
Am Fam Physician. 2001;63(11):2201-2209
People with valvular heart disease are living longer, with less morbidity, than ever before. Advances in surgical techniques and a better understanding of timing for surgical intervention account for increased rates of survival. Echocardiography remains the gold standard for diagnosis and periodic assessment of patients with valvular heart disease. Generally, patients with stenotic valvular lesions can be monitored clinically until symptoms appear. In contrast, patients with regurgitant valvular lesions require careful echocardiographic monitoring for left ventricular function and may require surgery even if no symptoms are present. Aside from antibiotic prophylaxis, very little medical therapy is available for patients with valvular heart disease; surgery is the treatment for most symptomatic lesions or for lesions causing left ventricular dysfunction even in the absence of symptoms.
Family physicians routinely encounter patients with valvular heart disease. Although major advances in our understanding of this disease have been made in the past two decades, only recently has there been evidence-based literature from which to draw conclusions about its diagnosis and management. The American College of Cardiology and American Heart Association (ACC/AHA) Task Force recently published comprehensive practice guidelines to help clinicians manage this challenging disorder.1,2 Improved surgical techniques, noninvasive techniques for surveillance of left ventricular function and guidelines for the timing of surgery have likely combined to contribute to improved survival and decreased morbidity in patients with chronic valvular heart disease.1,3
Aortic Stenosis
Aortic stenosis is usually idiopathic and results in calcification and degeneration of the aortic leaflets. Persons born with a bicuspid aortic valve are predisposed to develop aortic stenosis. The classic symptoms of angina, exertional syncope and dyspnea generally follow an extended latent period during which the patient is asymptomatic. The survival of patients with aortic stenosis is nearly normal until the onset of symptoms, when survival rates decrease sharply. After the onset of symptoms, average survival is only two to three years.1,3,4 Although the rate of progression of aortic stenosis is variable and difficult to predict,4 approximately 75 percent of patients with aortic stenosis will be dead three years after the onset of symptoms if the aortic valve is not replaced. Some patients with severe aortic stenosis remain asymptomatic, while others with moderate stenosis have symptoms attributable to the condition.
PHYSICAL EXAMINATION
The most common physical sign of aortic stenosis is a systolic ejection murmur that radiates to the neck. In mild aortic stenosis, the murmur peaks early in systole, but as the severity of stenosis increases, the murmur peaks progressively later in systole and may become softer as cardiac output decreases (Table 1). As the stenosis worsens, the aortic component of the second heart sound may become diminished. The timing and amplitude of the carotid pulse correlate with the severity of aortic stenosis.5(p118) Later in the disease, the carotid upstrokes become diminished and delayed1,2,5 (parvus et tardus; Figure 1).
| Findings | Murmur | S1 | S2 | Other findings | Maneuvers |
|---|---|---|---|---|---|
| Aortic stenosis | Mid to late systolic; may be soft or absent if severe | Normal | Single or paradoxically split | Carotid upstrokes diminished and delayed; S3or S4 may be present | Murmur softer with Valsalva maneuver |
| Mitral stenosis | Diastolic rumble | Loud | Normal | Opening snap may be present | Murmur increased with brief exercise |
| Aortic regurgitation | Blowing diastolic | Soft | Normal | Wide pulse pressure, systolic hypertension, hyperdynamic circulation | Murmur increased with handgrip or squatting |
| Mitral regurgitation | Holosystolic | Soft | Normal or split | S3 may be present; carotid upstrokesbrisk | Murmur louder with Valsalva maneuver |
| Mitral valve prolapse | Mid to late systolic | Normal | Normal | Mid-systolic click | Murmur increased with standing |
ECHOCARDIOGRAPHY
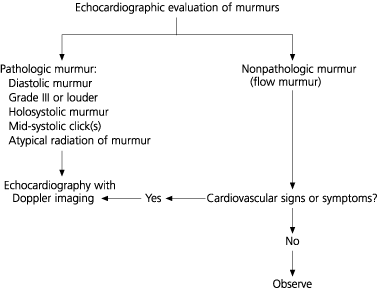
Echocardiography with Doppler provides an accurate assessment of aortic valve area and transvalvular gradient and also can be used to estimate left ventricular hypertrophy and ejection fraction.1,2 Patients with severe aortic stenosis should undergo echocardiography annually. This frequent monitoring is more for evaluation of left ventricular function than for estimation of valve area. In patients with mild to moderate aortic stenosis, performance of echocardiography every two to five years is appropriate for monitoring.1
MANAGEMENT
Patients with signs of aortic stenosis on physical examination should undergo testing, including chest x-ray, electrocardiograph (ECG) and echocardiograph.1 There is presently no medical therapy for aortic stenosis. Aortic valve replacement is the only effective treatment that will relieve this mechanical obstruction.3 The decision for surgery is based on the presence of at least moderately severe aortic stenosis with symptoms or signs of left ventricular dysfunction. The prognosis with surgery is excellent, even for elderly patients (in the absence of comorbid conditions). In fact, the age-adjusted survival rate in postoperative patients is similar to that of the general population.3 Cardiac catheterization with angiography is warranted in the appropriate age and risk groups before aortic valve replacement is undertaken.1
Exercise stress testing is not recommended in patients with aortic stenosis, especially those who are symptomatic. Asymptomatic patients may undergo exercise stress testing only under the careful supervision of an experienced physician and with close monitoring of blood pressure levels and ECGs.1 Patients with severe aortic stenosis should limit vigorous physical activity. Patients with aortic stenosis are at moderate risk for development of endocarditis and should receive endocarditis prophylaxis before selected procedures.6
Mitral Stenosis
The decline in rheumatic fever has reduced the incidence of mitral stenosis, a sequelae of rheumatic heart disease primarily affecting women.3 Mitral stenosis has a progressive, lifelong course that is slow and stable in the early years, with rapid acceleration later in life. There is generally a latent period of 20 to 40 years between the occurrence of rheumatic fever and the onset of symptoms.1
Increased left atrial pressure and decreased cardiac output produce the symptoms of mitral stenosis. Elevated left atrial pressure eventually causes pulmonary vasoconstriction, pulmonary hypertension and compromise of right ventricular function. Many patients remain asymptomatic until atrial fibrillation develops or until pregnancy occurs, when there is increased demand on the heart. Symptoms are generally those of left-sided heart failure: dyspnea on exertion, orthopnea and paroxysmal nocturnal dyspnea. Patients may also present with hemoptysis and signs of right-sided heart failure.
PHYSICAL EXAMINATION
An apical rumbling, diastolic murmur is characteristic and will immediately follow an opening snap, if present. The rumble is loudest in early diastole but, in patients with mild mitral stenosis or mitral stenosis with low cardiac output, the murmur may be difficult to hear (Table 1).1,3,5(p271) It can be accentuated by placing the patient in the left lateral decubitus position and using the bell of the stethoscope. Brief exercise (such as walking in the hallway) may also accentuate the murmur. A loud first heart sound is common. A right ventricular lift, elevated neck veins, ascites and edema are later signs of right ventricular overload with pulmonary hypertension.
ECHOCARDIOGRAPHY
Echocardiography is the study of choice for diagnosing and assessing the severity of mitral stenosis. It is also useful for obtaining an accurate valve area, assessing right ventricular function and determining suitability for balloon mitral valvotomy.
MANAGEMENT
In patients who are asymptomatic, an annual history and physical examination, as well as a chest x-ray and ECG, are recommended. Apart from endocarditis prophylaxis, no medical therapy is indicated.3,6 When mild symptoms develop, diuretics may be helpful in reducing left atrial pressure and decreasing symptoms. If atrial fibrillation develops, rate control is critical since tachycardia will further decrease left ventricular filling, reduce cardiac output and increase left atrial pressure, leading to more symptoms.1,3
The timing of surgery correlates with the clinical outcome. If symptoms are more than mild or if there is evidence of pulmonary hypertension, mechanical intervention is warranted and delaying intervention worsens prognosis.3
Unlike valvotomy for aortic stenosis, balloon valvotomy for mitral stenosis provides excellent mechanical relief with long-term benefit.3 Mitral balloon valvotomy is recommended for patients who have New York Heart Association class II, III or IV, moderate or severe mitral stenosis and favorable valve morphology. The valves must be pliable, noncalcified and free of subvalvular distortion.1,3 Candidates for valvotomy must have no significant mitral regurgitation.1
Aortic Regurgitation
Aortic regurgitation results from disease affecting the aortic root or aortic leaflets, preventing their normal closure. Common causes of aortic regurgitation include endocarditis, rheumatic fever, collagen vascular disease, aortic dissection and syphilis.3 Bicuspid aortic valves are also prone to regurgitation. In chronic aortic regurgitation, the stroke volume is increased, which in turn causes systolic hypertension, high pulse pressure and increased afterload. The afterload in aortic regurgitation may be as high as that occurring in aortic stenosis.1
Patients may be asymptomatic until severe left ventricular dysfunction has developed. The initial signs of aortic regurgitation are subtle and may include decreased functional capacity or fatigue. As the disease progresses, the typical presentation is that of left-sided heart failure: orthopnea, dyspnea and fatigue.
Systolic dysfunction is initially reversible, with full recovery of left ventricular size and function after aortic valve replacement. Over time, however, progressive chamber enlargement with decreased contractility make recovery of left ventricular function and improved survival impossible, even with surgery.1
PHYSICAL EXAMINATION
A diastolic blowing murmur heard along the left sternal border is characteristic of aortic regurgitation. A diastolic rumble may also be heard over the apex. The peripheral signs of hyperdynamic circulation indicate severe disease (Table 1).1,3,5 (p271) Some of these include Quincke's pulse (alternating blanching and erythema of the nailbed with gentle pressure applied) and Musset's sign (head bobbing).3
ECHOCARDIOGRAPHY
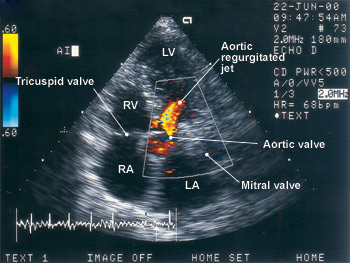
Echocardiography with Doppler ultrasonography provides information about aortic valve morphology and aortic root size, and a semiquantitative estimate of the severity of aortic regurgitation (Figure 2). It provides valuable information about left ventricular size and function. Asymptomatic patients with severe aortic regurgitation should undergo annual echocardiographic examination. Patients with milder forms of aortic regurgitation should have echocardiography if any change in symptoms occurs.2
MANAGEMENT
Aortic regurgitation should be corrected if the symptoms are more than mild. Compelling evidence supports surgical correction before the onset of permanent left ventricular damage, even in asymptomatic patients.1,3 As in patients with mitral valve regurgitation, timing of surgical intervention correlates with a good outcome. In patients with chronic aortic regurgitation, surgery should be performed before the ejection fraction falls below 55 percent or the end-systolic dimension exceeds 55 mm.1,3 The end-systolic dimension is another gauge of left ventricular function that helps determine contractility and is less dependent on preload than ejection fraction.
Afterload reduction with vasodilators has been shown to improve left ventricular performance and reduce aortic regurgitation. Therapy with nifedipine (Procardia) in particular has been shown to delay the need for surgery by two to three years in patients with severe aortic regurgitation and normal left ventricular function.3
Although the mainstay of surgical treatment has been replacement of the aortic valve with a tissue or mechanical prosthesis, interest in autograft procedures and aortic valve reconstruction is increasing.3 All patients with aortic regurgitation should receive appropriate endocarditis prophylaxis for selected procedures.6
Mitral Regurgitation
Causes of chronic mitral regurgitation include infective endocarditis, degenerative valvular disease (mitral valve prolapse) and rheumatic fever.3 Chronic mitral regurgitation is a state of volume overload leading to the development of left ventricular hypertrophy. The left atrium also enlarges to accommodate the regurgitant volume. This compensated phase of mitral regurgitation varies in duration but may last many years.1 The prolonged state of volume overload may eventually lead to decompensated mitral regurgitation. This phase is characterized by impaired left ventricular function, decreased ejection fraction and pulmonary congestion.
PHYSICAL EXAMINATION
A holosystolic murmur that may radiate to the axilla, the upper sternal borders or the subscapular region is apparent on physical examination. There may be displacement of the left ventricular impulse. A soft first heart sound and a widely split second heart sound may be present. An S3 indicates severe disease but does not necessarily indicate heart failure (Table 1).1,3,5(p271)
ECHOCARDIOGRAPHY
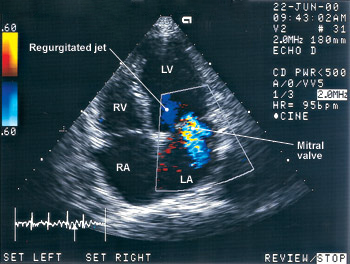
Echocardiography can be used to determine the etiology and morphology of mitral regurgitation, which are important in determining suitability for mitral valve repair (Figure 3). Although a variety of methods are used to quantify mitral regurgitation, echocardiography provides only a semiquantitative estimate of its severity.3 In patients with severe mitral regurgitation, annual or semiannual estimation of left ventricular function (ejection fraction and end-systolic dimension) is indicated, even in the absence of symptoms.
MANAGEMENT
In patients with chronic mitral regurgitation, left ventricular damage can occur while the patient remains asymptomatic. Therefore, surgery is indicated if left ventricular dysfunction has begun to develop, even in the absence of symptoms.1,3 Generally, the ejection fraction of patients with mitral regurgitation should be above normal. If the ejection fraction falls to below 60 percent, the prognosis worsens. End-systolic dimension is another helpful gauge in determining the need for surgery. An end-systolic dimension over 45 mm is also consistent with a poor prognosis.
In summary, patients should be referred for surgery if symptoms are moderate to severe, if the ejection fraction is less than 60 percent or if the end-systolic dimension approaches 45 mm, even in the absence of symptoms1,3 (Figure 3). Properly timed surgery gives patients with mitral regurgitation a postoperative survival rate similar to that of the general population. No studies have demonstrated successful use of medical therapy, other than as prophylaxis against endocarditis, in the treatment of chronic mitral regurgitation.1,3
Because mitral valve repair has a lower rate of mortality associated with surgery and a better late outcome than mitral valve replacement, repair should be performed whenever possible.1 The better outcome of repair is thought to be associated with the geometric preservation of the left ventricular and mitral valve apparatus. Mitral valve repair also eliminates the need for anticoagulation therapy in patients who are in sinus rhythm and prevents the chance of prosthetic valve failure. Mitral valve repair, however, is more technically demanding than mitral valve replacement, making the surgeon's skill and experience in performing the repair a predictor of success.1 The success of mitral valve repair surgery also depends on the location and type of mitral valve disease that caused the mitral regurgitation.
Mitral Valve Prolapse
Mitral valve prolapse occurs when varying portions of one or both leaflets of the mitral valve extend or protrude abnormally above the mitral annulus into the left atrium.
Although the prevalence of mitral valve prolapse was once thought to be as high as 15 percent in the general population, more recent studies using new echocardiographic criteria for diagnosis have suggested a prevalence of approximately 2.4 percent.7
Many symptoms have been attributed to mitral valve prolapse, including chest pain, dyspnea, anxiety and palpitations. Previous studies have linked mitral valve prolapse to serious long-term complications such as atrial fibrillation, syncope, stroke and sudden death. More recent research, however, reveals no increased risk of these complications when patients with mitral valve prolapse are compared with those who have no prolapse.7,8 In addition, the age-adjusted survival rate in patients with mitral valve prolapse is similar to that in patients without mitral valve prolapse.1
PHYSICAL EXAMINATION
The mid-systolic click, often accompanied by a late systolic murmur, is the auscultatory hallmark of mitral valve prolapse (Table 1). The click results from a sudden tensing of the mitral valve apparatus as the leaflets prolapse into the left atrium. When end-diastolic volume is decreased (as with standing), the clickmurmur complex occurs shortly after the first heart sound. Maneuvers that increase enddiastolic volume (such as squatting) cause the click-murmur complex to move toward the second heart sound.
ECHOCARDIOGRAPHY
New echocardiographic criteria for the diagnosis of mitral valve prolapse are based on the three-dimensional shape of the valve with clinical correlation. The new criteria have improved sensitivity and specificity for diagnosis of mitral valve prolapse.7 All patients with signs or symptoms of mitral valve prolapse should have an initial echocardiograph. Serial echocardiography is usually not necessary unless mitral regurgitation is present.1
MANAGEMENT
Reassurance is a major component of the management of patients with mitral valve prolapse. Patients should be reminded of the low incidence of serious complications associated with mitral valve prolapse, and physicians should attempt to allay fears of serious underlying heart disease. Patients with mitral valve prolapse and symptoms such as chest pain, palpitations or anxiety often respond to treatment with beta-blocking medications. In many cases, cessation of alcohol, tobacco and caffeine use may be sufficient to control symptoms.1
Use of prophylaxis against endocarditis has been controversial in patients with mitral valve prolapse. Currently, antibiotic prophylaxis is not recommended for use in patients with mitral valve prolapse in the absence of mitral regurgitation. Only patients with prolapsing and regurgitant valves, evidenced by audible clicks and murmurs or by mitral regurgitation demonstrated on Doppler imaging, should receive antibiotics.6
Final Comment
Great progress has been made in improving rates of morbidity and mortality in patients with valvular heart disease. Successful management of patients with valvular heart disease requires an evidence-based approach to echocardiography and to surgical intervention. Echocardiography should assess not only the valvular lesion but also the compensatory changes of the heart in response to the lesion. Timing of surgical intervention often correlates with outcome. Most patients with acquired valvular heart disease are at risk for developing endocarditis and should receive prophylactic antibiotics.