Infectious diseases account for one third of all deaths in people 65 years and older. Early detection is more difficult in the elderly because the typical signs and symptoms, such as fever and leukocytosis, are frequently absent. A change in mental status or decline in function may be the only presenting problem in an older patient with an infection. An estimated 90 percent of deaths resulting from pneumonia occur in people 65 years and older. Mortality resulting from influenza also occurs primarily in the elderly. Urinary tract infections are the most common cause of bacteremia in older adults. Asymptomatic bacteriuria occurs frequently in the elderly; however, antibiotic treatment does not appear to be efficacious. The recent rise of antibiotic-resistant bacteria (e.g., methicillin-resistant Staphylococcus aureus and vancomycin-resistant enterococcus) is a particular problem in the elderly because they are exposed to infections at higher rates in hospital and institutional settings. Treatment of colonization and active infection is problematic; strict adherence to hygiene practices is necessary to prevent the spread of resistant organisms.
Despite advances in antibiotic therapy, infectious diseases continue to be a major cause of mortality in older adults. The diagnostic and therapeutic nuances of managing infections in older adults create special challenges for physicians. Between 1980 and 1992, the infectious disease mortality rate in patients 65 years and older actually rose 25 percent.1 That comparative mortality rate is nine times the rate in patients between 25 and 44 years of age.1.
Many factors contribute to increased mortality in the elderly, including comorbid conditions, an increase in the number of invasive procedures performed and decreased physiologic reserves.
Assessment
The diagnosis of infection in older adults is more challenging, yet early diagnosis and treatment in these patients are imperative because of the higher incidence of morbidity and mortality. Many signs and symptoms of infection that are common in younger adults, particularly fever and leukocytosis, present less frequently or not at all in older adults.2,3 While 60 percent of older adults with serious infections develop leukocytosis, its absence does not rule out an infectious process.4 Because frail older adults tend to have poorer body temperature response, elevations in body temperature of 1.1°C (2°F) from their normal baseline temperature should be considered a febrile response. Fevers higher than 38.3°C (101°F) often indicate severe, life-threatening infections in older adults, and hospitalization should be considered for these patients.3
Changes caused by infection in the elderly are subtle, and nonspecific complaints may be the only indications. Elderly patients with infections commonly present with cognitive impairment or a change in mental status; frank delirium occurs in 50 percent of older adults with infections.5 Furthermore, anorexia, functional decline, falls, weight loss or a slight increase in respiratory rate may be the only signs indicating infection in older patients. A complete assessment is essential to identify older adults at higher risk of infection (Table 1).5
Bacterial Pneumonia
Pneumonia and influenza combined are the sixth leading cause of death in the United States, and about 90 percent of these deaths occur in adults 65 years and older.6 In fact, more than 60 percent of people 65 years and older are admitted to hospitals because of pneumonia.7 Changes in pulmonary reserve, decreased mucociliary transport, decreased cough reflex, decreased elasticity of alveoli and poorer ventilation—all of which lead to diminished cough and airway patency—cause older adults to be more susceptible to pneumonia. Table 28,9 describes risk factors that are associated with increased mortality in pneumonia acquired in community settings.
TABLE 2 Findings Associated with Poor Prognosis in Elderly Patients with Community-Acquired Pneumonia
| Age >50 years | Temperature <35°C (95°F) or ≥40°C (104°F) |
| Pa O2 <60 mm Hg | WBC elevation >13,000 per mm3 (13 × 109 per L) |
| O2 saturation <90 percent | WBC suppression <4,000 per mm3 (4 × 109 per L) |
| Altered mental status | Hematocrit <30 percent (0.3) |
| Nursing home resident | BUN ≥30 mg per dL (11 mmol per L) |
| Comorbid illness* | Glucose ≥250 mg per dL (13.9 mmol per L) |
| Tachycardia ≥125 beats per minute | Sodium <130 mEq per L (130 mmol per L) |
| Tachypnea ≥30 breaths per minute | Radiographic evidence of progressing or multilobar infiltrates |
| Hypotension <90 mm Hg systolic | Pleural effusion |
PaO2 = partial pressure of arterial oxygen; O2 = oxygen; WBC = white blood cell count; BUN = blood urea nitrogen.
*—Comorbid illness includes neoplastic disease, renal failure, liver failure, congestive heart failure or cerebrovascular disease.
Information from Fine MJ, Smith DN, Singer DE. Hospitalization decision in patients with community-acquired pneumonia: a prospective cohort study. Am J Med 1990;89:713–21, and Fine MJ, Auble TE, Yealy DM, Hanusa BH, Weissfeld LA, Singer DE, et al. A prediction rule to identify low-risk patients with community-acquired pneumonia. N Engl J Med 1997;336:243–50.
Attention to these risk factors may help the physician assess the severity of illness at presentation and determine the need for hospitalization.10 Because the diagnosis of pneumonia in older adults is difficult to make since the signs and symptoms can be subtle, the initiation of antibiotic therapy is often delayed, which may contribute to higher mortality rates.11
DIAGNOSIS
Routine laboratory testing is considered to have little value in determining the etiology of pneumonia. However, it may have prognostic significance in patients 60 years and older and, therefore, is recommended for them.12 The value of a Gram stain and routine bacterial cultures of sputum from patients with pneumonia is debated, and obtaining adequate sputum specimens in frail, older adults may be particularly difficult.12 Quantification of cultured pathogens and correlation with the Gram stain may help the physician interpret possible oropharyngeal contaminants versus true pneumopathogens.13 Blood cultures are recommended in hospitalized patients.
TREATMENT
Because of increased mortality and the larger number of potential pathogens causing pneumonia in older adults, an early etiologic diagnosis is optimal to guide pharmacotherapy. Regardless of age, bacterial causes of pneumonia can only be identified in 20 to 50 percent of patients.12,14 In the absence of specific bacterial etiology, pharmacotherapy of pneumonia is initially empiric and directed at the likely causative pathogens.
In older adults, Streptococcus pneumoniae is still the most common cause of pneumonia, followed by respiratory viruses, Haemophilus influenzae, gram-negative bacilli and Staphylococcus aureus.12 Moraxella catarrhalis, Legionella and Mycoplasma are less common but important causes of pneumonia in the elderly because these bacterium may not be covered by traditional empiric antibiotic regimens. As a group, gram-negative bacteria are responsible for more infections in the elderly than in younger adults.2,12 Therefore, in older adults, empiric antibiotic therapy should provide coverage for gram-positive and gram-negative bacteria.2 Table 313,15 summarizes the options for initial antibiotic therapy for pneumonia.
TABLE 3 Empiric Antibiotic Selection in Elderly Patients with Community-Acquired Pneumonia
| Outpatients | |||
| Generally preferred: doxycycline (Vibramycin), a macrolide* or a fluoroquinolone† | |||
| Modifying factors: | |||
| Suspected penicillin-resistant Streptococcus pneumoniae: fluoroquinolone† | |||
| Suspected aspiration: amoxicillin/clavulanate potassium (Augmentin) | |||
| Elderly, debilitated patients: some authorities prefer a fluoroquinolone as first choice | |||
| Hospitalized patients | |||
| General medical ward | |||
| Generally preferred: an extended-spectrum cephalosporin with a macrolide,* or a beta-lactam/beta-lactamase inhibitor combined with a macrolide, or a fluoroquinolone alone | |||
| Hospitalized in the intensive care unit with serious pneumonia | |||
| Generally preferred: erythromycin, azithromycin (Zithromax) or a fluoroquinolone† plus cefotaxime (Claforan), ceftriaxone (Rocephin) or a beta-lactam/beta-lactamase inhibitor‡ | |||
| Modifying factors | |||
| Structural diseases of the lung: antipseudomonal penicillin, a carbapenem or cefepime (Maxipime) plus a fluoroquinolone† | |||
| Penicillin allergy: a fluoroquinolone†, with or without clindamycin (Cleocin) | |||
| Suspected aspiration: a fluoroquinolone§, with or without clindamycin, metronidazole (Flagyl) or a beta-lactam/beta-lactamase inhibitor§ alone | |||
*—Azithromycin, clarithromycin (Biaxin) or erythromycin.
†—Levofloxacin (Levaquin), sparfloxacin (Zagam), grepafloxacin (Raxar), gatiflux (Tequin) or another fluoroquinolone with enhanced activity against S. pneumoniae.
‡—Ampicillin/sulbactam (Unasyn) or ticarcillin/clavulanate-potassium (Timentin) or piperacillin (Pipracil)/tazobactam (Zosyn); for structural disease of the lung: ticarcillin/clavulanate or piperacillin.
§—Cefotaxime, ceftriaxone or a beta-lactam/beta-lactamase inhibitor.
Adapted with permission from Bartlett JG, Breiman RF, Mandell LA, File TM. Community-acquired pneumonia in adults: guidelines for management. Infectious Diseases Society of America. Clin Infect Dis 1998;26:811–38, and Bartlett JG, Dowell SF, Mandell LA, File TM Jr, Musher DM, Fine AM. Practice guidelines for management of community-acquired pneumonia in adults. Clin Infect Dis 2000;31:347–82.
Because of their high cost, the newer macrolides should be reserved for use in patients who cannot tolerate erythromycin, when infection with H. influenzae is more likely (i.e., smokers) or when a drug interaction is anticipated.16 Trimethoprim-sulfamethoxazole (Bactrim, Septra) has a good spectrum of activity that includes S. pneumoniae, H. influenzae and M. catarrhalis; unfortunately, it does not provide activity against the atypical pathogens, and emerging resistance to this drug may limit its use. The newer fluoro-quinolones may also be suitable in the empiric treatment of pneumonia because most of them have a broad spectrum of activity against the likely pathogens for bacterial pneumonia, including atypical organisms. However, indiscriminate use of these antibiotic agents may increase the development of resistance.
In addition to the choice of antibiotic, physicians need to decide on the route of administration and duration of treatment. Unfortunately, little information exists regarding administration and duration of treatment. Frequently, no clear advantage is evident for the use of intravenous over oral antibiotics as long as the bioavailability and tissue penetration of the drug is adequate. Common practice has been to begin empiric therapy with an intravenous agent in hospitalized patients.13 A review of the literature suggests that a change to oral therapy can be made if the patient is hemodynamically stable, clinically improving and absorbing oral medication.13 Adequate hydration and oxygenation must also be assured for successful oral treatment. Generally, patients with S. pneumoniae should be treated for seven to 10 days; patients with mycoplasma or chlamydia infection, and those who are immunocompromised should be treated with longer regimens.12
Influenza
Influenza is a common respiratory infection that has an enormous impact worldwide and causes significant morbidity and mortality in older adults. Influenza is responsible for more than $1 billion in annual Medicare expenditures. Of deaths resulting from influenza, 80 to 90 percent occur in adults 65 years and older.17 Older adults are prone to severe and potentially fatal complications from this common illness because of co-existing chronic disease and weakened immunity. Older adults can benefit most from vaccination, early detection and aggressive therapy.
CLINICAL MANIFESTATIONS
The signs and symptoms of influenza infection in older adults are similar to those occurring in younger patients although, again, a febrile response may be absent. Influenza is typically associated with rapid onset of headache, fever, chills, muscle aches, malaise, cough and sore throat. Most people recover fully within one week, but older adults may develop a persistent weakness that can last for many weeks and are also at higher risk for developing complications, such as pneumonia.
DIAGNOSIS
Several commercially produced rapid-diagnostic tests intended for use in outpatient settings can detect influenza viruses within 30 minutes.17 In selected patients, obtaining a viral culture may be warranted to acquire specific information on influenza subtypes and strains.
TREATMENT
Four antiviral agents—amantadine (Symmetrel), rimantadine (Flumadine), zanamivir (Relenza) and oseltamivir (Tamiflu)—are approved for prevention or treatment of influenza (Table 4).18 These agents must be taken within 48 hours of the onset of illness, which is difficult because most patients treat their illness at home for several days before seeking treatment from a physician.19 Zanamivir and oseltamivir are not approved for prophylactic use.18 It is unknown whether therapy with amantadine or rimantadine can prevent complications of influenza A among persons at high risk, including older adults.20 Rimantadine costs more than amantadine but has fewer adverse effects on the central nervous system (i.e., confusion, nervousness, anxiety) and is less dependent on renal excretion.17,18 Table 517 lists indications for use and duration of therapy for these agents, and recommended dosage adjustments when renal impairment is present.
TABLE 4 Comparison of Antiviral Agents for Treatment of Influenza
| Agent | Types of influenza viruses inhibited | Route of administration | Dosage | Cost* (daily) | Cost* (five-day regimen) |
|---|---|---|---|---|---|
| Amantadine (Symmetrel) | A | Oral (tablet, capsule, syrup) | 200 mg as a single dose or 100 mg twice daily† | $ 1.96 | $ 9.82 |
| Rimantadine (Flumadine) | A | Oral (tablet, syrup) | 100 mg twice daily† | 3.78 | 18.90 |
| Zanamivir (Relenza) | A and B | Oral inhalation‡ | 2 inhalations (one 5-mg blister per inhalation for a total dose of 10 mg) twice daily | 8.88 | 44.00 |
| Oseltamivir (Tamiflu) | A and B | Oral (capsule) | 75 mg twice daily§ | $10.60 | $53.00 |
*—Estimated cost to the pharmacist based on average wholesale prices in Red book. Montvale, N.J.: Medical Economics Data, 2000. Cost to the patient will be higher, depending on prescription filling fee.
†—In elderly nursing home patients, a dose reduction to 100 mg daily is recommended.
‡—Zanamivir is administered by using a specially designed plastic oral inhalation device called a Diskhaler. The device and instructions for its use are included in the package with the medication.
§—A dosage of 75 mg once daily is recommended for patients with creatinine clearance less than 30 mL per minute (0.5 mL per second).
Adapted from Winquist AG, Fukuda K, Bridges CB, Cox NJ. Neuraminidase inhibitors for treatment of influenza A and B infections. MMWR Morb Mortal Wkly Rep 1999;48(RR-14):1–9 [Published erratum appears in MMWR Morb Mortal Wkly Rep 1999;48:1139].
TABLE 5 Influenza Treatment with Amantadine (Symmetrel) or Rimantadine (Flumadine)
| Adults ≥65 years with normal renal function | ||
| Active (documented) influenza A infection: 100 mg daily for three to five days | ||
| Prophylaxis | ||
| Unvaccinated patient exposed to suspected influenza illness: 100 mg daily for at least two weeks or up to one week following end of exposure | ||
| Newly vaccinated patient exposed to suspected influenza illness: 100 mg daily until two weeks have elapsed since vaccination | ||
| Vaccinated residents of long-term care facilities during confirmed outbreak: 100 mg daily for at least two weeks or up to one week following end of exposure | ||
| Adults with renal impairment | ||
| Creatinine clearance | ||
| 20 to 30 mL per min (0.33 to 0.50 mL per s): 200 mg twice weekly | ||
| 10 to 20 mL per min (0.17 to 0.33 mL per s): 100 mg three times weekly | ||
| Less than 10 mL per min: 200 mg alternating with 100 mg every seven days | ||
Adapted with permission from Kuhle C, Evans JM. Prevention and treatment of influenza infections in the elderly. Clin Geriatr 1999;7(2):35.
Zanamivir and oseltamivir are equally effective neuroaminidase inhibitors, but their place in therapy is yet to be determined. Their potential benefit (decrease in duration of symptoms by one to one-and-one-half days) must be considered relative to cost, compliance and adverse effects. In some trials, these agents demonstrated effects in patients 65 years and older that are similar to effects in younger adults. It has not yet been determined whether early initiation of treatment reduces hospital admission and mortality, particularly in elderly and high-risk patients.21 The recommended dosage for zanamivir is two inhalations twice daily for five days. Oseltamivir should be taken at a dosage of 75 mg orally twice daily for five days (or once daily in patients with a creatinine clearance less than 30 mL per minute [0.5 mL per second]).
Urinary Tract Infections
Urinary tract infections (UTIs) are the most frequent bacterial infection and the most common source of bacteremia in older adults.22 Table 64 compares UTI characteristics in older and younger patients. Factors that predispose older adults to UTIs include the use of urethral or condom catheters, and neurogenic bladders with increased residual urine. Contributing factors specific to gender include prostate enlargement in men, an increase in vaginal pH, vaginal atrophy that is due to postmenopausal estrogen depletion, and incomplete emptying of the bladder in women. These factors provide the opportunity for bacterial colonization and are likely to contribute to the higher rates of asymptomatic bacteriuria and UTIs in the elderly.23
CLINICAL MANIFESTATIONS
Because classic clinical manifestations of UTIs such as dysuria, fever, urinary frequency and suprapubic tenderness may be absent or masked in older adults, treating these infections in this group is a special challenge for physicians. Vague symptoms such as nausea, vomiting and decreased urinary output must be distinguished from symptoms of other common illnesses.24 Older adults with UTIs and comorbidities (e.g., diabetes) and those who are at risk for dehydration (because of fever, nausea or vomiting) should be strongly considered for hospitalization.
DIAGNOSIS
The same process is followed for the diagnosis of UTIs in older adults as in younger adults. An important caveat is the controversy regarding treatment of asymptomatic bacteriuria, which is common in the elderly. Most authorities advocate withholding antibiotics in patients who are completely asymptomatic.23 Clinical findings and a urinalysis are usually sufficient for diagnosis in older adults; however, a urine Gram stain and culture should be considered to direct therapy.
TREATMENT
Treatment for UTIs should be directed at the organism identified by Gram stain and culture. Unfortunately, polymicrobial infections occur in about 30 percent of patients and more often if the UTI is related to the use of a catheter.24 In these patients, use of a broad-spectrum antibiotic may be necessary. In general, seven days is an adequate duration of therapy in older women and 14 days in older men. The duration of therapy is routinely doubled for infections considered to be serious.24
Treatment of asymptomatic bacteriuria does not appear to reduce morbidity or mortality and may increase the likelihood of development of drug-resistant microorganisms and adverse reactions to antibiotics. Exceptions to treatment of asymptomatic bacteriuria are patients scheduled for a genitourinary procedure who have obstructive uropathy, stones or a history of recurrent symptomatic infections.22
Skin Infections
HERPES ZOSTER
Infection with herpes zoster caused by a reactivation of varicella virus dormant in dorsal root ganglia is also common in older adults. As cellular immunity wanes with advancing age, clinical reactivation of the virus can occur.
The hallmark of herpes zoster is skin lesions that progress from discrete patches of erythema to grouped vesicles in a dermatomal pattern that pustulate and crust within seven to 10 days. Diagnosis of suspicious lesions can be confirmed by giant cells noted on Tzanck test preparation of lesion scrapings or a positive viral culture of the vesicular fluid.
Pain is the most common symptom associated with herpes zoster, and it can be debilitating in frail elderly patients. Postherpetic neuralgia develops in 10 to 70 percent of patients and can be difficult to treat.25
Antiviral therapy (famciclovir [Famvir], valacyclovir [Valtrex], acyclovir [Zovirax]) initiated within 72 hours after the appearance of the rash can reduce the acute pain (to less than one month) associated with herpes zoster and reduce the duration of the eruption.25,26 The use of antiviral agents has been shown to reduce the duration of an outbreak of herpes zoster by one to two days. The acute pain may be reduced with the addition of corticosteroid therapy if no contraindications exist.
More recent controlled trials of corticosteroids have not shown any reduction in the incidence of postherpetic neuralgia but have shown them to improve the quality of life (e.g., use of analgesics, normal sleep patterns, normal activity) when used in combination with antiviral agents in patients older than 50 years.27 An approach to the treatment and prevention of acute pain associated with herpes zoster and postherpetic neuralgia has been developed28,29 (Figure 1).27,29 Aspirin and other mild analgesics appear to have limited value.
FIGURE 1. Approaches to Treatment of Postherpetic Neuralgia
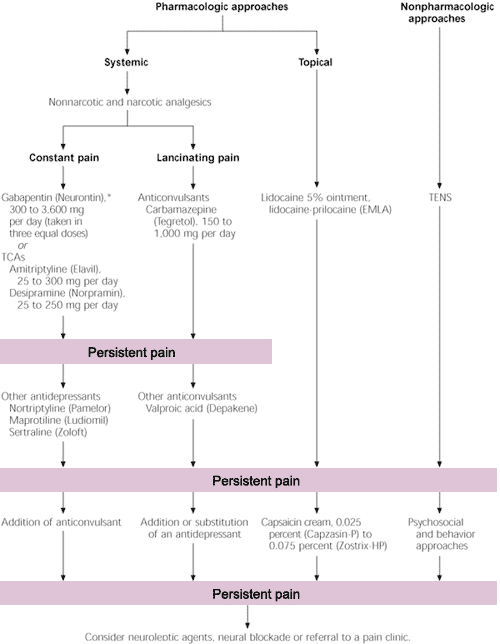
Approaches to treatment of postherpetic neuralgia. (TCAs = tricyclic antidepressants; TENS = transcutaneous electrical nerve stimulation)
Adapted with permission from Kost RG, Strauss SE. Postherpetic neuralgia—pathogenesis, treatment, and prevention. N Engl J Med 1996;335:32–42, with added information from Rowbotham M, Harden N, Stacey B, Bernstein P, Magnus-Miller L. Gabapentin for the treatment of postherpetic neuralgia: a randomized controlled trial. JAMA 1998:28:1837–42.
METHICILLIN-RESISTANT STAPHYLOCOCCUS AUREUS
Methicillin-resistant Staphylococcus aureus (MRSA) presents a major problem for elderly patients, especially those in institutional settings. People colonized with MRSA are at increased risk of MRSA infection. They also have a higher risk of death from MRSA resulting from its resistance to typical antibiotics. While MRSA infection is more likely to occur in hospitalized patients than in nursing home residents, poor functional status is associated with being an MRSA carrier. Therefore, nursing homes and other institutional settings must be especially careful to prevent the spread of infection caused by this organism.
Handwashing, isolation of infected patients and proper handling of bodily secretions are essential to prevent the spread of MRSA. The most common reservoirs for MRSA colonization are the nasal mucosa and oropharynx. Skin contamination from persons already colonized in these areas may also be a source for MRSA infection.
While colonization by MRSA does not require systemic treatment, active infection with MRSA is treated with vancomycin (Vancocin) as the preferred antibiotic. Vancomycin is administered intravenously in a 1-g dose every 12 hours to normal adults. Older adults may require dosage adjustment based on renal function. Other regimens include vancomycin plus gentamicin (Garamycin) or rifampin (Rifadin). Table 730,31 reviews therapeutic options for MRSA and vancomycin-resistant enterococcus (VRE). Attempts to identify the original infected person (source case) should be made by swabbing the nasopharynx of patients and staff near to the outbreak and treating those found to have MRSA infection. Staff and patients who are MRSA carriers should be isolated, and some authorities recommend treatment with topical mupirocin (Bactroban), which is applied twice daily for two weeks to the nares or other areas of skin carriage (e.g., wounds) to reduce the shedding of MRSA.32 Colonization recurs in about one half of treated subjects.
TABLE 7 Therapeutic Options for MRSA and VRE
| MRSA | ||
| Prevention | ||
| Careful handwashing | ||
| Isolation of infected patients | ||
| Removal of colonized catheters | ||
| Eradication of nasal carriage with mupirocin (Bactroban) | ||
| Treatment of systemic infections | ||
| Vancomycin (Vancocin) plus gentamicin (Garamycin) if strain is susceptible to the latter | ||
| Teicoplainin (Targocid)* if vancomycin is not well-tolerated | ||
| VRE | ||
| Prevention | ||
| Careful handwashing | ||
| Isolation of patients | ||
| ± Eradication of carriage with oral bacitracin (Bacticin) | ||
| Treatment of systemic infection: vanA type | ||
| Susceptible to ampicillin—(MIC 64 mg per mL) | ||
| Ampicillin or ampicillin/sulbactam (Unasyn) plus gentamicin or streptomycin, if sensitive to either of the latter | ||
| Europe only: teicoplanin (Targocid) plus ampicillin plus gentamicin or streptomycin | ||
| High resistance to ampicillin | ||
| Quinupristin/dalfopristin (Synercid) | ||
| Teicoplanin* plus streptomycin or gentamicin | ||
| Ciprofloxacin (Cipro) plus rifampin (Rifadin) plus gentamicin | ||
| Cefotaxime (Claforan) or ceftriaxone (Rocephin) plus fosfomycin (Monurol) | ||
| Chloramphenicol (Chloromycetin) or doxycycline (Vibramycin) plus rifampin plus quinupristin/dalfopristin | ||
| Treatment of systemic infections: vanB | ||
| Susceptible to ampicillin and gentamicin | ||
| Ampicillin or ampicillin-sulbactam plus gentamicin or streptomycin | ||
| Other possibilities | ||
| Ampicillin plus imipenem (Primaxin I.V.) ± gentamicin or streptomycin | ||
| Betalactam plus vancomycin plus gentamicin or streptomycin | ||
| Ampicillin plus a fluoroquinolone (unless fluoroquinolone-resistant) | ||
| Future approaches | ||
| MRSA | ||
| Amoxicillin-clavulanate potassium (Augmentin) | ||
| Cefotaxime (Claforan) plus fosfomycin | ||
| Injectable quinupristin/dalfopristin | ||
| Carbapenem with PBZa affinity | ||
| Oxzolidinones | ||
| VRE | ||
| Quinupristin/dalfopristin | ||
| New fluoroquinolones (moxifloxacin,† clinafloxacin,‡ gemifloxacin,‡ sitafloxacin†) | ||
| Glycylcyclines | ||
| Linezolid (Zyvox) | ||
| New glycopeptides (LY19145) and (LY333328) | ||
MRSA = methicillin-resistant Staphylococcus aureus; VRE = vancomycin-resistant Enterococcus; vanA, vanB = phenotypes of VRE; MIC = mean inhibitory concentration; PBZa = antipseudomonal beta-lactam.
*—Teicoplanin is not available in the United States.
†—Investigational drug.
‡—In development.
Adapted with permission from Michel M, Guttmann L. Methicillin-resistant Staphylococcus aureusand vancomycin-resistant enterococci: therapeutic realities and possibilities. Lancet 1997;349:1901–6, and Murray BE. Drug therapy: vancomycin-resistant enterococcal infections. New Engl J Med 2000;342:710–21.
VANCOMYCIN-RESISTANT ENTEROCOCCUS
As with MRSA, VRE presents a major problem in older patients, especially when an outbreak occurs in an institutional setting. Enterococci are the second most common organism in nosocomial urinary tract and wound infections and the third most common cause of nosocomial bacteremia in the United States.30,33,34 Over the past two decades, most enterococci have become resistant to betalactam antibiotics and, recently, resistance to aminoglycosides has become widespread.34 Thus, glycopeptide antibiotics, such as vancomycin in the United States and teicoplanin (Targocid) in Europe, have become the most reliable drugs to treat infections caused by multidrug-resistant enterococci.
In 1988, the first outbreak of VRE was reported. Currently, 10 percent of isolates reported to the Centers for Disease Control and Prevention are vancomycin-resistant.30,34 Bacteremia from these isolates have a mortality rate approaching 50 percent. Multiple strains of VRE have been identified, with phenotypes vanA, vanB and vanC described to date.
Therapeutic options for VRE vary by strain and resistance to other drugs. For vanA VRE, combinations of ampicillin and imipenem (Primaxin I.V.), vancomycin and ciprofloxacin (Cipro), ceftriaxone (Rocephin) with fosfomycin (Monurol), and chloramphenicol (Chloromycetin) have been used with limited success. For vanB VRE, the combination of teicoplanin (available in Europe but not in the United States) with gentamicin has been successful. Quinupristin/dalfopristin (Synercid) has been used against vancomycin-resistant Enterococcus faecium. For UTIs, nitrofurantoin (Macrodantin) has been used against susceptible VRE. Ongoing research shows that two semisynthetic glycopeptides, as well as two new fluoroquinolones, have in vitro activity against VRE.34
Because of the high level of antibiotic resistance, prevention of outbreaks and spread of VRE is crucial. As with MRSA, the best way to prevent an outbreak of VRE infection is by handwashing and proper handling of bodily secretions. In addition, limiting the use of oral and parenteral vancomycin will help control the further spread of resistance. Patients who are found to be colonized with VRE should be isolated, and proper infection control should be instituted (Table 7).30,31
Prevention
Most of the infectious diseases discussed in this article are preventable. Pneumococcal vaccine decreases the risk of pneumococcal bacteremia by 75 percent in immunocompetent persons 65 years or older.35,36 In nursing home residents, the vaccine is 50 to 60 percent effective in preventing pneumococcal bacteremia and 80 percent effective in preventing death from pneumonia.35,37 Influenza vaccines target the strains of influenza A and B that are most likely to cause illness during a particular season and reduce the risk of influenza infection by 30 to 70 percent among elderly persons in home settings. Preventive therapy with influenza vaccination has been shown to be about 58 percent effective in reducing influenza infection and has resulted in a 39 to 69 percent decrease in mortality from all causes during the influenza season in older adults.37–39
The influenza vaccine is usually administered as an inactivated virus preparation given intramuscularly. The optimal time for vaccination is from October to mid-November; however, the vaccination can be given throughout the influenza season and during outbreaks. Recently, a live attenuated virus preparation was developed that reduced the number of infections by 19 to 24 percent when given intranasally to healthy adult volunteers.40
UTIs are preventable by limiting the use of urinary catheters, and providing topical estrogen therapy for women and pharmacologic or surgical relief of prostatic hypertrophy for men. Physician awareness of these components of preventive measures is important to the care of the older adult.

