Foot ulcers are a significant complication of diabetes mellitus and often precede lower-extremity amputation. The most frequent underlying etiologies are neuropathy, trauma, deformity, high plantar pressures, and peripheral arterial disease. Thorough and systematic evaluation and categorization of foot ulcers help guide appropriate treatment. The Wagner and University of Texas systems are the ones most frequently used for classification of foot ulcers, and the stage is indicative of prognosis. Pressure relief using total contact casts, removable cast walkers, or “half shoes” is the mainstay of initial treatment. Sharp debridement and management of underlying infection and ischemia are also critical in the care of foot ulcers. Prompt and aggressive treatment of diabetic foot ulcers can often prevent exacerbation of the problem and eliminate the potential for amputation. The aim of therapy should be early intervention to allow prompt healing of the lesion and prevent recurrence once it is healed. Multidisciplinary management programs that focus on prevention, education, regular foot examinations, aggressive intervention, and optimal use of therapeutic footwear have demonstrated significant reductions in the incidence of lower-extremity amputations.
Foot disorders such as ulceration, infection, and gangrene are the leading causes of hospitalization in patients with diabetes mellitus.1,2 Approximately 15 to 20 percent of the estimated 16 million persons in the United States with diabetes mellitus will be hospitalized with a foot complication at some time during the course of their disease.3 Unfortunately, many of these patients will require amputation within the foot or above the ankle as a consequence of severe infection or peripheral ischemia. Neuropathy is often a predisposing factor to ulceration and amputation.
The diabetic foot and its sequelae account for billions of dollars in direct medical expenditures, as well as lengthy hospital stays and periods of disability.3,4 The most characteristic lesion of the diabetic foot is a mal perforans ulceration (Figure 1), which consequently is one of the major risk factors for amputation. Approximately 85 percent of all diabetes-related lower-extremity amputations are preceded by foot ulcers.5,6
FIGURE 1.
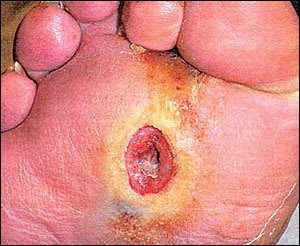
Typical diabetic foot ulcer caused by high plantar pressures at the second metatarsal head.
Etiology of Foot Ulceration
The etiology of diabetic foot ulcers usually has many components.4,7 A recent multicenter study8 attributed 63 percent of diabetic foot ulcers to the critical triad of peripheral sensory neuropathy, trauma, and deformity. Other factors in ulceration are ischemia, callus formation, and edema. Although infection is rarely implicated in the etiology of diabetic foot ulcers, the ulcers are susceptible to infection once the wound is present. Many of the risk factors for foot ulcer are also predisposing factors for amputation, because ulcers are primary causes leading to amputation.5,7,9 Recognized risk factors for diabetic foot ulceration are listed in Table 1.4
Ulcer Evaluation
A thorough evaluation of any ulcer is critical and should direct management.4 An adequate description of ulcer characteristics, such as size, depth, appearance, and location, also provides for the mapping of progress during treatment.6 The evaluation should determine the etiology of the ulcer and ascertain whether the lesion is neuropathic, ischemic, or neuro-ischemic. Failure to perceive the pressure of a 10-g monofilament is a proven indicator of peripheral sensory neuropathy and loss of protective sensation.9,10 Other common modalities that can detect insensitivity are a standard tuning fork (128 cycles per second) and a neurologic reflex hammer.
After describing the dimensions and appearance of the ulcer, the physician should examine the ulcer with a blunt sterile probe. Gentle probing can detect sinus tract formation, undermining of ulcer margins, and dissection of the ulcer into tendon sheaths, bone, or joints. A positive probe-to-bone finding has a high predictive value for osteomyelitis.11 Failure to diagnose underlying osteomyelitis often results in failure of wound healing. The existence of odor and exudate, and the presence and extent of cellulitis must be noted.12
Generally, limb-threatening infections can be defined by cellulitis extending beyond 2 cm from the ulcer perimeter, as well as deep abscess, osteomyelitis, or critical ischemia.4,6,13 Aerobic and anaerobic cultures should be taken when signs of infection, such as purulence or inflammation, are present.6 Cultures are best taken from purulent drainage or curetted material from the ulcer base.
Because all ulcers are contaminated, culture of noninfected wounds is generally not recommended.6,14 Polymicrobial infections predominate in severe diabetic foot infections and include a variety of aerobic gram-positive cocci, gram-negative rods, and anaerobes.14,15
Radiographs should be obtained in most patients with deep or longstanding ulcers to rule out osteomyelitis; however, radiographs are not a very sensitive indicator of acute bone infection.6,16 When clinical suspicion indicates osteomyelitis but radiographs are negative, additional bone or leukocyte scanning is helpful in ascertaining bone involvement. However, in the neuropathic patient, bone scans are often falsely positive because of hyperemia or Charcot's arthropathy. Leukocyte scanning or magnetic resonance imaging offers better specificity in this situation.16 Ultimately, bone biopsy is necessary to firmly establish the diagnosis of osteomyelitis.
Vascular status must always be assessed because ischemia portends a poor prognosis for healing without vascular intervention. The simple palpation of both pedal pulses and popliteal pulses is the most reliable indication of arterial perfusion to the foot. The absence of pedal pulses in the presence of a palpable popliteal pulse is a classic finding in diabetic arterial disease because of the selective involvement of the tibial arteries below the knee.6,13 Noninvasive Doppler studies should be used to augment the clinical examination as needed, although even with these tests, the severity of arterial insufficiency can be underestimated.13 Vascular surgical consultation is warranted when there is significant suspicion of ischemia.
Classification of ulcerations can facilitate a logical approach to treatment and aid in the prediction of outcome.2,4,6 Several wound classification systems have been created, based on parameters such as extent of infection, neuropathy, ischemia, depth or extent of tissue loss, and location. The most widely accepted classification system for diabetic foot ulcers and lesions is the Wagner ulcer classification system, which is based on the depth of penetration, the presence of osteomyelitis or gangrene, and the extent of tissue necrosis (Table 2).17 The drawback of the Wagner classification system is that it does not specifically address two critically important parameters: ischemia and infection.
The University of Texas diabetic wound classification system assesses the depth of ulcer penetration, the presence of wound infection, and the presence of clinical signs of lower-extremity ischemia.18 This system uses four grades of ulcer depth (0 to 3) and four stages (A to D), based on ischemia or infection, or both (Table 3).19 The University of Texas system is generally predictive of outcome, because wounds of increasing grade and stage are less likely to heal without revascularization or amputation.
Treatment
The primary goal in the treatment of diabetic foot ulcers is to obtain wound closure. Management of the foot ulcer is largely determined by its severity (grade) and vascularity, and the presence of infection.2,4,12 A systematic approach to treatment should be taken for all diabetic foot lesions, as indicated in Table 4.4 A multidisciplinary approach should be employed because of the multifaceted nature of foot ulcers and the numerous comorbidities that can occur in these patients.4,6 This approach has demonstrated significant improvements in outcomes, including reduction in the incidence of major amputation.20,21
Rest, elevation of the affected foot, and relief of pressure are essential components of treatment and should be initiated at first presentation. Ill-fitting footwear should be replaced with a postoperative shoe or another type of pressure-relieving footwear.4 Crutches or a wheelchair might also be recommended to totally off-load pressure from the foot. Although total contact casting (TCC) is considered the optimal method of management for neuropathic ulcers, it must be reapplied weekly and requires considerable experience to avoid iatrogenic lesions.22 Acceptable alternatives to TCC are removable walking braces and the “half shoe.”4,23,24 Table 54 lists several off-loading modalities that are useful in relieving pressure from foot ulcers.
A mainstay of ulcer therapy is debridement of all necrotic, callus, and fibrous tissue.6,9 Unhealthy tissue must be sharply debrided back to bleeding tissue to allow full visualization of the extent of the ulcer and detect underlying abscesses or sinuses. Topical enzymes have not been proved effective for this purpose and should only be considered as adjuncts to sharp debridement. Soaking ulcers is controversial and should be avoided because the neuropathic patient can easily be scalded by hot water.6
Although numerous topical medications and gels are promoted for ulcer care, relatively few have proved to be more efficacious than saline wet-to-dry dressings.4,6,25 Topical antiseptics, such as povidone-iodine, are usually considered to be toxic to healing wounds.6,12 Generally, a warm, moist environment that is protected from external contamination is most conducive to wound healing. This can be provided by a number of commercially available special dressings, including semipermeable films, foams, hydrocolloids, and calcium alginate swabs.25
The genetically engineered platelet-derived growth factor becaplermin (Regranex gel) is approved for use on neuropathic diabetic foot ulcers and can expedite healing.26 [Evidence level A: randomized controlled trial] Growth factors stimulate chemotaxis and mitogenesis of neutrophils, fibroblasts, and monocytes, as well as other components that form the cellular basis of wound healing.
Bioengineered skin (Apligraf) and human dermis (Dermagraft) are new types of biologically active implants for ulcers that are derived from fibroblasts of neonatal foreskins.25,27 These bioengineered products enhance healing by acting as delivery systems for growth factors and extracellular matrix components through the activity of live human fibroblasts contained in their dermal elements.
Treatment of the underlying ischemia is critical in achieving a successful outcome, regardless of topical therapies. Vascular surgical consultation should be obtained when a patient presents with an ischemic wound and when ulcers show no sign of progress despite appropriate management. A major component of the limb salvage strategy in these patients is extreme distal arterial reconstruction to restore pulsatile flow to the foot.13,20 The role of isolated distal endovascular procedures in this setting has not been determined. Vasodilator drugs have not been beneficial in promoting healing of ischemic lesions.13 Hyperbaric oxygen therapy has been used as adjunctive treatment of foot ulcers; however, support for its use is limited by the small number of carefully controlled clinical trials.28
When infection is present, aerobic and anaerobic cultures should be obtained, followed by initiation of appropriate broad-spectrum antibiotic therapy6,13,14 (Table 629).
TABLE 6 Selected Empiric Antibiotic Regimens for Diabetic Foot Ulcers
| Scenario | Drug of choice | Alternatives* |
|---|---|---|
| Mild to moderate, localized cellulitis (outpatient) | Dicloxacillin (Pathocil) | Cephalexin (Keflex); amoxicillin/clavulanate potassium (Augmentin); oral clindamycin (Cleocin) |
| Moderate to severe cellulitis (inpatient) | Nafcillin (Unipen) or oxacillin | Cefazolin (Ancef); ampicillin/sulbactam (Unasyn); clindamycin IV; vancomycin (Vancocin) |
| Moderate to severe cellulitis with ischemia or significant local necrosis | Ampicillin/sulbactam | Ticarcillin/clavulanate (Timentin); piperacillin/tazobactam (Zosyn); clindamycin plus ciprofloxacin (Cipro); ceftazidime (Fortaz) or cefepime (Maxipime) or cefotaxime (Claforan) or ceftriaxone (Rocephin) plus metronidazole (Flagyl); cefazolin (for Staphylococcus aureus); nafcillin (Unipen); oxacillin |
| Life- or limb-threatening infection | Ticarcillin/clavulanate or piperacillin/tazobactam, with or without an aminoglycoside | Clindamycin plus ciprofloxacin or tobramycin (Nebcin); clindamycin plus ceftazidime or cefepime or cefotaxime or ceftriaxone; imipenem/cilastin (Primaxin) or meropenem (Merrem); vancomycin plus aztreonam (Azactam) plus metronidazole; vancomycin plus cefepime; ceftazidime plus metronidazole |
IV = intravenous.
*— Persons with serious beta-lactam allergy may be given alternative agents.
Adapted with permission from Deery HG 2d, Sangeorzan JA. Saving the diabetic foot with special reference to the patient with chronic renal failure. Infect Dis Clin North Am 2001;15:953–81.
Antibiotic coverage should subsequently be tailored according to the clinical response of the patient, culture results, and sensitivity testing. Surgical drainage, deep debridement, or local partial foot amputations are necessary adjuncts to antibiotic therapy of infections that are deep or limb-threatening.4,30 [Reference 30—Evidence level B: uncontrolled study]
Underlying osteomyelitis is frequently present in patients with moderate to severe infections and requires aggressive bony resection of infected bone and joints followed by four to six weeks of culture-directed antibiotic therapy.13–16 The presence of deep infection with abscess, cellulitis, gangrene, or osteomyelitis is an indication for hospitalization and prompt surgical drainage. Even in the absence of bone infection, foot-sparing reconstructive procedures might be necessary to achieve final healing of the foot ulcer, especially in areas subject to exceedingly high plantar or shoe pressures.2,4,13 Table 74 illustrates clinical practice pathways that incorporate various assessment and treatment parameters that should be considered in the management of a diabetic foot ulcer or infection.
Prevention
Prevention of an initial or subsequent foot lesion is crucial to avoiding amputation.2,9,31 The best approach is to make use of a team of multidisciplinary professionals who are committed to limb salvage. Centers that have instituted teams specifically for this purpose have subsequently reported dramatic reductions in lower-extremity amputation and improved rates of primary-ulcer healing.20,21 Patient education has a central role in treatment and should include instruction on foot hygiene, daily inspection, proper footwear, and the necessity of prompt treatment of new lesions.2,9,31
Regular foot-care examinations, including debridement of calluses and ingrown toe-nails, provide an opportunity to reinforce appropriate self-care behaviors and allow for early detection of new or impending foot problems.4,6 Therapeutic shoes with pressure-relieving insoles are an essential element of ulcer prevention and have been associated with significant reductions in their development.12,22,31
Elective surgery to correct structural deformities that cannot be accommodated by therapeutic footwear can be performed as needed in certain patients.5,13,32 Common procedures include hammertoe repair, metatarsal osteotomies, plantar exostectomies, and Achilles tendon lengthening.4,32 In patients with neuropathy, these procedures can be easily performed under local anesthesia.
Working in unison with a vascular surgeon, these foot-sparing reconstructive procedures can even be performed after revascularization in an ischemic patient who might otherwise have needed amputation.13 Table 84 summarizes the important components of an amputation prevention program. Diabetic foot ulcers can be managed without amputation by following the principles discussed in this article and having a thorough understanding of the pathogenesis of these ulcers.

