Oral cancers account for approximately 3 percent of all cases of cancer in the United States. An estimated 30,000 people will be diagnosed with oral cancer this year, and about one half of them will eventually die of the disease. The most common type of oral cancer is squamous cell carcinoma. Sixty percent of oral cancers are well advanced by the time they are detected, even though physicians and dentists frequently examine the oral cavity. The two most important risk factors for oral cancer are tobacco use and heavy alcohol consumption. The keys to reducing mortality are prevention and control. The earlier any intraoral or extraoral abnormalities or lesions are detected and biopsied, the more lives can be saved. Controversy exists whether screening programs effectively reduce the mortality rate. Specific step-by-step guidelines should be followed to perform an adequate examination of the head and neck.
Forty percent of head and neck cancers occur in the oral cavity, including the tongue, floor of the mouth, palate, lips and oropharynx. Oral cancer is the sixth most common cancer worldwide.1 The incidence rate of oral cancer in the United States is about 11 cases per 100,000 persons per year, and the incidence increases with age.2,3 Cancers of the head and neck region account for approximately 3 percent of cancers in men and less than 2 percent in women.4
Oral cancer is responsible for 2 percent of all cancer deaths in the United States.5 Sixty percent of these cancers are well advanced by the time they are detected. Therefore, cancers of the oral mucous membranes continue to cause considerable mortality and morbidity in the United States, with a five-year survival rate of about 30 percent.6 Lip cancer, while included in statistics for oral cancer, is more similar to skin cancers, with a survival rate of more than 90 percent.3
This article reviews the methods of assessing oral cancer and the controversy surrounding screening programs.
History
The first step in patient assessment is obtaining a thorough history including the presence of risk factors and symptoms. Most neoplastic diseases occur from an inherent tendency or enhanced genetic susceptibility combined with multiple exposures to substances that contribute to the initiation or promotion of carcinogenic change.7 The two major risk factors for oral cancer—tobacco use and heavy alcohol consumption—are responsible for about 75 percent of oral cancers. The remaining 25 percent are caused by other factors, which may include nutritional and genetic influences. The etiologic role of these factors is not well understood, and methods for modifying them need to be developed.
All forms of tobacco have been implicated as causative agents, including cigarette, cigar and pipe tobacco as well as chewing tobacco and snuff. Although oral cancers occur in patients who do not use tobacco, this constitute a very small percentage of cases.
Patterns of alcohol consumption, particularly among smokers, also are considered important. There is a higher incidence of oral cancer in people who are heavy smokers and heavy drinkers. The reason for the increased cancer risk associated with increasing alcohol consumption is not completely understood.8 It may be due to the carcinogenic effect of the first metabolite of ethanol, acetaldehyde.8,9 Women who drink heavily and smoke have a 100-fold increase in oral cancer, whereas men who are heavy drinkers and smokers have a 38-fold increase.10 Sun exposure is the primary risk factor for lip cancer, but pipe smoking also is a risk factor.
Although early oral cancers are usually asymptomatic, progression of the cancer may be accompanied by difficulty in chewing or swallowing, a history of sores in the mouth that do not disappear, hoarseness, soreness or a sensation of something in the throat and difficulty with speech.
Physical Examination
The principal screening test for oral cancer is a physical examination that consists of systematic inspection and palpation.2 The extraoral soft tissues are examined first, followed by the intraoral soft tissues. Any deviations, such as swelling, tenderness or fluctuance, are recorded.
During the extraoral examination, the lips, salivary glands and cervical lymph nodes are evaluated. The closed lips are inspected for texture and color. The vermilion border should be observed and palpated for ulceration, blistering, induration and swelling.
The parotid gland, the largest of the salivary glands, lies in front of the ear and wraps around the posterior border of the mandible. The parotid gland is palpated intraorally and extraorally, either bimanually (placing one hand on the cheek and two fingertips of the other hand on the buccal mucosa) or bidigitally (placing the gland between the fingertips of one hand).
The submandibular and sublingual glands are palpated by either pressing four fingertips bilaterally on the soft tissue under the chin (Figure 1a) or resting the thumbs of each hand near the inferior mandibular border while pressing the fingertips inferior and medial to the mandibular border (Figure 1b). The finger-tips are moved inferiorly to the hyoid bone and then medially and superiorly until the inferior part of the submandibular gland is felt.
FIGURE 1A.
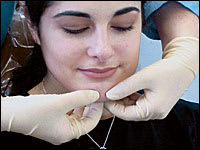
Palpation of the submandibular and sublingual glands.
FIGURE 1B.
Palpation of the submandibular and sublingual glands.
When oral cancer metastasizes, it most commonly spreads through the lymphatic system to the cervical chain of lymph nodes in the neck. Spread to the cervical lymph nodes is more common in oropharyngeal neoplasms than in oral cavity neoplasms. Thus, the status of the cervical lymph nodes, especially the upper deep nodes, at presentation is important.
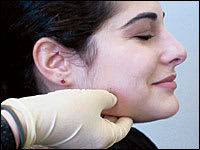
The anterior deep cervical lymph nodes are palpated with the patient's head hyperextended and turned to distend the sternocleidomastoid muscle (Figure 2). Using two hands, the fingertips of one gently retracts the sternocleidomastoid backward while the fingertips of the other hand, hooked around the front of the neck, palpate the region of the carotid sheath. This examination also can be performed with one-hand palpation by placing aligned fingertips along the posterior border of the sternocleidomastoid while the thumb provides counter pressure from the anterior aspect of the muscle. The fingertips are gradually moved inferiorly along the muscle.
FIGURE 2.
Examination of the cervical lymph nodes.
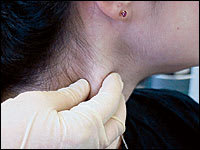
The preauricular and posterior auricular lymph nodes are examined next, using bilateral placement of both hands (Figure 3). The preauricular lymph nodes are located in front of the ear and the postauricular nodes are located behind the ear. The supraclavicular nodes are located on the inferior part of the front of neck, superior to the clavicle. The occipital lymph nodes are located at the base of the skull. The auricular lymph nodes are palpated by the bilateral placement of both hands on the skin surface with the fingertips arranged to cover a large surface area.
FIGURE 3.
Examination of the preauricular lymph nodes.
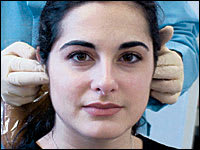
Examination of the intraoral tissues includes inspection and palpation of the lips, buccal mucosa, tongue, floor of the mouth, gingiva and palate. The intraoral labial mucosa is inspected and then palpated using the fingertips of two hands to invert the upper and lower lips (Figure 4). The buccal mucosa is retracted for inspection and palpated using a bimanual or bidigital technique.
FIGURE 4.
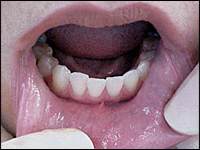
Visualization of the intraoral labial mucosa of the lips.
The lateral borders of the tongue are common areas for oral malignancies to develop. The borders should be inspected for ulceration, inflammation, swelling or white patches. The tongue is inspected by grasping the tip with a gauze square and pulling out and moving it to the sides and upward to permit complete visualization of the dorsal lateral borders and ventral surfaces (Figure 5a). At the same time, palpation using the index finger of the other hand is performed (Figure 5b).
FIGURE 5A.
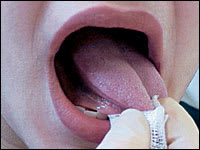
Visualization of the dorsal surface and lateral borders of the tongue.
FIGURE 5B.
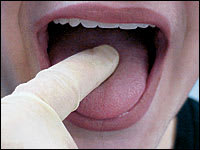
Palpation of the dorsum of the tongue.
Next, the floor of the mouth is inspected and palpated. The submandibular glands located in the anterolateral floor of the mouth are palpated by placing the fingertips of one hand in the floor of the mouth while the fingertips of the other hand are placed under the chin to support the mandible. The sublingual glands are more difficult to palpate than the submandibular glands because they are more compressible and less distinct.
The hard palate is another site of oral malignancies. Smokers, especially pipe smokers, may develop nicotinic stomatitis on the hard posterior palate, which makes this a high-risk area. With the mouth wide open and the patient's head tilted back, the hard and soft palates should be carefully inspected and palpated.
Screening
The U.S. Preventive Services Task Force (USPSTF) states that insufficient evidence exists to recommend for or against routine screening (secondary prevention) for oral cancer in asymptomatic patients.2 However, the American Cancer Society (ACS) and the National Cancer Institute (NCI) strongly emphasize that oral examinations could prevent many deaths.11 According to the USPSTF, little information is available on the sensitivity of screening in detecting oral cancer or the frequency of false-positive results when a lesion is found.2 Additionally, there is no evidence for the efficacy of screening in reducing the incidence of oral cancer and mortality from the disease.12 Sufficient evidence concerning the efficacy is needed to justify universal screening programs. For patients at high risk (e.g., smokers and heavy alcohol users), examinations by a physician or dentist should be considered. All high-risk patients should be counseled to discontinue the use of all forms of tobacco and to limit their consumption of alcohol.
Any small (less than 1 cm), suspicious lesions can be prophylactically removed by a family physician, an otolaryngologist or an oral surgeon. Any lesions that persist longer than two weeks should be biopsied for a definitive diagnosis.
Prevention
Although the role of secondary prevention is controversial, primary prevention through avoiding or limiting tobacco and alcohol use is strongly recommended. It cannot be overemphasized that elimination of all tobacco use is the single most important way to reduce the incidence of oral cancer. Former smokers are only half as likely to develop oral cancer as those who continue to smoke.13 If alcohol is used, it should be consumed only in moderation.
Oral Cavity Neoplasms
Because the oral cavity is composed of many types of tissue, different types of benign or malignant neoplasms can occur in this site. The more common premalignant and malignant lesions of the oral cavity are reviewed in the following sections.
LEUKOPLAKIA
Leukoplakia has been labeled the most common “precancerous” lesion of the oral cavity. Leukoplakia is defined as a white plaque-like lesion of the oral mucous membrane that cannot be rubbed off and cannot be diagnosed clinically or pathologically as any specific disease. It is strictly a clinical term and does not designate a histologic appearance.
Although leukoplakia has been overemphasized in its relationship to cancer, the cause of any white lesion should be determined and the lesion monitored. Most leukoplakias are caused by hyperkeratosis or a thickening of the outer keratin layer. There is little scientific evidence that physical irritation plays a role in the development of oral cancer.14 The more common sites of involvement are the buccal mucosa, floor of the mouth, ventral or lateral border of the tongue, and lower lip. Leukoplakia involving the tongue or the floor of the mouth tends to be premalignant or malignant.
Removal of the associated risk factors—tobacco and alcohol use—is of utmost importance. If the lesion does not disappear after about two weeks, a biopsy must be performed.
ERYTHROPLAKIA
Erythroplakia is the red counterpart to leukoplakia. The term refers to a red patch of tissue that cannot be diagnosed clinically or pathologically as a specific disease or condition15 and is not associated with inflammation. Clinically, erythroplakia may appear as a red, velvet-like lesion or may have white patches on or peripheral to the lesion (speckled leukoplakia), which has a high risk of carcinoma. The boundaries of these lesions may be poorly defined; many are irregular, with a blending of inflamed and normal mucosa.
Erythroplakia is less common than leukoplakia; however, it is more frequently associated with epithelial dysplasia or squamous cell carcinoma. Most suspicious erythroplakias are found in patients considered to be at high risk for oral cancer, such as heavy smokers and drinkers 40 years and older. Approximately 90 percent of early carcinomas are predominantly red or have a red component. In high-risk patients, the red, velvety lesion appears long before any ulceration, bleeding, induration, dysfunction, pain or lymphadenopathy arises.
Biopsy is mandatory for any persistent red lesion without an obvious cause. Treatment depends on the results of a histologic examination. Avoidance of tobacco and alcohol is of utmost importance.
EPITHELIAL NEOPLASMS
Squamous cell carcinoma is a malignant neoplasm of squamous epithelial cells and is the most common primary malignancy of the oral cavity. The more common sites of involvement are the lateral border of the tongue, lower vermilion border of the lip, floor of the mouth and alveolar ridge (Figure 6).
FIGURE 6.
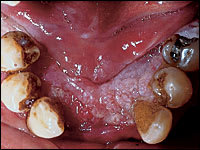
Squamous cell carcinoma of the alveolar ridge.
Clinically, squamous cell carcinoma starts as a nonhealing, painless, red ulceration with white rolled borders. Early lesions may be erythroplakia, leukoplakia or speckled leukoplakia. If leukoplakia is left untreated, it can transform to squamous cell carcinoma. Growth progresses very rapidly. As with all oral cancers, it spreads from the oral cavity to the submandibular and cervical lymph nodes.
SALIVARY GLAND NEOPLASMS
Neoplasms of the salivary gland have different risk factors and are relatively rare. Neoplasms can occur in the parotid, sublingual or submandibular glands, or in the minor salivary glands. Intraorally, the posterior hard palate (lateral to the midline) is the most common site of involvement. These neoplasms also can occur on the buccal mucosa and lips. Growth is rapid. Biopsy is required to establish a diagnosis.
Adenoid cystic carcinoma and mucoepidermoid carcinoma are the two most common intraoral malignant neoplasms of the accessory salivary glands. Mucoepidermoid carcinoma occurs most frequently in persons between 20 and 50 years of age; adenoid cystic carcinoma usually occurs after age 50. Clinically, these carcinomas appear as painless masses or nodules.
Pleomorphic adenoma is a benign neoplasm that occurs in major or minor salivary glands. The palate is the most common intraoral site when accessory salivary glands are involved. Clinically, this neoplasm appears as a slow-growing, nonulcerated, painless mass. It occurs most frequently in women between 30 and 60 years of age. Excisional biopsy is recommended, although there is a high rate of recurrence.
Final Comments
Currently, it is unclear whether screening patients for oral cancer will achieve a reduction in mortality. However, this does not preclude family physicians and dentists from performing frequent extraoral and intraoral clinical examinations. Suspicious lesions should be biopsied by the family physician or referred to a dental specialist (e.g., oral surgeon or oral pathologist) or an otolaryngologist. The most important way to reduce the incidence of oral cancer is to minimize or eliminate the use of tobacco and alcohol.

