West Nile virus is a mosquito-borne flavivirus and human neuropathogen. Since the virus was recognized in New York City in 1999, it has spread rapidly across the United States, with human disease documented in 39 states and the District of Columbia. West Nile virus can cause a broad range of clinical syndromes, including fever, meningitis, encephalitis, and a flaccid paralysis characteristic of a poliomyelitis-like syndrome. Approximately one in 150 infections results in severe neurologic illness. Advanced age is the greatest risk factor for severe neurologic disease, long-term sequelae, and death. Physicians should consider West Nile virus infection when evaluating febrile patients who have unexplained neurologic symptoms, muscle weakness, or erythematous rash during late spring through early fall, or throughout the year in warm climates. West Nile virus infection has no characteristic findings on routine laboratory tests, although anemia, leukocytosis, or lymphopenia may be present. Testing for IgM antibody to West Nile virus in serum or cerebrospinal fluid (samples from the acute and convalescent phases, submitted at least two weeks apart) is the most common diagnostic method. Local or state health departments usually can perform the test within 24 to 36 hours of submission. Treatment is supportive. Prevention relies on comprehensive mosquito-control programs and measures to avoid mosquito bites, including the use of mosquito repellents containing N,N-diethyl-m-toluamide.
West Nile virus, a mosquito-borne RNA flavivirus and human neuropathogen, was first identified in 1937 in the West Nile district of Uganda. The virus was recognized for the first time in the Western hemisphere in 1999, when it caused illness in 59 patients in New York City.1 Clinical syndromes have included fever, meningitis, encephalitis, and flaccid paralysis. During 2000 and 2001, West Nile virus spread rapidly through the South and Midwest, establishing enzootic activity (i.e., evidence of the virus in mosquitoes, animals, or humans) in 28 states. However, large numbers of human infections were not detected.
In 2002, West Nile virus spread westward. Activity was reported in all but six states (i.e., Arizona, Utah, Nevada, Oregon, Alaska, and Hawaii) and triggered the largest human arboviral encephalitis epidemic in U.S. history. From June 10 to December 31, 2002, there were 4,156 cases of West Nile virus infection (including 284 deaths) reported in 39 states and the District of Columbia.2 Because of the increasing prevalence of West Nile virus, family physicians should have a solid understanding of the epidemiology, clues to early diagnosis, clinical implications, and preventive strategies in humans (Table 1).
TABLE 1 Facts About West Nile Virus
| The 2002 West Nile virus epidemic was the largest human encephalitis outbreak ever documented in the Western hemisphere. |
| West Nile virus has been found in 44 states and the District of Columbia; human cases have been reported in 39 states and the District of Columbia. |
| Mosquito-borne infection is maintained in nature by birds. |
| Person-to-person transmission can occur through organ transplantation and blood and blood-product transfusions; it also may occur transplacentally and via breast milk. |
| Advanced age is the greatest risk factor for severe neurologic disease, long-term sequelae, and death from West Nile virus infection. |
| Of patients infected with West Nile virus, approximately 80 percent are asymptomatic, approximately 20 percent have a mild, self-limited, febrile illness, and less than 1 percent develop meningitis, encephalitis, or an acute flaccid paralysis. |
| Key diagnostic clues include residence in an area with West Nile virus–infected dead birds, onset of febrile illness with unexplained neurologic symptoms in summer or early fall, profound muscle weakness, and headache. |
| IgM antibody–capture enzyme-linked immunosorbent assay of serum or cerebrospinal fluid is the best diagnostic test when West Nile virus infection is suspected. |
| Rapid reporting of suspected cases of West Nile virus infection to local health departments is essential to guide control measures. |
Epidemiology
West Nile virus is indigenous to Africa, Asia, Europe, and Australia.3 Human outbreaks of infection with this virus were recorded during the 1990s in Europe and the Mediterranean area, with the largest outbreaks documented in Russia, Israel, and Romania. The viral strain introduced into the United States in 1999 likely originated from a West Nile virus homologue that was circulating in Israel during 1998.4
The West Nile virus epidemic of 2002 was concentrated in the Ohio and Mississippi river valleys (Figure 1). The geographic distribution was similar to that of a large epidemic of St. Louis encephalitis that caused 2,131 cases of disease in the United States during 1975.5 In the southern states, human cases of West Nile virus disease occurred from the summer months through December of 2002, which is beyond the characteristic arboviral disease-transmission season of June through October. In the northern states, the epidemic started later in the summer and ended earlier in the fall, most likely because of seasonal cooler weather changes, and the peak number of cases was higher than in the southern states (Figure 2).2
FIGURE 1. Distribution of West Nile Virus

Geographic distribution of West Nile virus in the United States, 1999 to 2002.
Adapted fromwww.cdc.gov/ncidod/dvbid/westnile/surv&control03maps99_02.htm.
FIGURE 2. West Nile Meningitis and Encephalitis Cases
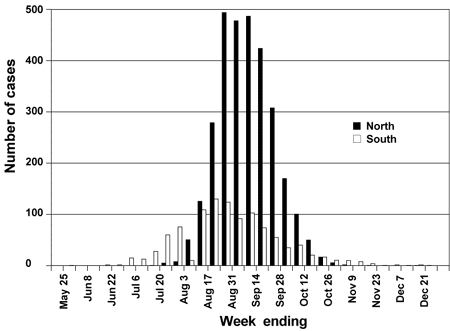
Human West Nile meningitis and encephalitis cases in 2002, by location and time of illness onset. As of April 15, 2003, there were 4,156 reported cases. Southern states included Alabama, Arkansas, California, Delaware, District of Columbia, Florida, Georgia, Kentucky, Louisiana, Maryland, Mississippi, North Carolina, Oklahoma, South Carolina, Tennessee, Texas, West Virginia, and Virginia. Northern states included Colorado, Connecticut, Illinois, Indiana, Iowa, Kansas, Massachusetts, Michigan, Minnesota, Missouri, Montana, Nebraska, New Jersey, New York, North Dakota, Ohio, Pennsylvania, Rhode Island, South Dakota, Vermont, Wisconsin, and Wyoming.
Unpublished data compiled by ArboNET. Centers for Disease Control and Prevention, Center for Infectious Diseases, Division of Vector-Borne Infectious Diseases, Fort Collins, Colo.
West Nile meningitis and encephalitis are notifiable diseases and should be reported to local health departments. The Centers for Disease Control and Prevention (CDC) collects data through ArboNET, a secure, Web-based surveillance network comprising 54 state and local public health departments. Local health departments and cooperating state and federal agencies collect and test specimens from ill animals, dead birds, captive sentinel animals (mostly chickens), mosquitoes, and live birds trapped in the wild. Demographic and clinical information about patients diagnosed with West Nile virus infection also is compiled and forwarded to the CDC through ArboNET.6 ArboNET data are used to track the epidemic temporally and geographically. Data generated locally are used to direct public health resources for research and control activities.
TRANSMISSION
Humans typically acquire West Nile virus through a bite from an infected adult mosquito. In the United States, the virus is maintained in an enzootic mosquito-bird-mosquito cycle that primarily involves Culex mosquito species.3 In temperate regions, adult mosquitoes arise in the spring from developmental aquatic stages and infect birds; this cycle continues into early fall. Infected adult mosquitoes also may overwinter in culverts and catch basins.7 Year-round transmission of West Nile virus is possible in tropical climates.
In the United States, West Nile virus appears to be particularly virulent in the corvids (e.g., crows, jays), which become ill and die at high rates.8 Unexpected deaths of large numbers of birds have been sentinel events preceding some outbreaks of West Nile virus infection in humans.9
When weather or climate promotes significant viral amplification in the enzootic cycle, bridge vector mosquitoes (i.e., mosquitoes that bite humans and birds, which may include non-Culex species) become infected in late summer and pose an additional threat to humans.10 Hot, dry summers with brief periods of unseasonably cool temperatures are thought to be favorable for the transmission of West Nile virus; epidemics of St. Louis encephalitis virus have occurred during similar conditions.11
Humans and horses usually are incidental hosts because they may not develop sufficient viremia to propagate virus amplification after a mosquito bite. During the 2002 epidemic, human viremia was implicated for the first time in person-to-person transmission through blood, blood products, and organ transplantation, and possibly through breast milk and intrauterine infection.12–14 (Figure 3).
FIGURE 3. Transmission Cycle of West Nile Virus
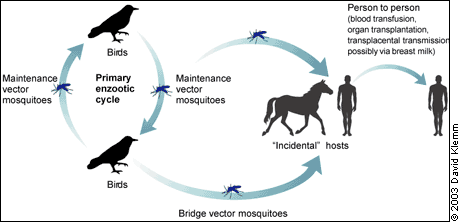
Transmission cycle of West Nile virus.
RISK FACTORS
West Nile virus infection appears to be equally distributed among all age groups and in both sexes. However, the incidence of encephalitis and death increases 20-fold among persons 50 years of age and older.1,15 During the 2002 epidemic, approximately 9 percent of patients with meningitis or encephalitis died.2
In recent urban epidemics, risk factors associated with West Nile virus infection included length of time spent outdoors, failure to apply mosquito repellent regularly, living in a building with a flooded basement, and presence of mosquitoes in the home.16,17 Physiologic factors (e.g., hypertension, diabetes, cerebrovascular disease) do not appear to be risk factors.18
West Nile virus has been reported in laboratory workers exposed to infected animals. Standard droplet and contact precautions, including safe disposal of sharp instruments and minimizing the generation of aerosols, should be used by laboratory workers who handle potentially infected animals or tissue specimens.19
In December 2002, the U.S Food and Drug Administration (FDA) recommended the voluntary withdrawal of blood and blood products that might carry West Nile virus. The risk of blood-product contamination was thought to be low but may have varied regionally.20 This year, data requested by ArboNET have been modified to help remove potentially infectious blood components from the U.S. blood supply, and use of nucleic acid–based assays was implemented in July 2003 to screen blood donors for West Nile virus.
In May 2003, the FDA revised guidelines on deferring blood donations from persons with a medical diagnosis of West Nile virus infection and from persons who report postdonation illness with symptoms suggestive of infection with the virus.21 Questions about symptoms compatible with West Nile virus infection now are included in the blood donor–history questionnaire; the FDA is evaluating the impact of these added questions. As recently introduced assays for blood-donor screening are evaluated, donor suitability guidelines may be revised.
Clinical Manifestations
Most persons with West Nile virus infection are asymptomatic.10,16 Approximately one in five infected persons develops a mild, febrile illness, and approximately one in 150 infected persons manifests neurologic symptoms.16,22 Symptomatic illness generally follows an incubation period of two to 15 days.
The majority of symptomatic patients have a self-limited, febrile illness, occasionally with headache, nausea, and vomiting. Chills, myalgias, and overwhelming fatigue are common complaints. Although the acute illness usually lasts less than seven days, fatigue may persist for several weeks. Some patients may have lymphadenopathy and conjunctivitis. Occasionally, patients develop a brief, nonpruritic, papular rash, often on the arms, legs, or trunk. Rarely, hepatitis, pancreatitis, and myocarditis have been associated with West Nile virus infection.10
A study1 of 59 hospitalized patients in New York state found that the most common symptoms were fever (90 percent), weakness (56 percent), nausea and vomiting (51 to 53 percent), headache (47 percent), and changes in mental status (46 percent). Rash, cough, stiff neck, myalgia, or arthralgia was found in less than 20 percent of these patients; only 2 percent had lymphadenopathy.
Approximately 5 percent of patients with symptomatic West Nile virus infection (less than 1 percent of all infected persons) develop more severe neurologic illness, including meningitis, encephalitis, and acute flaccid paralysis characteristic of a poliomyelitis-like syndrome.18 West Nile meningitis is characterized by acute onset of headache, nuchal rigidity, photophobia, or other signs of meningeal irritation. Patients with West Nile encephalitis have altered mental status and may have signs of meningeal irritation; those with the most severe encephalitis may progress to coma.
Patients with West Nile poliomyelitis–like syndrome have acute onset of asymmetric weakness and diminished or absent deep tendon reflexes in the absence of pain or other sensory symptoms. Electrodiagnostic studies generally show lower motor neuron involvement with preserved sensory potentials.23,24 In patients presenting with flaccid paralysis, appropriate diagnostic tests, including cerebrospinal fluid (CSF) examination and electrodiagnostic studies, should be performed before treatments are considered for other conditions, such as Guillain-Barré syndrome, stroke, or myopathy. The poliomyelitis-like syndrome may be present in the absence of fever, meningismus, or other symptoms that raise suspicion for West Nile virus infection.25
Patients with nervous system involvement may have tremor, which is generally static or kinetic in nature, and myoclonus, which often affects the upper extremities and face. Patients also may have signs of parkinsonism, including cogwheel rigidity, bradykinesia, and postural instability.
A prospective study26 conducted during the 2002 West Nile virus epidemic in Louisiana found that the median age was 70 years in patients with encephalitis, 56 years in those with acute flaccid paralysis, and 36 years in those with meningitis. Age-related underlying risk factors for invasive neurologic disease likely account for this difference.
Diagnosis
Family physicians should consider West Nile virus infection in febrile patients who have unexplained neurologic symptoms, muscle weakness, erythematous rash, or headache during late spring through early fall, or at any time of the year in areas with warmer climates, such as the Mississippi Delta. Initial laboratory tests should include the following: a complete blood count; serologic testing for West Nile virus; CSF analysis for cell count, glucose level, and protein level; and, if indicated, neuroimaging and electrodiagnostic studies.
Laboratory results from patients during recent outbreaks of West Nile virus disease have shown normal or elevated total white blood cell counts and, sometimes, anemia.1,27,28 Occasionally, patients have lymphocytopenia, an unusual feature of viral encephalitis that may indicate West Nile virus infection.1,14 Lymphocytic-predominant pleocytosis with elevated protein and normal glucose levels are common CSF findings. Computed tomography of the brain usually does not demonstrate acute abnormalities, but magnetic resonance imaging may show leptomeningeal or parenchymal changes.1,27,28
The best laboratory test for diagnosing West Nile virus infection is an IgM antibody–capture enzyme-linked immunosorbent assay (MAC-ELISA) of serum or CSF collected eight to 21 days after the onset of symptoms. This simple, sensitive (95 percent) diagnostic test is available commercially and through local or state health departments.10 State public health laboratories usually can perform the test within 24 to 36 hours of submission. Testing of serum in the acute and convalescent phase (i.e., sera collected at least two weeks apart) may provide laboratory confirmation of infection. Since IgM antibody usually does not cross an intact blood-brain barrier, a single West Nile virus–specific IgM positive antibody titer in CSF can confirm central nervous system infection.
Viral culture of CSF and polymerase chain reaction (PCR) testing of serum should not be used as routine screening tools because of their low sensitivity for West Nile virus diagnosis.29 The CDC has established multiple serologic, PCR-based, and viral isolation criteria for confirmation of West Nile virus infection (Table 2).30 [Evidence level C, consensus/expert opinion]
TABLE 2 Criteria for Confirmation of West Nile Virus Infection
| Isolation of West Nile virus from tissue, blood, CSF, or other body fluid, or demonstration of West Nile viral antigen or genomic sequences |
| IgM antibody to West Nile virus in CSF as detected by MAC-ELISA |
| At least a fourfold serial change in plaque-reduction neutralizing antibody titer to West Nile virus in paired, appropriately timed (i.e., at least 14 days apart) serum or CSF samples |
| West Nile virus–specific IgM (by MAC-ELISA) and IgG antibody (screened by ELISA and confirmed by plaque-reduction neutralizing tests) in a single serum specimen |
CSF = cerebrospinal fluid; MAC-ELISA = IgM antibody–capture enzyme-linked immunosorbent assay.
Information from reference 30.
Four points about serum serology further underscore the importance of following the testing of sera from the acute phase with testing of sera from the convalescent phase:
- Approximately 10 percent of West Nile virus–infected patients in New York City (1999 and 2000) tested negative for serum IgM antibody when the acute specimen was collected within eight days of symptom onset.
- Because IgM antibodies may persist for one year or longer after infection, the presence of a single IgM antibody titer may be unrelated to a patient's current illness.29
- Antibodies to St. Louis encephalitis virus, which is enzootic in many states, may cross-react with West Nile virus antigen in serologic tests.
- When interpreting serologic tests, physicians should ask patients if they recently have been vaccinated against or infected with related flaviviruses (e.g., yellow fever virus, Japanese encephalitis virus, dengue virus), because antibody to these viruses may cause nonspecific positive MAC-ELISA results.31 Plaque reduction neutralization tests, the most specific serologic tests available for arthropod-borne flavivirus infection, can help identify false-positive or cross-reaction results using acute-phase and convalescent-phase specimens.10
Treatment
Treatment of West Nile virus infection is supportive. Patients with volume depletion because of nausea and vomiting may need oral or intravenous fluids. Patients with meningitis or encephalitis may need pain control for headache. Patients with profound encephalopathy or acute flaccid paralysis may require mechanical ventilation.
In vitro studies have suggested that ribavirin and interferon alfa-2b may be useful in the treatment of West Nile virus disease, but the efficacy of these agents has not been demonstrated in controlled clinical trials.32,33 An FDA-approved clinical trial has been launched to study the effectiveness of interferon alfa-2b in patients with West Nile encephalitis.34 To date, no controlled trials have studied the use of corticosteroids, anticonvulsants, or osmotic agents in the treatment of neurologic disease caused by West Nile virus.
Prevention
No vaccine is licensed for the prevention of West Nile virus disease in humans, although laboratory development is currently underway. Thus, comprehensive mosquito-control programs and personal protective measures are paramount in preventing West Nile virus infection in endemic areas. State and local agencies should identify vector species and breeding sites, and implement aggressive surveillance and targeted control early in the transmission season to restrict enzootic viral amplification.
In southern areas such as the Mississippi Delta, the predominant mosquito vector for West Nile virus is Culex quinquefasciatus, which breeds in drainage ditches and septic ponds. Municipal mosquito-abatement programs should emphasize preventive strategies such as larvicide treatment.
In northern regions, the principal mosquito vector is Culex pipiens, which develops in standing water. Eliminating household stagnant-water sources, such as flowerpots, pet bowls, clogged rain gutters, discarded tires, swimming pool covers, and birdbaths, can reduce breeding sites.
Protective measures to limit exposure to potentially infected mosquitoes include avoiding outdoor activity during peak mosquito-biting periods (usually dusk to dawn), wearing long-sleeved shirts and pants when outdoors during the early-evening and night hours, and applying mosquito repellent containing N,N-diethyl-m-toluamide (DEET) to clothing and exposed skin. Some mosquito-repellent preparations (e.g., Avon's Skin-So-Soft®, citronella) are minimally effective at best.35
Final Comments
West Nile virus likely will continue to advance throughout the western United States within the next few years and is expected to follow local and regional flyway patterns of viremic birds. Physicians provide a vital link in recognizing new and unexpected disease activity. Few data exist regarding long-term sequelae from West Nile virus infection, the magnitude of risk of transmission associated with organ transplantation and blood transfusion, potential therapeutic interventions, and vaccine options. Physicians engaged in diagnosing, treating, and reporting West Nile virus infection may greatly enhance knowledge of this emerging infectious disease.

