
Am Fam Physician. 2002;65(7):1341-1347
Epidural abscess of the spinal column is a rare condition that can be fatal if left untreated. Risk factors for epidural abscess include immunocompromised states such as diabetes mellitus, alcoholism, cancer, and acquired immunodeficiency syndrome, as well as spinal procedures including epidural anesthesia and spinal surgery. The signs and symptoms of epidural abscess are nonspecific and can range from low back pain to sepsis. The treatment of choice in most patients is surgical decompression followed by four to six weeks of antibiotic therapy. Nonsurgical treatment may be appropriate in selected patients. The most common causative organism in spinal epidural abscess is Staphylococcus aureus. Spinal epidural abscess involving actinomycosis is rare.
Spinal epidural abscess has an estimated incidence rate of 0.2 to 2.8 cases per 10,000 per year, with the peak incidence occurring in people who are in their 60s and 70s. The most common causative agent is Staphylococcus aureus.1,2 Epidural abscess caused by actinomycosis is rare; fewer than 80 cases have been reported since the organism was identified in 1878.3,4
The incidence of spinal epidural abscess appears to have increased in the United States since the 1980s, possibly because of an increase in the age of the population, the number of spinal procedures performed,5,6 intravenous (IV) drug abuse, and the number of patients with acquired immunodeficiency syndrome (AIDS).7 Associated predisposing conditions include a compromised immune system such as occurs in patients with diabetes mellitus, AIDS, chronic renal failure, alcoholism, or cancer,8-10 or following epidural anesthesia, spinal surgery, or trauma11 (Table 1). No predisposing condition can be found in 20 percent of patients with spinal epidural abscess, and the condition has been reported in patients with no predisposing risk factors.12
| Immunodeficiency | |
| AIDS | |
| Alcoholism | |
| Chronic renal failure | |
| Diabetes mellitus | |
| Intravenous drug abuse | |
| Malignancy | |
| Spinal procedure or surgery | |
| Spinal trauma | |
Illustrative Case
A 72-year-old woman presented with decreased appetite, severe low back pain, urinary incontinence, and difficulty ambulating after a fall three weeks earlier. Her significant prior medical history included diabetes mellitus, hypertension, and depression. The patient was alert and oriented, and had stable vital signs but was mildly febrile. Positive physical findings included poor dentition, periodontal disease, diffusely tender abdomen, decreased rectal sphincter tone, absence of deep tendon reflex in both lower extremities, and mild tenderness on palpation of the lower thoracic spine and upper lumbar region with no external evidence of injury.
Chest and abdominal radiographs were unremarkable. Initial laboratory investigation showed leukocytosis with a left shift (white blood cell count [WBC], 22,000 per mm3 [22 × 109 per L] with 68 percent neutrophils and 14 percent bands) and an elevated erythrocyte sedimentation rate of 102 mm per hour (Wintrobe hematocrit method). Urinalysis showed many white blood cells. Urine and blood cultures were obtained, and the patient was hospitalized for empiric treatment of a suspected urinary tract infection with IV cefotaxime (1 g every eight hours).
On the second day of hospitalization, the patient's temperature increased to 38.8° C (102° F), and the WBC increased to 26,000 per mm3 (26 × 109 per L). She became agitated and intermittently confused. A urine culture showed Escherichia coli that was sensitive to cefotaxime; a blood culture was negative. Because the patient's condition worsened while she was receiving the appropriate antibiotic therapy for a urinary tract infection, other sites of infection were considered.
A transesophageal echocardiogram was negative, and a magnetic resonance image (MRI) of the brain was unremarkable. MRI of the spine, with and without contrast, showed osteomyelitis at T12, and a paraspinal soft tissue fluid collection at T12-L1 with epidural extension from T12 through L2 and displacement of portions of the underlying thecal sac (Figures 1 through 4). The radiologist reported that the abnormal soft tissue fluid collection was probably infectious.
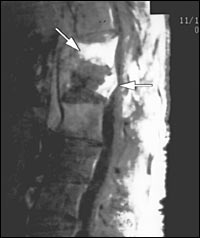
Computed tomography (CT)–guided needle aspiration of the paravertebral collection showed purulent material that grew Actinomyces odontolyticus after one week. A blood culture grew the organism after 10 days. The patient's antibiotic therapy was changed to IV penicillin G (20 million U per day in four divided doses for 10 weeks).
With resolution of the leukocytosis, the patient became afebrile, her depression appeared to improve, and the back pain was reduced significantly. She was discharged to a nursing home after 10 weeks of IV antibiotic therapy and continued to receive oral penicillin V (4 g per day in four divided doses) for six months. A repeat MRI of the spine showed resolution of the infection.
Discussion
In this patient, risk factors for spinal epidural abscess included age, diabetes mellitus, and vertebral trauma. A hematoma formation from the vertebral fracture may have become infected. It is likely that the source of actinomycosis bacteremia was periodontal disease because A. odontolyticus is the most common actinomyces present on the tongue.13 The decision to treat the patient medically was based on the higher surgical risk (because of age, comorbid condition, and uncertain duration of cord dysfunction) when compared to the expected benefit. Because the organism was isolated, nonsurgical treatment was preferred.
Pathology and Pathophysiology
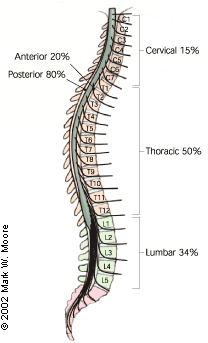
Most epidural abscesses are located posteriorly in the thoracic or lumbar spine (Figure 3). Most posterior spinal epidural abscesses are thought to originate from a distant focus such as a skin infection, pharyngitis, or dental abscess.8,9 Anterior epidural abscesses are commonly associated with discitis or vertebral osteomyelitis.1,2 These abscesses can also be caused by direct extension from retropharyngeal or retroperitoneal abscess14 through communication with intervertebral foramina. Blunt trauma is reported to precede the symptoms of spinal epidural abscess in 15 to 35 percent of cases,11 and it is postulated that trauma may result in the formation of a vertebral hematoma, which serves as a rich nutrient source for infection.
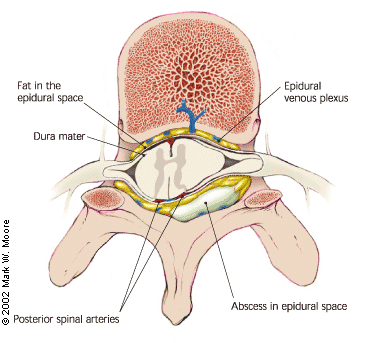
The mechanisms leading to neurologic deficit and destruction within the spinal cord, as well as the major complications of spinal epidural abscess, such as central nervous system dysfunction, are not completely understood. Although cord and nerve root compression from the extradural mass within the rigid spinal canal might appear to be the obvious explanation, neurologic dysfunction is often disproportionate to the observed degree of compression.11 Many authors have postulated that the edema and inflammation in the epidural space may involve the epidural venous plexus, which may compromise circulation and result in cord ischemia10,15 (Figure 4). A combination of compressive and ischemic effects may act in synergy to produce the disastrous sequelae of epidural abscess.
Diagnosis
The diagnosis of epidural abscess must be made promptly because delay in treatment can result in irreversible neurologic damage or death. The progression of neurologic dysfunction varies from a few hours to several months. The presentation of spinal epidural abscess can be nonspecific. Fever, malaise, and back pain are the most consistent early symptoms (Table 2). Local tenderness, with or without neurologic deficit, is the usual physical finding, and leukocytosis may be the only abnormal laboratory finding. Epidural abscess should be suspected in patients presenting with fever and back pain, especially if they have risk factors as listed in Table 1.
| Fever | Bladder/bowel dysfunction |
| Malaise | Plegia |
| Back pain | Sepsis/mental status change |
| Radiculopathy/paresis |
Once the diagnosis is suspected, neurosurgery consultation is recommended. A blood culture should be obtained (blood cultures are positive in more than 60 percent of patients) to confirm the diagnosis.10 Gadolinium-enhanced MRI is the imaging choice for the diagnosis of epidural abscess. It is useful in distinguishing epidural abscess from the adjacent compressed thecal sac and other potential compressive lesions, such as a herniated disc.
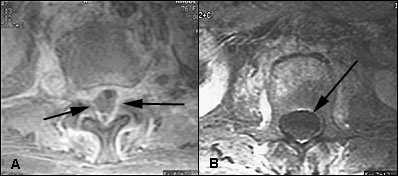
MRI reveals two basic patterns.16,17 First, the phlegmonous stage of infection results in homogenous enhancement of the abnormal area. Pathologically, this correlates to granulomatous-thickened tissue with embedded microabscesses without a significant pus collection. The second pattern is that of a liquid abscess surrounded by inflammatory tissue, which shows varying degrees of peripheral enhancement with gadolinium (Figure 5).
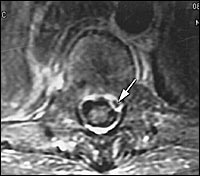
Once the abscess is diagnosed, a culture specimen can be obtained by CT-guided aspiration or open biopsy, so the causative organism can be determined. Lumbar puncture, once a routine diagnostic tool in patients with suspected spinal epidural abscess, has largely been replaced by MRI. If MRI is not available, CT with myelography can be performed.
Treatment
Surgical decompression remains the mainstay of treatment for spinal epidural abscess. Endoscopy-assisted surgery18 and percutaneous drainage19 are reported to be successful treatments. Nonsurgical treatment is indicated in patients who present with minimal neurologic deficit or are poor surgical candidates.2,20–22
If the patient is treated medically, serial MRIs and close neurologic examination are important so the physician can follow the progression of the disease.23 If neurologic deficit progresses or MRI shows an increase in the size of the abscess, prompt surgical intervention is indicated. There are no studies indicating whether patients should be treated surgically or medically because, if treatment is initiated expeditiously, both modalities show good outcomes.21,22
A recent study22 analyzed the predictive value of the symptoms and outcomes of patients with spinal epidural abscess. The duration of pain or radiculopathy location of abscess, and degree of granulation tissue do not appear to affect the outcome of the disease as long as the condition is treated in a timely manner. The authors of this study suggest that surgery provides a good outcome when patients' cord symptoms (bowel and bladder dysfunction, paresis, or “plegia”) are present for fewer than 72 hours, when extent of abscess (the degree of thecal sac compression) is less than 50 percent, and when patients are younger than 60 years of age (Table 3). However, these authors did not address possible outcomes in patients with the same criteria who were treated medically.21
| Factor | Good outcome | Poor outcome |
|---|---|---|
| Age | <60 years | >60 years |
| Degree of thecal sac compression | <50% | > 50% |
| Duration of cord symptoms | < 72 hours | > 72 hours> |
| Comorbid condition | None present | One or more |
Actinomyces and Treatment
Actinomyces are susceptible in vitro to a number of antimicrobial agents. The therapy of choice is IV penicillin G given in high doses for six to eight weeks. Oral treatment should be continued for at least an additional four to six months because A. odontolyticus infection easily recurs with premature termination of antibiotic therapy.
Final Comment
The incidence of spinal epidural abscess is increasing, and the most common causative agent is S. aureus. Actinomycosis should be suspected in patients with poor dentition and those who have had recent dental work. The most common presenting symptoms are back pain and fever.
Once epidural abscess is suspected, neurosurgery consultation is recommended. The diagnostic study of choice is MRI with gadolinium contrast. Once a diagnosis is made, empiric antibiotic therapy should be started. If a patient's neurologic deficit progresses or MRI shows an increase in the size of the abscess following medical treatment, emergency surgical decompression is indicated. Both surgery and medical treatment produce good outcomes when patients are treated early.