Sporotrichoid lymphocutaneous infection is an uncommon syndrome that is often misdiagnosed and improperly treated. Of the several hundred cases seen each year in the United States, the majority are caused by Sporothrix schenckii, Nocardia brasiliensis, Mycobacterium marinum or Leishmania brasiliensis. The “sporotrichoid” disease begins at a site of distal inoculation and leads to the development of nodular lymphangitis. Systemic symptoms are characteristically absent. By recognizing the distinct pattern of nodular lymphangitis and focusing on the diverse but limited etiologies, the physician can obtain the appropriate histologic and microbiologic studies and start targeted antimicrobial therapy. Therapy is generally continued for two to three months after the resolution of cutaneous disease.
Sporotrichoid lymphocutaneous infection is a syndrome characterized by the development of superficial cutaneous lesions that progress along dermal and subcutaneous lymphatics. It is usually preceded by a well-defined episode of cutaneous inoculation or trauma (historical information that can be a helpful clue to the diagnosis). The classic pattern is described as “sporotrichoid” after the infection caused by the dimorphic fungus Sporothrix schenckii. However, the microbiologic differential diagnosis of this syndrome includes a variety of common and uncommon microorganisms.
The syndrome is also known as nodular lymphangitis, a term that will be used in this article because it more accurately reflects the diverse microbiology and anatomic pathology of the condition. The infection is frequently confused with the more common forms of cellulitis and lymphangitis, typically caused by staphylococci and streptococci. Secondary infections by these common microbes can obscure the underlying cause, resulting in misdiagnosis and delay in the appropriate antimicrobial therapy.
Familiarity with the characteristics of nodular lymphangitis allows the family physician to narrow the microbiologic differential diagnosis, obtain appropriate laboratory studies and initiate directed antimicrobial therapy. If complicating factors exist (such as infection in immunocompromised patients, pregnant women, or children), referral to an appropriate subspecialist may be necessary.
Although the list of microorganisms is diverse, when considered in the context of exposure history, the differential diagnosis is, fortunately, limited (Table 11). The most common causes of nodular lymphangitis in the United States are S. schenckii, Nocardia brasiliensis, Mycobacterium marinum and Leishmania brasiliensis. Microbiologic diagnosis can usually be made when appropriate histologic stains and cultures of biopsied tissues are obtained. The clinical microbiology laboratory should be notified in advance so that optimal culture techniques can be performed.
TABLE 1 Organisms That Cause Nodular Lymphangitis
| Common causes |
| Sporotrichosis schenckii |
| Nocardia brasiliensis |
| Mycobacterium marinum |
| Leishmania brasiliensis |
| Uncommon causes |
| Nocardia asteroides |
| Atypical mycobacteria |
| Francisella tularensis |
| Blastomyces dermatitidis |
| Coccidioides immitis |
| Cryptococcus neoformans |
| Histoplasma capsulatum |
| Streptococcus pyogenes |
| Staphylococcus aureus |
| Pseudomonas pseudomallei |
| Bacillus anthracis |
| Cowpox virus |
Adapted with permission from Kostman JR and DiNubile MJ. Nodular lymphangitis: a distinctive but often unrecognized syndrome. Ann Int Med 1993;118:884.
Because of the diversity of microorganisms and the lack of official reporting of cases, it is impossible to establish the true incidence of nodular lymphangitis. Rates of infection will vary substantially as a function of exposure-prone activities, geographic location, travel history, occupation and hobbies. Practice experience and the lack of large case series suggest that nodular lymphangitis is a relatively uncommon condition.
Illustrative Case
A 69-year-old woman was admitted to a local hospital for treatment of refractory cellulitis involving her left index finger and extending up her arm. Two weeks before, she had pricked her index finger on a thorny cactus. Within two days a painful papule appeared at the inoculation site, and over the next two weeks tender lymphangitis developed. Despite the use of oral antistaphylococcal antibiotics, lymphangitis involving the forearm progressed. Subcutaneous nodules developed along the erythematous lymphangitic streak. The nodules and the papule on the affected finger began to ulcerate and produce a purulent discharge (Figure 1a and 1b).
FIGURE 1A.
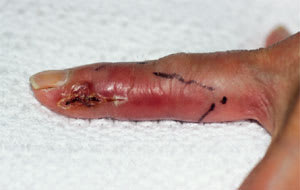
Erythema, induration and suppuration involving the left index finger.
FIGURE 1B.
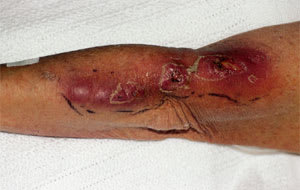
Nodular lymphangitis caused by Nocardia brasiliensis, with areas of suppuration extending up the left forearm and arm.
A variety of intravenous antistaphylococcal antibiotics were administered and surgical debridement of the proximal forearm was performed. Gram's stain of purulent fluid reportedly showed many neutrophils but no microorganisms. Therapy with a combination of supersaturated potassium iodide (SSKI) and itraconazole (Sporanox) was initiated empirically for a presumptive diagnosis of sporotrichosis.
The patient was transferred to a different hospital four days later for further management. At the time of admission, a physical examination confirmed the previous findings. In addition, she was noted to have bilateral conjunctival erythema and periorbital edema. Cultures obtained at the previous hospital were growing an Actinomyces-like organism.
The presumptive diagnosis was Nocardia-induced nodular lymphangitis, and therapy with trimethoprim-sulfamethoxazole (TMP-SMX) was started. The patient also showed signs of potassium iodide–induced conjunctivitis, which resolved within two days of discontinuing the combination of SSKI and itraconazole. The Actinomyces-like microbial isolate was obtained from the referring hospital and was subsequently identified as N. brasiliensis. The lesions dramatically improved within a week after initiation of TMP-SMX therapy and resolved completely after one month (Figure 2). The TMP-SMX therapy was continued for three months.
FIGURE 2.
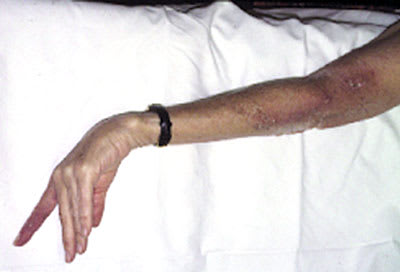
Improvement of nodular lymphangitis in left index finger, forearm and arm after two weeks of trimethoprim-sulfamethoxazole therapy.
Microbiology, Diagnosis and Therapy
The microbiologic diagnosis focuses on the four major causes of nodular lymphangitis in the United States: S. schenckii, N. brasiliensis, M. marinum and L. brasiliensis. Although these microorganisms have diverse taxonomic classifications, they do have several characteristics in common. First, they tend to be ubiquitous in soil or water, environmental niches where primary inoculation is likely to occur. (In the case of leishmaniasis, infection is geographically limited to areas of the world where transmitting sandflies are endemic.) Second, after primary inoculation, all of these micro-organisms are capable of eliciting granulomatous inflammation, necrosis and abscess formation. Third, systemic symptoms are characteristically absent.
The diagnosis and treatment of nodular lymphangitis caused by these organisms are summarized in Table 2.2–12
TABLE 2 Diagnosis and Treatment of the Major Causes of Nodular Lymphangitis
| Organism | Exposure history | Diagnosis | Therapy | Alternative therapy | Duration of treatment |
|---|---|---|---|---|---|
| Sporothrix schenckii | Gardening, soil contamination, splinters, animal scratches or bites | Biopsy and culture | Itraconazole (Sporanox), 200 mg daily2–4 | SSKI, 5 drops in water three times daily, slowly increased to 40 to 50 drops three times daily as tolerated2,5 Terbinafine (Lamisil), 250 mg twice daily4 | Two months after resolution of lesions |
| Nocardia brasiliensis | Gardening, soil contamination, splinters | Biopsy and culture | TMP-SMX (Bactrim, Septra), 2 double-strength tablets three times daily5,6 | Minocycline* (Minocin), 200 mg twice daily5–8 See text for additional alternatives. | Three months symptoms |
| Mycobacterium marinum | Aquariums, fish-handling, swimming in oceans, lakes or pools | Biopsy and culture | Rifampin (Rifadin), 15 mg per kg, daily with ethambutol (Myambutol), 25 mg per kg daily5,9,10 | Minocycline*,100 mg twice daily5,9,10 Rifampin, 10 to 15 mg per kg daily, with clarithromycin* (Biaxin), 30 mg per kg daily10 | Two to three months after resolution of |
| Leishmania brasiliensis | Residence in or travel to endemic areas | Biopsy and culture | Stibogluconate sodium, 20 mg per kg daily,5,11,12 or meglumine antimonate, 20 mg per kg daily11,12 | Amphotericin B (Fungizone IV), 0.25 to 1.00 mg per kg daily11 See text for additional alternatives. | 20 days |
SSKI = supersaturated potassium iodide; TMP-SMX = trimethoprim-sulfamethoxazole.
*—Sensitivities should be obtained before instituting therapy.
†—Only available through the Centers for Disease Control and Prevention in Atlanta.
Sporothrix schenckii
In the United States, S. schenckii is the most commonly recognized cause of nodular lymphangitis. It is a dimorphic fungus that exists saprophytically in nature and has a worldwide distribution. Infection may result from contact with soil and plant debris, thorns, sphagnum moss and timber.2,5,7,13 Inoculation has also been associated with scratches and bites from a variety of animals.5,7,14–17
Following a one- to 12-week incubation period, a nontender, erythematous, papulonodular lesion develops at the site of minor trauma or inoculation. Secondary erythematous, nodular lesions develop along the proximal lymphatic chain. The primary and secondary lesions frequently ulcerate and may discharge a thin, serous fluid. The lymphatic channels between nodules may be thickened and palpable. Regional adenopathy and fever are uncommon. Dissemination is rare, except in immunocompromised patients.
The characteristic appearance along with an exposure history should prompt consideration of the diagnosis. The clinical features of sporotrichosis overlap with those of other causes of nodular lymphangitis, and definitive diagnosis depends on microbiologic studies. Cultures obtained from fluid from the wound are usually negative for the organism. Routine and fungal histologic stains of biopsied tissue are nonspecific and rarely demonstrate the presence of the fungi. However, cultures of biopsied tissue frequently yield positive results and are therefore the diagnostic method of choice.18
SSKI has historically been used to treat cutaneous sporotrichosis,2,7 but it is associated with a number of adverse effects, including inflammation of lacrimal and parotid glands, gastrointestinal intolerance, hypothyroidism and rash.2,3,18 Recent studies have shown that itraconazole is an excellent alternative to SSKI.2,4,19 The drug is well tolerated and, in most cases, should be considered the drug of choice. Heat application is a useful adjunct to treatment. Hot compresses placed over the lesion for 40 to 60 minutes per day have been shown to lessen the severity of the symptoms of the disease.2
Although the duration of therapy is not firmly established, treatment should continue for approximately two months after resolution of the lesions. If the lesions recur after discontinuation, itraconazole can be reinstituted.
NOCARDIA SPECIES
Nocardia are filamentous gram-positive bacteria that are widely distributed in soil and water. Several species of Nocardia can cause lymphocutaneous infection, but N. brasiliensis is most often implicated in nocardial nodular lymphangitis. Infection may result from wound contamination by plant and soil matter.5,7
Following a one- to six-week incubation period, a tender nodule or subcutaneous abscess develops at the inoculation site. Secondary lesions may occur along proximal lymphatics. Purulent drainage and sinus formation may develop, and tender regional adenopathy and fever may occur.
Exposure history and clinical presentation may be indistinguishable from those of sporotrichosis. The characteristic morphology of the microorganism may occasionally be identified on Gram's stain of wound fluid and tissue. Nocardia species are partially acid-fast, a characteristic that may help distinguish them from other members of the Actinomyces group of organisms. Cultures of fluid and biopsy tissue are the diagnostic methods most likely to yield positive results. Because the microorganism tends to grow slowly, the microbiology laboratory should be notified of the possible presence of Nocardia in the clinical specimen so that cultures may be kept for several weeks.6,18
Sulfonamides are the preferred antibiotics, and the ease of use and oral bioavailability of TMP-SMX (Bactrim, Septra) makes it the mainstay of therapy. No well-controlled trials clearly define the dosage and duration of therapy. A high initial dosage of two double-strength tablets three times per day is reasonable. Monitoring of complete blood counts should be performed on a weekly basis, and if cytopenia develops, the dosage should be decreased. A three-month course of therapy is usually adequate to prevent relapse.
In patients who are unable to take sulfonamides, and in the rare instance of sulfonamide resistance, alternative antimicrobial therapy may be used after susceptibility testing. Minocycline (Minocin), amikacin (Amikin), carbapenems and quinolones are frequently active antimicrobials.
Mycobacteria
Various mycobacteria are capable of causing lymphocutaneous infection.5,7 M. marinum is the species most often implicated as a cause of nodular lymphangitis. M. marinum is found in fresh and saltwater environments, as well as on the fish and animals inhabiting them. Inoculation of microorganisms typically occurs in fish handlers and tropical aquarium hobbyists. Infection has also occurred after swimming in oceans, lakes and pools.9,20
Following a two- to eight-week incubation period, papules develop that may ulcerate or evolve into abscesses. Nodules may form along proximal lymphatics in a sporotrichoid manner. Systemic symptoms, regional adenopathy and dissemination are rare. The microorganisms are seldom visualized in tissue stains. The diagnosis is established by growing the microorganisms from cultures of biopsied tissue. Because specific incubation temperatures are required for optimal growth, the clinical microbiology laboratory should be alerted to the possible presence of M. marinum in the tissue specimen.
Most strains of M. marinum are resistant to isoniazid. Rifampin (Rifadin) and ethambutol (Myambutol) are active against the organism and have been used clinically. TMP-SMX, macrolides and the fluoroquinolones may be active, although testing the sensitivity of the isolate is recommended before use of these agents.10 As in sporotrichosis, the application of warm compresses is a useful adjunct to therapy. The recommended duration of therapy is two to three months beyond resolution of clinical symptoms.9,20
Leishmania
Leishmania are protozoal parasites that are transmitted to humans by Phlebotomus sandflies. Leishmaniasis is a major public health concern in Central and South America, the Middle East, Africa and Asia. There is also an endemic focus of Leishmania in south central Texas, where several cases of cutaneous infection have been reported.21 Various clinical syndromes can result after infection with the parasite. The cutaneous form of leishmaniasis occurs from two weeks to six months after the bite of a sandfly. A small nodule at the inoculation site usually evolves into a mildly painful, well-demarcated ulcer. Nodular lymphangitis may occur and is seen most frequently after infection caused by L. brasiliensis. In the lymphocutaneous form of the disease, systemic symptoms and regional adenopathy are rare.
Leishmaniasis should be considered in any person from an endemic area who has chronic localized skin lesions. The edge of a lesion should be biopsied and tissue submitted for histology and culture. Giemsastained tissue and cultures may reveal the presence of the parasite. Serologic tests for cutaneous leishmaniasis are of low sensitivity.
In many cases, cutaneous leishmaniasis resolves spontaneously over months to years. However, healed cutaneous L. brasiliensis infection can emerge months to years later as mucocutaneous infection. For this reason, the current recommendation is to start treatment at the time of initial diagnosis. The drugs of first choice are the pentavalent antimonial sodium stibogluconate or meglumine antimonate.11,12 Treatment is given for 20 days, during which time patients should be observed for side effects including pancreatitis and electrocardiogram abnormalities. Although not evaluated in a systematic way, amphotericin B (Fungizone IV), pentamidine (Pentam), allopurinol (Zyloprim) and azole antifungal compounds are alternative therapies.11,12,21 Warm compress application for two to three hours daily may lessen the severity of the symptoms of the disease.5
Other Causes of Nodular Lymphangitis
Other causes of nodular lymphangitis (Table 11) are exceedingly rare and should be considered primarily in the context of a suggestive exposure history. For a more detailed discussion of uncommon causes of nodular lymphangitis, the reader is referred to several articles on the topic.1,5,7,22 Microbiologic diagnosis can usually be made when appropriate histologic stains and cultures of biopsied tissue are obtained. These uncommon forms of nodular lymphangitis may require referral to an appropriate subspecialist.

