Pain, which is often inadequately treated, accompanies the more than 23 million surgical procedures performed each year and may persist long after tissue heals. Preemptive analgesia, an evolving clinical concept, involves the introduction of an analgesic regimen before the onset of noxious stimuli, with the goal of preventing sensitization of the nervous system to subsequent stimuli that could amplify pain. Surgery offers the most promising setting for preemptive analgesia because the timing of noxious stimuli is known. When adequate drug doses are administered to appropriately selected patients before surgery, intravenous opiates, local anesthetic infiltration, nerve block, subarachnoid block and epidural block offer benefits that can be observed as long as one year after surgery. The most effective preemptive analgesic regimens are those that are capable of limiting sensitization of the nervous system throughout the entire perioperative period.
Pain is thought to be inadequately treated in one half of all surgical procedures.1 In addition to immediate unpleasantness, painful experiences can imprint themselves indelibly on the nervous system (Figure 1), amplifying the response to subsequent noxious stimuli (hyperalgesia) and causing typically painless sensations to be experienced as pain (allodynia). A chronic condition sometimes develops that produces continuous pain long after surgery. Prior painful experiences are a known predictor of increased pain and analgesic use in subsequent surgery.2,3
FIGURE 1. Pain Sensitization
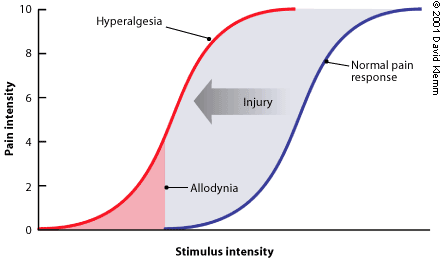
Noxious stimuli can sensitize the nervous system response to subsequent stimuli. The normal pain response as a function of stimulus intensity is depicted by the curve at the right, where even strong stimuli are not experienced as pain. However, a traumatic injury can shift the curve to the left. Then, noxious stimuli become more painful (hyperalgesia) and typically painless stimuli are experienced as pain (allodynia).
The process by which the nervous system becomes sensitized is active early in life, as demonstrated by the enhanced pain-related behavior that occurs in circumcised boys during subsequent vaccination compared with boys who were not circumcised4 and boys who were circumcised after application of a local anesthetic cream.5 Long-term painful sequelae of surgery in adults occur following amputation of an extremity,6 thoracotomy,7 laparotomy,8,9 herniorrhaphy10 and mastectomy.11 Moreover, even low levels of residual pain are associated with decreased physical and social function as well as a diminished perception of overall health.9 Questions that confront a physician whose patient is about to undergo surgery include: “What is the process by which painful stimuli sensitize the nervous system?” and “How can this sensitization be prevented to reduce short-term and long-term pain and its consequences?”
Pain Response
Both the peripheral and the central nervous systems (CNS) are involved in the perception of pain, with the spinal and supraspinal components of the CNS playing key roles12 (Figure 2).13 The transduction of noxious stimuli begins with peripheral nociceptors. Signals from these nociceptors travel primarily along small myelinated A and unmyelinated C fibers with soma lying in the dorsal root ganglion. Their axons synapse in the dorsal horn of the spinal cord, where the neurons of laminae I, II and V are most involved in the perception of pain.
FIGURE 2. Providing Postoperative Pain Relief
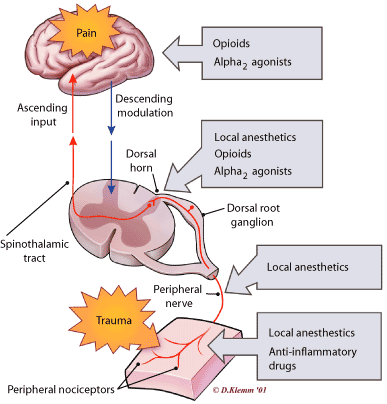
The pain pathway and interventions that can modulate activity at each point.
Redrawn with permission from Kehlet H, Dahl JB. The value of “multimodal” or “balanced analgesia” in postoperative pain treatment. AnesthAnalg 1993;77:1049.
The signals then travel along the spinothalamic tract of the spinal cord to the thalamus and the cortex. Large fiber inputs from other sensory modalities and descending pathways can modulate activity in the dorsal horn, where these descending pathways may provide a physiologic explanation for the increased pain experienced by patients who have high levels of depression and anxiety.3,9 Painful stimuli ultimately cause activity in both the somatotopically appropriate portion of the sensory cortex and the limbic system.14
The response to noxious stimuli can be modulated by their repeated application.12 For example, peripheral nociceptors become more responsive with the repeated application of noxious stimuli. Their sensitivity can be further enhanced by many tissue factors and inflammatory mediators released in the course of tissue injury. The response of neurons in the dorsal horn of the spinal cord of experimental animals has been found to be biphasic. The initial response to a noxious stimulus is brief and correlates with the sharp, well-localized initial pain. The second phase of the response is more prolonged and correlates with the dull, diffuse pain experienced after the initial injury. Experimentally, this second phase is associated with a growing region of hypersensitivity around the point where the noxious stimulus was initially applied.
The process through which the neurons of the dorsal horn of the spinal cord become sensitized by prior noxious stimuli is often referred to as “windup” or “central sensitization. “Much less is known about pain-induced sensitization of the supraspinal components of the CNS. Collectively, however, the above mechanisms enhance sensitivity to noxious stimuli and may increase the level of pain experienced following surgery.
Preemptive Analgesia
One of the most critical observations concerning central sensitization is the role played by the first phase of the pain response. Opiates administered before the first phase and reversed with the opiate antagonist naloxone (Narcan) before the expected onset of the second phase were capable of preventing this late stage of the pain response.15 Thus, preventing the initial neural cascade could lead to long-term benefits by eliminating the hypersensitivity produced by noxious stimuli.
Animal experiments demonstrated the benefits of preventing central sensitization by infiltrating with local anesthetics,16 an approach that was particularly effective with pain associated with deafferentation, as might occur with amputation.17 Collectively, results like these led to the concept of preemptive analgesia—initiating an analgesic regimen before the onset of the noxious stimulus to prevent central sensitization and limit the subsequent pain experience.18
Surgery may be the clinical setting where preemptive analgesia techniques will be the most effective because the onset of the intense noxious stimulus is known (Figure 3).19 To appreciate the design of clinically effective strategies in this setting, it is essential to recognize that otherwise adequate levels of general anesthesia with a volatile drug such as isoflurane (Forane) do not prevent central sensitization.20 Thus, the potential for central sensitization exists even in unconscious patients who appear to be clinically unresponsive to surgical stimuli.
FIGURE 3. Comparison of Analgesic Regimens
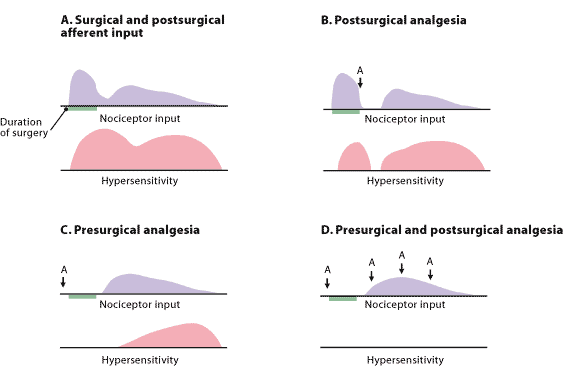
Schematic of preemptive analgesia with an emphasis on preventing sensitization of the nervous system throughout the perioperative period. A typical experience without intervention is shown in A, which depicts pain from the initial surgery and the hypersensitivity that subsequently develops. In B, analgesia (A) administered after sensitization may decrease pain somewhat but has little long-term benefit. Analgesia administered before surgery limits the pain from that stimulus and decreases subsequent hypersensitivity, as shown in C. However, the most effective preemptive analgesic regimen is initiated before surgery and continued throughout the postoperative period, as illustrated in D. Although timing of the intervention is important, it must also be capable of preventing sensitization of the nervous system.
Redrawn with permission from Woolf CJ, Chong MS. Preemptive analgesia—treating postoperative pain by preventing the establishment of central sensitization. Anesth Analg 1993;77:368.
Efficacy
Despite solid demonstrations of its effects in some animal models, considerable controversy surrounds the use of preemptive analgesia in clinical settings. This controversy exists because not all clinical trials of preemptive analgesia have resulted in clear demonstrations of its efficacy. In evaluating clinical trials of preemptive analgesia, the timing of the intervention is only one factor. It is also essential to consider the ability of the intervention to prevent central sensitization and whether other aspects of the perioperative pain experience may be of sufficient duration and intensity to mask any intraoperative benefits from the preemptive analgesia.
Many clinical protocols have mirrored the laboratory studies using animals that gave birth to the concept of preemptive analgesia. However, these animal experiments employed painful stimuli of intensity, duration and somatotropic extent that were generally far less than that experienced during even relatively minor surgery in human patients.21 Therefore, it should not be surprising that interventions with a limited capacity for preventing central sensitization, when applied for only a small portion of the perioperative period, fail to demonstrate a preemptive analgesia effect.22
Strategies
Preemptive analgesia strategies have involved interventions at one or more sites along the pain pathway (Figure 2).13 These strategies have included infiltration with local anesthetics,23,24 nerve block,25 epidural block,8,26–30 subarachnoid block,23 intravenous analgesics31 and anti-inflammatory drugs.32 For example, infiltrating the incision site with the long-acting local anesthetic bupivacaine (Marcaine) after administering general anesthesia and before incision was found to be more effective for hernia repair pain than either spinal anesthesia or general anesthesia alone, and these benefits appeared to last many days.23
Although spinal anesthesia clearly provides a better intraoperative block of the surgical stimulus, the more effective postoperative analgesia produced by infiltration with a longacting local anesthetic may have been an important factor in preventing central sensitization. During routine laparoscopy with general anesthesia, infiltrating with local anesthetic before incision was found to be more effective than infiltrating with saline before incision or infiltrating with local anesthetic at the conclusion of the procedure.24 There were no differences among patients who received either saline or local anesthetic at the conclusion of the procedure. Blockade of peripheral nerves with local anesthetics can have a beneficial effect on pain after hernia repair, outlasting the duration of the nerve block even when the repair is performed with spinal anesthesia.25
Intravenous opiates or ketamine (Ketalar) administered before incision can lead to decreases in wound hyperalgesia days after the surgery.31 Anti-inflammatory drugs may play an important role in perioperative pain management by reducing the inflammatory response in the periphery and thereby decreasing sensitization of the peripheral nociceptors. This should help attenuate central sensitization.13,32 However, clear demonstrations of this last hypothesis have yet to be made.
EPIDURAL TECHNIQUE
One of the most important techniques for perioperative pain control involves the use of an epidural catheter. These catheters are usually placed at a lumbar or thoracic interspace before the start of major thoracic, abdominal or orthopedic procedures and can be maintained for several days to provide postoperative analgesia. Typically, local anesthetics and opiates, alone or in combination, are administered through the epidural catheter as an infusion or a bolus to provide analgesia.
Because a surgical level of anesthesia can be achieved for procedures on the lower abdomen and lower extremities with epidural anesthesia alone, its intraoperative use might be expected to provide one of the best means of blocking noxious stimuli and preventing central sensitization. However, even when an epidural catheter has been placed for the purpose of postoperative pain control, many physicians hesitate to use it intraoperatively, and others employ epidural drug regimens of limited efficacy. One reason for this is concern about hypotension related to epidurally administered local anesthetic blockade of the sympathetic nervous system,33 despite data demonstrating that neuroaxial blockade with local anesthetics is beneficial in hemorrhagic shock.34 Another reason that indwelling epidural catheters are not used to full effect is uncertainty about the efficacy of preemptive analgesia and how to achieve it.
One of the earliest studies of the efficacy of preemptive epidural analgesia involved lower extremity amputation.28 Patients in the intervention group received several days of epidural analgesia for their painful lower extremity before receiving epidural anesthesia during surgery. This was followed by several days of postoperative epidural analgesia. In the control group, the amputation was performed with epidural anesthesia, and patients received intravenous and oral opiates for analgesia.
One year after surgery, the intervention group demonstrated dramatic reductions in phantom limb pain, stump pain and phantom sensation when compared with the control group. However, even the control group experienced one-year postoperative phantom limb pain at one half of the historical rate of 70 percent,6 presumably because performing the amputation with regional blockade limited central sensitization.
A number of subsequent studies reported mixed results for lower extremity amputation, although a recent editorial that accompanied one of the negative-outcome studies made a strong argument that the likelihood of success improves with the ability to effectively prevent central sensitization throughout the entire perioperative period.35
Preemptive epidural analgesia using opiates, local anesthetics or a combination of the two is effective for both thoracic and abdominal procedures. Short-term and long-term benefits have been demonstrated for thoracotomy,27,30 although again, the extent of benefit varies with the ability to prevent central sensitization. The benefits of preemptive epidural analgesia during abdominal surgery8,29 include decreased pain during hospitalization, reduced length of hospital stay, more rapid return to preoperative levels of activity and less long-term residual pain.
Some benefit has been observed when using epidural opiates alone for mastectomy,26 thoracotomy,27 extremity surgery26 and lower abdominal surgery.8 In evaluating these and other studies, it is essential to assess the quality of postoperative pain relief. Aggressive postoperative pain therapy with an epidural catheter may be essential to preserve any benefit obtained intraoperatively in the intervention group, although it may bias the control group in the direction of the intervention group.22
Final Comments
Thus, the process by which the nervous system becomes sensitized by noxious stimuli offers both an explanation for the pain that persists after a traumatic experience, and a means for reducing the short-term and long-term painful effects of such stimuli. The available studies indicate that modest short-term interventions are not likely to provide meaningful benefits in the face of massive tissue injury. However, preincisional infiltration with a long-acting local anesthetic can be beneficial in appropriately chosen patients undergoing minor surgery.
Analgesic strategies for more extensive procedures require interventions capable of preventing central sensitization throughout the perioperative period and, therefore, require a commitment from the entire surgical team. Ultimately, multimodal approaches13 that address multiple sites along the pain pathway may prove necessary to adequately prevent central sensitization in many surgical procedures. Unfortunately, the resources to provide outstanding pain relief following surgery may only become available once the clinical and economic benefits of pain relief are clearly demonstrated.36

