Medical and surgical options for the treatment of benign prostatic hyperplasia have expanded in recent years. Saw palmetto, the most widely used complementary medication, is less effective than standard medical therapy but has fewer side effects. Although nonselective alpha blockers provide rapid relief of symptoms and are relatively inexpensive, they can cause dizziness and orthostatic hypotension. These effects occur less often with tamsulosin, a more selective alpha blocker. Finasteride, a 5α-reductase inhibitor, slowly reduces prostatic volume but is not as effective as alpha blockers, especially in men with a smaller prostate. Dutasteride, a new 5α-reductase inhibitor, has recently been labeled for the treatment of benign prostatic hyperplasia. Surgery may be appropriate initial treatment in patients with severe symptoms who are not at high risk for complications. Surgery may also be indicated in patients who have failed medical therapy or have recurrent infection, hematuria, or renal insufficiency. Transurethral resection of the prostate is effective in most patients, but it carries some risk of sexual dysfunction, incontinence, and bleeding. Surgical procedures that use thermal microwave or laser energy to reduce hyperplastic prostate tissue have recently become available. In general, the newer procedures are less expensive than transurethral resection of the prostate and have fewer complications; however, the need for retreatment is somewhat greater with these less invasive techniques.
Because of the aging of the U.S. population in general and the “baby boomers” in particular, the prevalence of benign prostatic hyperplasia (BPH) is increasing. Recent studies have shown that more than 80 percent of men older than 80 years have BPH.1,2 When symptoms of urinary obstruction interfere with quality of life, treatment is warranted.
Diagnosis
In 1994, the Agency for Health Care Policy and Research (AHCPR; now the Agency for Healthcare Research and Quality)1 published a carefully researched clinical practice guideline on BPH. The diagnosis of this condition is presumptive based on the patient's symptoms. These symptoms include urinary frequency, nocturia, urgency, hesitancy, weak or intermittent urine stream, straining to void, and sensation of incomplete voiding. It is common for an enlarged prostate to be found on rectal examination, but enlargement is not necessary for the diagnosis of BPH, in that palpable prostate size does not correlate with the degree of obstruction or the severity of symptoms.
Although the symptoms of BPH vary from patient to patient and can be difficult to grade, they are key to the diagnosis and treatment of this condition. Instead of using traditional BPH terminology, some physicians may refer to these symptoms as lower urinary tract symptoms.
To grade urinary symptoms, most physicians use the urinary symptom scoring system developed by the American Urological Association (AUA).1,3 This system allows patients to rate various urinary symptoms on a scale of zero to five. The total point score is then matched to the point range for mild, moderate, or severe BPH (Figure 1).3
FIGURE 1. BPH Symptom Score
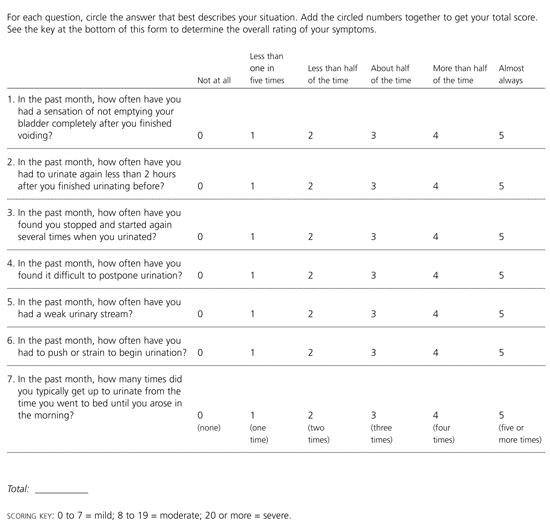
Scoring system developed by the American Urological Association to evaluate the severity of urinary symptoms. (BPH = benign prostatic hyperplasia)
Adapted with permission from Barry MJ, Fowler FJ Jr, O'Leary MP, Bruskewitz RC, Holtgrewe HL, Mebust WK, et al. The American Urological Association symptom index for benign prostatic hyperplasia. The Measurement Committee of the American Urological Association. J Urol 1992;148:1549–57.
The initial evaluation should include a detailed medical history, with particular attention given to the urinary tract and comorbid conditions that may influence or complicate treatment. Uncontrolled diabetes mellitus, urinary tract infection, neurogenic bladder, urethral strictures, bladder cancer, and congestive heart failure may cause symptoms similar to those in BPH. Medications that can increase obstructive urinary symptoms include tricyclic antidepressants and other anticholinergic agents, diuretics, narcotics, and first-generation antihistamines and decongestants (common cold medications).
The physical examination should include a digital rectal examination to check for palpable nodules, induration, or irregularities associated with malignancy or infection. An irregularity that is suspicious for cancer requires a discussion of the patient's preferences for further investigation.4
All patients with suspected BPH should undergo dipstick urinalysis testing or microscopic urinalysis to screen for infection or hematuria, and a serum creatinine determination to evaluate renal function. In addition, prostate-specific antigen (PSA) testing may be offered to patients at risk for prostate cancer who prefer to be screened for the malignancy. The AUA5 and the American Cancer Society6 recommend that PSA testing and rectal examination be offered annually to men 50 years of age and older if they are expected to live at least 10 more years. Black men and men who have a first-degree relative with prostate cancer are at high risk for prostate cancer. These men should be offered screening at 45 years of age.
Optional tests include urinary flow rate measurements, postvoid residual urine measurements, and pressure flow studies. Ultrasonography, intravenous pyelography, filling cystometrography, and cystoscopy are not routinely recommended in the evaluation of suspected BPH.
Treatment Options
WATCHFUL WAITING
Watchful waiting is appropriate in patients with a low AUA symptom score (zero to seven) because studies have shown that medications are not significantly more effective than placebo in these patients.1 However, follow-up monitoring is important, because spontaneous exacerbations and remissions of BPH (even without treatment) can occur. Patients with higher AUA symptom scores should be given information on appropriate treatment options.1
COMPLEMENTARY MEDICINE
Interest in alternative treatments for BPH increased after epidemiologic studies showed a lower incidence of BPH and prostate cancer in Asians compared with persons from Western countries. One postulated explanation is the higher soy content of the typical Asian diet. Genistein, a major isoflavone ingredient of tofu, has been found to decrease the growth of hyperplastic prostate tissue in histoculture.7
Because natural soy food products are not readily available or accepted worldwide, a standardized isoflavone product containing genistein has been marketed as a nonprescription tablet (Trinovin). This product is derived from red clover, not soy. Initial short-term company-sponsored studies8 showed rapid relief of BPH symptoms within one month and no adverse events when this product was given in a dosage of 40 or 80 mg per day. However, long-term and independent studies are not available.
Saw palmetto (Serenoa repens) is a popular complementary treatment for BPH. Physicians may want to become familiar with this treatment even if they do not recommend it. The mechanism of action is uncertain. Although it has been shown to inhibit the enzyme 5α-reductase in some in vitro studies, this has not been confirmed clinically.9 In patients with BPH, saw palmetto has been shown to be as effective as finasteride (Proscar) but not as effective as other medical treatments.9 In an analysis of 18 studies,10 saw palmetto had fewer side effects than traditional medications, and serial ultrasound examinations showed that treatment with this medicinal herb decreased prostate size without changing serum PSA levels. The usual dosage of saw palmetto is 160 mg twice daily. Side effects are rare (incidence of less than 3 percent) and usually consist of mild headaches or gastrointestinal upset.
Throughout the world, other herbal or complementary medicines are used to treat BPH. However, many of these medicines are not standardized or have not been well studied for efficacy. Commonly used agents include African plum, South African star grass, stinging nettle, and rye pollen.7 A rye grass pollen extract (Cernilton) has recently received attention. In short trials,11 this compound produced mild improvement of BPH symptoms, but further studies are needed.
MEDICAL TREATMENTS
Nonselective Alpha Blockers
Doxazosin (Cardura), prazosin (Minipress), and terazosin (Hytrin) reduce prostatic smooth muscle tone and, thus, have an immediate effect on urinary flow. Although these medications quickly improve BPH symptoms, AUA symptom scores improve less than with surgery.1
Side effects such as dizziness, postural hypotension, fatigue, and asthenia affect from 7 to 9 percent of patients treated with nonselective alpha blockers. Side effects can be minimized by bedtime administration and slow titration of the dosage. Alpha blockers can be used with other therapies as needed. Prazosin has the cost advantage of generic availability; however, unlike doxazosin and terazosin, it is not available in a once-daily formulation.
Selective Alpha Blocker
Tamsulosin (Flomax) is a highly selective alpha1A-adrenergic antagonist that was developed to avoid the side effects of nonselective agents. Some patients who do not respond to nonselective alpha blockers may respond to tamsulosin and, because of the selectivity, may have fewer side effects, including hypotension. Tamsulosin is initiated in a dosage of 0.4 mg once daily, with a maximum dosage of 0.8 mg per day.12 Tamsulosin has no antihypertensive effect and is more expensive than nonselective alpha blockers13 (Table 1).
5α -Reductase Inhibitors
Finasteride slowly induces an 80 to 90 percent reduction in the serum dihydrotestosterone level. As a result, prostatic volume decreases by about 20 percent over three to six months of treatment.
Although an early study showed similar efficacy for finasteride and placebo, the larger Proscar Safety Plus Efficacy Canadian Two Year Study (PROSPECT)14 found that treatment with finasteride led to significant improvements in urinary symptoms and flow rates. However, in the PROSPECT study, the improvements with finasteride were significantly less than those with any alpha blocker or surgery.1 Analyzed together, the results of multiple studies suggest that finasteride may work best in men with a large gland, whereas alpha blockers are effective across the range of prostate sizes.15
The incidence of side effects with finasteride is similar to that with placebo (4 to 5 percent). Adverse effects include decreased libido, ejaculatory disorder, and impotence.14
Finasteride decreases PSA levels by 40 to 50 percent. In a patient taking finasteride who has PSA screening, PSA levels should be doubled and then compared in the usual fashion to age-related norms. There is no change in sensitivity or specificity for the diagnosis of prostate cancer.16
The U.S. Food and Drug Administration recently labeled dutasteride (Duagen), in 0.5-mg capsule form, for the treatment of BPH (labeling for male-pattern baldness is pending). This drug has a distinct mechanism of action, in that it blocks both types 1 and 2 5α-reductase.17 Sexual side effects are similar to those of finasteride.
SURGICAL TREATMENTS
Surgery should be considered in patients who fail medical treatment, have refractory urinary retention, fail catheter removal, or have recurrent urinary tract infections, persistent hematuria, bladder stones, or renal insufficiency.1 Surgery can also be the initial treatment in patients with high AUA symptom scores who want surgical treatment and are good candidates for surgery.
Open Prostatectomy
Surgical removal of the inner portion of the prostate using a suprapubic or retropubic approach is the oldest and most effective treatment for relieving the symptoms of BPH and increasing maximum urinary flow. Symptomatic improvement occurs in 98 percent of patients who undergo this procedure, and the retreatment rate is only 2 percent. However, open prostatectomy is the most invasive treatment for BPH and is associated with the most morbidity. Therefore, this procedure is typically reserved for use in patients with a very large prostate gland or structural problems such as a large median lobe that protrudes into the bladder or a large bladder calculus or urethral diverticulum.18
Transurethral Resection of the Prostate (TURP)
The most commonly employed surgical procedure for BPH, TURP reduces symptoms in 88 percent of patients. The most frequent complications of the procedure are inability to void, clot retention, and secondary infection. Bleeding, the most morbid complication, occurs in only 1 percent of patients. Long-term complications include retrograde ejaculation (70 percent of treated patients), impotence (14 percent; range: 3 to 32 percent), partial incontinence (6 percent), and total incontinence (1 percent).1,19 Approximately 10 percent of patients require retreatment within five years.1
Newer Procedures
Many elderly patients with BPH are poor surgical candidates, and many younger patients find the risk of sexual dysfunction associated with TURP to be unacceptable. “Minimally invasive” surgical techniques have been developed in an effort to obtain the same results as TURP, but at a lower cost and with less morbidity. The availability and use of these newer procedures vary regionally.
Transurethral incision of the prostate (TUIP) is an endoscopic procedure using only one or two incisions to reduce constriction of the urethra without removing any of the prostate gland. TUIP can be performed as an outpatient procedure. It is generally offered as a treatment option for younger patients in whom fertility and antegrade ejaculation are important issues. Outcomes are similar to those with TURP, but the procedure takes less time to perform and causes less bleeding. Long-term satisfaction and retreatment rates have not been adequately studied.
Transurethral microwave thermotherapy (TUMT) is a single-session, minimally invasive outpatient treatment in which a microwave antenna is placed in a urethral catheter. Microwave energy causes deep, rapid tissue heating, while a cooling system circulates water to protect adjacent tissue. General or spinal anesthesia is not needed, and the procedure takes about one hour. Limited studies have shown a 65 percent decrease in subjective urinary symptoms and a 45 percent improvement in objective flow rates. Two years after undergoing TUMT, 7.3 percent of patients required retreatment.20
No major complications, including incontinence and sexual dysfunction, have been reported in patients treated with TUMT. When temperatures higher than 60°C (140°F) were used, temporary urinary retention secondary to swelling of the prostate occurred in 40 percent of patients.21
Another minimally invasive procedure is ablation of the prostate by electrode or laser. Some early studies showed that this procedure was associated with an increase in methicillin-resistant Staphylococcus aureus infections, possibly because all necrotic tissue was not removed or tissue coagulation was overzealous.22 Advances in electrodes, lasers, and training have decreased the incidence of this complication. Transurethral vaporization of the prostate (TUVP) or transurethral electrovaporization of the prostate (TVP) is now performed using endoscopic electrosurgical equipment to remove prostatic tissue with limited coagulation. The procedure provides urinary symptom reduction similar to that of TURP, with less postoperative irritation, urinary retention, blood loss, or risk of hyponatremia. As with TUMT, no tissue is obtained; thus, it is not possible to assess patients for concurrent prostatic malignancy.
Transurethral needle ablation of the prostate (TUNA) involves the placement of radiofrequency needles in the prostate. The procedure is safe and can be performed using local anesthesia. However, results may be limited, because the bladder neck and median prostate lobe cannot be treated. Initial studies showed that AUA symptom scores decreased from 21 to 7 and maximum urinary flow rates increased from 10 to 15 mL per second in patients who underwent TUNA. The 14 percent treatment failure rate at two-year follow-up is similar to that for TURP.23
In the past, transurethral balloon dilation of the prostate was used as a safe, minimally invasive treatment for BPH. However, the retreatment rate was high. Urethral stents can be placed to maximize the success of the dilation, but the stents are susceptible to infection or encrustation and reblockage. Because of the high complication rate, stent placement is usually reserved for use in patients who are high-risk surgical candidates with a short life expectancy.24,25
Final Comments
To date, no large-scale studies have compared the results of the many treatments for BPH. Treatment costs vary widely (Tables 1 and 226), and the availability of less invasive surgical options also varies considerably. Although surgery tends to be more expensive than medical treatment, it is often more effective.27 In a 1997 Gallup survey,28 only 3 percent of urologists reported performing TUMT and TUNA, whereas 95 percent performed TURP. However, a more recent Canadian study29 showed increasing use of the less invasive surgical procedures.
TABLE 1 Medical Therapy for Benign Prostatic Hyperplasia
| Drug | Tablet strengths (mg) | Cost (generic)* |
|---|---|---|
| Doxazosin (Cardura) | 2, 4, 6, 8 | $32 (28) |
| Prazosin (Minipress) | 1, 2, 5 | 15 (8 to 9) |
| Terazosin (Hytrin) | 1, 2, 5, 10 | 59 (48) |
| Tamsulosin (Flomax) | 0.4, 0.8 | 50 |
| Finasteride (Proscar) | 5 | 73 |
*— Estimated cost to the pharmacist, rounded to the nearest dollar, for 30 days of treatment using the lowest given dosage, based on average wholesale prices in Red book. Montvale, N.J.: Medical Economics Data, 2002. Cost to the patient will be higher, depending on prescription filling fee.
TABLE 2 Costs of Selected Treatments for Benign Prostatic Hyperplasia
| Treatment | Estimated cost over 5 years |
|---|---|
| Medical (drug) therapy | $6,294 |
| TUMT | 7,035 |
| TURP | 7,334 |
TUMT = transurethral microwave thermotherapy; TURP = transurethral resection of the prostate.
Adapted with permission from Blute M, Ackerman S, Rein AL, Beusterien K, Sullivan EM, Tanio CP, et al. Cost effectiveness of microwave thermotherapy in patients with benign prostatic hyperplasia: part II—results. Urology 2000;56:981–7.
With the wide range of treatments that are available for BPH, patient preference plays an important role. Some patients may be willing to live with the residual symptoms of a less efficacious therapy that has fewer side effects, whereas others may consider the same symptoms to represent treatment failure.

