
Am Fam Physician. 2002;65(9):1817-1823
A more recent article on adolescent idiopathic scoliosis is available.
Adolescent onset of severe idiopathic scoliosis has traditionally been evaluated using standing posteroanterior radiographs of the full spine to assess lateral curvature with the Cobb method. The most tilted vertebral bodies above and below the apex of the spinal curve are used to create intersecting lines that give the curve degree. This definition is controversial, and patients do not exhibit clinically significant respiratory symptoms with idiopathic scoliosis until their curves are 60 to 100 degrees. There is no difference in the prevalence of back pain or mortality between patients with untreated adolescent idiopathic scoliosis and the general population. Therefore, many patients referred to physicians for evaluation of scoliosis do not need radiographic evaluation, back examinations, or treatment. Consensus recommendations for population screening, evaluation, and treatment of this disorder by medical organizations vary widely. Recent studies cast doubt on the clinical value of school-based screening programs.
Adolescent idiopathic scoliosis is lateral and rotational spinal curvature in the absence of associated congenital or neurologic abnormalities. Longitudinal studies1,2 estimate the prevalence of idiopathic scoliosis as 2 percent of the adolescent population, using a definition of a spinal curve greater than 10 degrees.3 However, clinically significant curves in the range of 40 to 100 degrees are rare. Controversy surrounds clinical recommendations for evaluating and managing patients with a wide range of curve sizes. Recent debate has centered on the value of school-based screening programs.4
School-Based Screening
School-based scoliosis screening programs are currently mandated in 26 states, with many other states having voluntary programs. Several studies1,5,6 and the 1996 U.S. Preventive Services Task Force (USPSTF) Report7 question the value and cost-effectiveness of these programs. Few children referred for medical evaluation from these programs receive any form of therapy.8 Furthermore, the long-term health outcomes for treated versus untreated patients with scoliosis have not been well studied, making it difficult to determine the health impact of these screening programs. Patients with severe curves are not difficult to diagnose. Although some advocates still recommend school-based screening of adolescents, there is no evidence to support these programs. The American Academy of Pediatrics recommends screening at physician visits. The USPSTF and the Canadian Task Force on the Periodic Health Examination state that insufficient evidence exists to support universal school-based screening. Radiographic decision-making skills will help primary care physicians evaluate severe scoliosis accurately.
Etiology
The etiology of adolescent idiopathic scoliosis is believed to be multifactorial, including genetic factors. One study9 showed a 73 percent phenotypic concordance of scoliosis in monozygotic twins. Eleven percent of first-degree relatives of patients with scoliosis are affected. Inheritance of scoliosis varies, and no single pattern of genetic transmission has been accepted. The physiologic causes of scoliosis have not been elucidated. Muscular, nervous system, hormonal, and connective tissue defects have been noted in subgroups of patients with scoliosis, but these abnormalities may be a result of the disorder rather than a cause.10
Natural History
Studies11–13 of adolescent-onset scoliosis have demonstrated that patients with scoliosis show minimal progression in the magnitude of the curve in adulthood if the curve is less than 30 degrees at skeletal maturity. Although curves in different regions of the spine progress differently, curves measuring 40 to 50 degrees at skeletal maturity progress an average of 10 to 15 degrees during a normal lifetime, while curves measuring greater than 50 degrees at skeletal maturity progress at a rate of approximately 1 to 2 degrees per year.11 Because of the controversy surrounding ideal treatment strategies for patients with moderate curves, estimation of curve progression can aid in clinical management and counseling patients' families about prognoses.
Estimation of skeletal maturity can be determined by assessing the epiphyseal status on wrist radiographs, the Risser sign, Tanner stages, progressive sitting and standing height measurements, and age at menarche.
Risser sign is defined by the amount of calcification present in the iliac apophysis and measures the progressive ossification from anterolaterally to posteromedially. A Risser grade of 1 signifies up to 25 percent ossification of the iliac apophysis, proceeding to grade 4, which signifies 100 percent ossification (Figure 1). A Risser grade of 5 means the iliac apophysis has fused to the iliac crest after 100 percent ossification. Children usually progress from a Risser grade 1 to a grade 5 over a two-year period. One study8 found that immature patients (Risser grades 0 and 1) with a spinal curvature measuring 20 to 29 degrees had a 68 percent probability of progression of 6 degrees or more during remaining growth. Patients closer to maturity (Risser grades 2 to 4) and with the same degree of scoliosis had a 23 percent probability of progression.8 Curves measuring 5 to 19 degrees in immature patients had a 22 percent probability of progression, while small curves in mature patients had only a 1.6 percent probability of progression.8
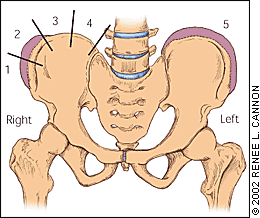
If other clinical markers of maturity such as Tanner staging or age at menarche are not consistent with the Risser grade, curve progression may proceed at a different rate. Thus, multiple measures of maturity are important to the clinical assessment of these patients.
Although a recent study14 showed an increased incidence of precocious puberty, the average female reaches a Tanner stage 1 at 11 years of age, the beginning of the growth spurt at 11.5 years of age, a Risser grade 1 at 12 years of age, and has an onset of menarche between 12 and 13 years of age. A female patient who matures consistent with these averages will have a relatively higher risk of curve progression before 12 years of age and a relatively lower risk of curve progression after 12.5 years of age.15
A scoliotic curve is more likely to progress in females, and thoracic curves or curves with a higher apex vertebral level are more likely to progress than other types of curves.8,14 Severe curves or moderate curves expected to progress beyond 100 degrees can lead to restrictive pulmonary disease and a possible reduction of life expectancy.11 Curves of this magnitude usually have an infantile or juvenile onset rather than an adolescent onset.
Studies16,17 show an equal incidence of back pain and mortality in the general population and patients with adolescent idiopathic scoliosis. There are few recent studies18–20 that evaluate the long-term cosmetic and psychosocial consequences of progressive spinal curves from the perspective of patients with scoliosis.
Clinical Presentation and Evaluation
Family physicians may need to examine children for scoliosis who have been referred from school-based screening programs. Because diastematomyelia (congenital splitting of the spinal cord), syringomyelia (cavity in the spinal cord), a tethered cord, or a spinal tumor can cause spinal curvature, physicians should ask the patient questions concerning neurologic symptoms. Neurofibromatosis can be associated with scoliosis, and a unilateral cavus foot can be a manifestation of intraspinal pathology.
Magnetic resonance imaging should be obtained in patients with an onset of scoliosis before eight years of age, rapid curve progression of more than 1 degree per month, an unusual curve pattern such as left thoracic curve, neurologic deficit, or pain. Spine abnormalities may present during routine examinations of 10- to 19-year-olds. Many examination techniques are used to evaluate patients presenting with spinal curvatures. Traditionally, the Adam's test with level plane and ruler or a scoliometer evaluation of the patient while bending forward was used to guide clinical decision-making.19 These measurements are difficult to standardize and should only be obtained when they will affect management decisions for an individual patient or to reassure the patient and family.6
Height measurements of the patient sitting and standing should be taken in the physician's office every three to four months. Documenting rapid height increases helps the physician determine the onset of the adolescent growth spurt and gauge the risk of rapid progression of the spinal curve. Sitting heights can be measured with the patient sitting in a standard chair and the height of the seat subtracted from the total height. Changes in sitting height can be less than changes in standing height and give a better estimate of truncal growth rate.
Any examination data must be combined with a thorough history to assess skeletal and sexual maturation.21
If an examination of the back is conducted, the physician should begin with a survey of the back while the patient is standing. Physicians may be misled by scapular or shoulder asymmetry and should focus on waist crease asymmetry or spine deviation during the upright examination. When measuring waist crease asymmetry, subtract perpendicular height from the iliac crests on each side. Radiographs should only be considered when a patient has a curve that might require treatment or could progress to a stage requiring treatment (usually 40 to 100 degrees).22
Radiologic Evaluation and Classification
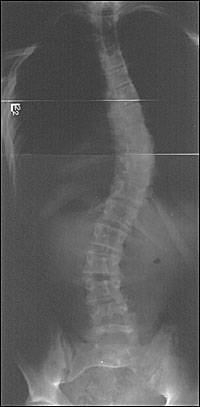
The standard radiologic evaluation of adolescent idiopathic scoliosis consists of standing posteroanterior radiographs of the full spine. Follow-up is necessary in those patients with severe curves who are at risk for significant curve progression or require some form of treatment. Any discrepancy in leg length should be corrected with a block placed under the patient's shorter leg when radiographs are taken. One study23 has shown that long-term management of scoliosis poses no radiograph-related risks to patients, but posteroanterior views assure maximal safety by minimizing radiation to the breasts. The Cobb method is used to measure the degree of scoliosis on the posteroanterior radiograph (Figure 2). In addition to curve degree, physicians should describe curves as “right” or “left,” based on their curve convexity. Standard measurement error is 3 to 5 degrees for the same observer and 5 to 7 degrees for different observers when the same end vertebrae are used for measurements.24,25 Thus, physicians should use the same end vertebrae for subsequent measurements and assume that some measurement change may be caused by error rather than true curve progression.
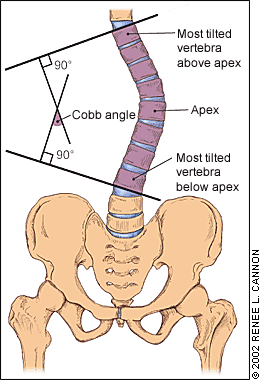
Posteroanterior radiographs should be viewed in reverse to normal chest radiographs with the patient's right side on the physician's right side. Curves are named for the location of the apex vertebrae, and may be described as thoracic (Figure 3), lumbar, thoracolumbar, cervical, or double major (two curves in different spinal regions). A thoracolumbar curve (Figure 4) has an apex vertebrae at T12 or L1. Thoracic and lumbar curves have apex vertebrae in the middle of the thoracic and lumbar regions, respectively. A double curve (Figure 5) has a major and a minor curve (based on size and flexibility) and a primary and secondary curve (based on respective development). A compensatory curve is nonstructural and develops to balance out a primary curve. A nonstructural curve differs from a structural curve because it can correct on lateral bending, distraction, or sitting.
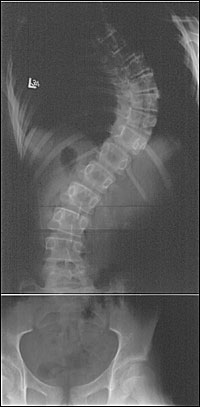
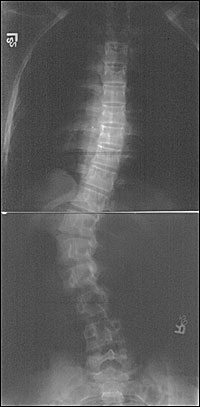
Management and Follow-up
The primary goal of treating adolescent idiopathic scoliosis is preventing progression of the curve magnitude. Curves less than 10 to 15 degrees require no active treatment and can be monitored, unless the patient's bones are very immature and progression is likely. Moderate curves between 25 and 45 degrees in patients lacking skeletal maturity used to be treated with bracing, but this treatment has never been proven to prevent curve progression. Poor compliance with wearing a brace obviates any potential usefulness of the therapy.26 Much controversy surrounds brace indications, and trends over the past 20 years have moved toward no bracing or bracing only the more significant curves (20 to 50 degrees). Evidence27–29 showing the low symptomatic burden of patients with curves less than 60 degrees has influenced this trend away from treatment with bracing.
Most patients with adolescent idiopathic scoliosis who require treatment with a brace may use a thoracolumbar-sacral orthosis (TLSO) or a cervicothoracolumbar-sacral orthosis (CTLSO). Recommendations for optimal use of braces vary from eight to 24 hours a day depending on the style of brace chosen.
In patients with a curve severe enough to require surgery (greater than 45 degrees in adolescents and greater than 50 degrees in adults), rod placement and bone grafting may be necessary to achieve partial or complete correction. Patient preference is essential in deciding on a surgical treatment, and primary care physicians should work closely with patients and their families to reach optimal individual outcomes.