Thrombolytic therapy in acute myocardial infarction has decreased the mortality rate, but it is linked to the rare, potentially devastating complication of intracranial hemorrhage. Of the various techniques developed to deliver thrombolytic therapy, bolus injection is reported to provide more rapid and higher patency rates than standard infusion. Bolus injection has, however, been linked to an increased rate of intracranial hemorrhage. Mehta and colleagues combined data from several randomized controlled trials to establish the potential risks of bolus administration of thrombolytic agents.
A search of electronic literature databases, published abstracts of leading scientific conferences and references of review articles identified 39 randomized, controlled trials of different aspects of thrombolytic therapy. Only seven trials met the authors' criteria for size, focus on bolus therapy, selection of patients, follow-up duration, selection of outcome measures and methodologic quality. Data from these seven trials (see the accompanying figure) were pooled to assess rates of intracranial hemorrhage associated with bolus thrombolytic therapy.
FIGURE
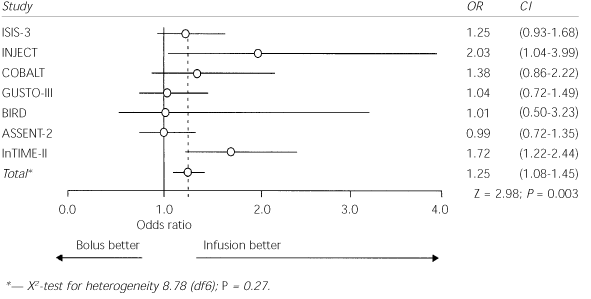
Intracranial hemorrhage in trials of bolus versus infusion of thrombolytic agents. (OR = odds ratio; CI = confidence interval; df = degrees of freedom; Z = distance between a value and the mean of a distribution expressed as the number of standard deviations from the mean; P = P value; X2 = chisquare test)
Reprinted with permission from Mehta SR, Eikelboom JW, Yusuf S. Risk of intracranial haemorrhage with bolus versus infusion thrombolytic therapy: a meta-analysis. Lancet 2000;356:451.
The seven trials involved 103,972 patients. More than 70 percent of the participants were men, and the median age was between 61 and 63 years. About one third of the patients were hypertensive. Although intracranial hemorrhage occurred in less than 1 percent of cases, it was significantly more common following bolus administration than following standard infusion (0.8 compared with 0.6 percent). Rates of nonhemorrhagic stroke and the overall 30-day mortality rate were not changed by bolus administration.
The authors conclude that bolus regimens carry an increased risk of intracranial hemorrhage with no evidence of reduction in mortality or reinfarction rates. The number of patients harmed is estimated to be small, approximately two per 1,000 treated, but all seven trials showed no effect or an increase in the consistent incidence of intracranial hemorrhage.
editor's note: At first glance, the increase in risk of hemorrhagic stroke from 0.6 to 0.8 percent, although a 25 percent increase and statistically significant, may not seem like a big deal. On reflection, however, this study should make us pause before switching to bolus techniques. First, the increased risk of hemorrhage and the lack of benefit in terms of mortality and reinfarction are consistently reported and appear to be real effects. Second, this small increase in risk translates into large numbers of patients potentially harmed, because approximately 1.5 million persons are admitted to U.S. hospitals every year with acute myocardial infarction. Third, intracranial hemorrhage is a significant event and not one that patients should be exposed to for little or no proven benefit. Finally, without reading each study, we cannot know how cases of intracranial hemorrhage were detected. It is not uncommon for patients to have residual physical or cognitive problems following myocardial infarction and the dramatic treatments now administered. How many of these are due to undiagnosed intracranial bleeding? Even a few missed cases would make a dramatic difference in the conclusions of this study. The bottom line appears to be primum non nocere, especially when we are striving to do everything possible to help patients survive acute myocardial infarction without deficits.—A.D.W.

