral vascular disease. Beta1 selective agents should be used, and patients should be monitored closely for worsening of symptoms. Because patients with Raynaud's phenomenon or severe arterial vascular disease were excluded from the meta-analysis, beta blockers cannot be recommended for use in these patients.
Practical Considerations
Dosage recommendations for beta blockers after acute myocardial infarction are summarized in Table 3. Although many physicians are concerned about the potential for beta blockers to precipitate heart failure, reduce exercise tolerance or otherwise impair quality of life, these risks can be minimized by using cardio-selective agents, initiating therapy at a low dosage and slowly increasing the dosage based on the patient's tolerance of the agent. Many adverse effects, such as fatigue or even worsening of left ventricular function, may be transient and resolve with continued therapy.
TABLE 3 Dosages of Beta Blockers for Short-Term and Long-Term Treatment After Myocardial Infarction
| Agent | Dosage | Cost* |
|---|---|---|
| Acute treatment | ||
| Atenolol (Tenormin) | Two 5-mg IV doses 10 minutes apart, then 50 mg orally every 12 hours, beginning 10 minutes after the second IV dose | NA |
| Metoprolol (Lopressor) | Three 5-mg IV doses 5 minutes apart, then 50 mg orally every 6 hours, beginning 15 minutes after the third IV dose | NA |
| Long-term therapy (beta1-selective agents preferred)† | ||
| Atenolol | 100 mg once daily or 50 mg twice daily | $30 to 42 |
| Metoprolol | 100 mg twice daily | 41 |
| Alternative nonselective agents | ||
| Propranolol (Inderal) | 60 mg three or four times daily | 17 to 22 |
| Timolol (Blocadren) | 20 mg twice daily | 21 |
IV = intravenous; NA = not applicable.
*—Estimated cost to the pharmacist of long-term therapy for one month based on average wholesale prices (rounded to the nearest dollar in Red Book. Montvale, N.J.: Medical Economics Data, 1999. Cost to the patient may be greater, depending on prescription filling fee.)
†—Initiate beta-blocker therapy at lower dosages and titrate slowly in elderly patients and in patients with heart failure.
Adverse effects on quality of life have been reported more frequently with nonselective beta blockers such as propranolol than with selective agents. Beta1-selective blockers such as metoprolol or atenolol may produce fewer side effects and are generally preferred for use in patients with COPD, diabetes, lipid abnormalities or peripheral vascular disease. Beta blockers with intrinsic sympathomimetic activity, such as pindolol (Visken) and acebutolol (Sectral), should not be used for cardioprotection after infarction. Agents with peripheral alpha-blocking activity, such as labetalol (Trandate) and carvedilol, have not yet been evaluated and labeled for use following myocardial infarction.
In addition to appropriate dosing and monitoring, patient education is essential to maximize efficacy and minimize adverse effects. Along with verbal counseling, giving the patient written information about the importance of beta-blocker therapy after a myocardial infarction may be useful in enhancing compliance and preventing complications. Physicians, pharmacists and nurses can all play an important role in educating and monitoring patients.

