Hearing loss is a common problem that can occur at any age and makes verbal communication difficult. The ear is divided anatomically into three sections (external, middle, and inner), and pathology contributing to hearing loss may strike one or more sections. Hearing loss can be categorized as conductive, sensorineural, or both. Leading causes of conductive hearing loss include cerumen impaction, otitis media, and otosclerosis. Leading causes of sensorineural hearing loss include inherited disorders, noise exposure, and presbycusis. An understanding of the indications for medical management, surgical treatment, and amplification can help the family physician provide more effective care for these patients.
More than 28 million Americans have some degree of hearing impairment. The differential diagnosis of hearing loss can be simplified by considering the three major categories of loss. Conductive hearing loss occurs when sound conduction is impeded through the external ear, the middle ear, or both. Sensorineural hearing loss occurs when there is a problem within the cochlea or the neural pathway to the auditory cortex. Mixed hearing loss is concomitant conductive and sensorineural loss.
Evaluation
A thorough history and a careful physical examination are essential to the diagnosis and treatment of hearing loss. Pertinent questions to ask patients are listed in Table 1.
TABLE 1 Questions for Evaluating Hearing Loss
| When did your hearing loss begin? |
| Was your hearing loss sudden, or has your hearing slowly been getting worse? |
| Does your hearing loss involve one or both ears? |
| Have you been having ringing in your ear, fullness in your ear, dizziness, ear drainage, or ear pain? |
| Is there a history of hearing loss in your family? |
| What is your job? What is the noise level in your workplace? |
| Do you have a history of ear infections, ear injury, or straining to hear? |
| Do you have a history of stroke, diabetes, or heart disease? |
| What medicines are you currently taking? |
| Have you received any intravenous antibiotics, diuretics, salicylates, or chemotherapy? |
The physical examination begins with visualization and palpation of the auricle and periauricular tissues. An otoscope should be used to examine the external auditory canal for cerumen, foreign bodies, and abnormalities of the canal skin. The mobility, color, and surface anatomy of the tympanic membrane should be determined (Figure 1). A pneumatic bulb is required to accurately assess the tympanic membrane and the aeration of the middle ear.
FIGURE 1.
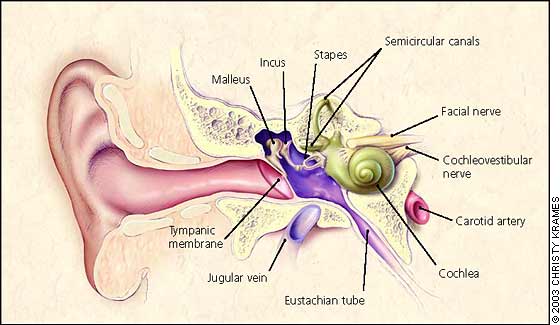
Anatomy of the ear.
Weber's test is performed by softly striking a 512-Hz tuning fork and placing it midline on the patient's scalp, or on the forehead, nasal bones, or teeth. If the hearing loss is conductive, the sound will be heard best in the affected ear. If the loss is sensorineural, the sound will be heard best in the normal ear. The sound remains midline in patients with normal hearing.
The Rinne test compares air conduction with bone conduction. The tuning fork is struck softly and placed on the mastoid bone (bone conduction). When the patient no longer can hear the sound, the tuning fork is placed adjacent to the ear canal (air conduction). In the presence of normal hearing or sensorineural hearing loss, air conduction is better than bone conduction. Therefore, sound is still heard when the tuning fork is placed adjacent to the ear canal. In the presence of conductive hearing loss, bone conduction is better than air conduction, and the sound is not heard when the tuning fork is placed adjacent to the canal.
Whispering softly into the patient's ear or holding a softly ticking wristwatch close to the ear can be helpful in making a gross evaluation of hearing.
Once the ear has been examined and initial hearing tests have been done, the head and neck are examined, and the cranial nerves are evaluated.
Formal audiography is more sensitive and specific than a tuning fork examination and thus is essential in most patients with hearing loss. Audiograms objectively measure hearing levels and compare them with standards adopted by the American National Standards Institute in 1969.1 Normal hearing levels are 20 dB or better across all frequencies. The audiogram measures air conduction and bone conduction and presents them graphically across the hearing frequencies. Audiographically demonstrated conductive hearing loss results in the air line falling below the bone line, creating an air-bone gap.
Speech testing should be performed using standard word lists. The speech reception threshold is the sound level at which 50 percent of presented words are understood. The speech recognition score is the percentage of words understood at 40 dB above the speech reception threshold.
Conductive Hearing Loss
EXTERNAL EAR
Complete occlusion of the ear canal by cerumen is a frequent cause of conductive hearing loss. Cotton-tipped applicators are notorious for worsening cerumen impaction. Warm water (body temperature) irrigation is a safe method of removing cerumen in patients who have no history of otitis media, perforation of the tympanic membrane, or otologic surgery. Use of an otoscope and a curette allows cerumen to be removed under direct vision. The distance to the tympanic membrane must be kept in mind, because otoscopes do not allow for depth perception. A variety of softening preparations are available if cerumen is too firm to remove. Aqueous-based preparations, including docusate sodium, sodium bicarbonate, and hydrogen peroxide, are effective cerumenolytics.2,3
Foreign bodies in the external auditory canal also can cause unilateral conductive hearing loss. These foreign bodies can be removed by irrigation or with a curette. If the object is not impacted or hygrostatic, warm water irrigation probably should be attempted first. If this approach is not effective, the foreign body can be removed with an instrument if the patient is cooperative. If the patient is uncooperative, removal in an operating room may be necessary.
Otitis externa is an infection of the skin of the external auditory canal. Patients with otitis externa experience pain on manipulation of the pinna or tragus, and their ear canal is edematous and filled with infectious debris. Conductive hearing loss may occur if swelling and debris occlude the canal. The most common pathogens in otitis externa are Pseudomonas aeruginosa and Staphylococcus aureus.4 Treatment involves debridement of the canal, followed by the application of ototopical drops. In patients with severe otitis externa, a wick is placed in the ear for two to three days to ensure delivery of the medication. Oral antibiotics that are effective against P. aeruginosa and S. aureus are helpful in patients with severe infection. The conductive hearing loss resolves after the inflammation subsides.
Exostoses and osteomas are benign bony growths of the external auditory canal that interfere with normal cerumen migration, leading to occlusion and conductive hearing loss. Exostoses are multiple and bilateral, and are found adjacent to the tympanic membrane. Patients with exostoses often report a history of cold-water swimming. Osteomas are single and unilateral, and are found at the bony-cartilaginous junction (Figure 2). If symptomatic, exostoses and osteomas are removed surgically, but this is rarely necessary.
Figure 2.
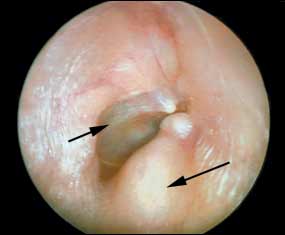
Otoscopic view of osteoma of the external auditory canal, showing tympanic membrane (short arrow) and an osteoma (long arrow).
Uncommon causes of external auditory canal obstruction include cysts and tumors. Sebaceous cysts, fibromas, papillomas, adenomas, sarcomas, carcinomas, and melanomas also have been reported. If a malignancy is suspected, prompt biopsy is indicated.
MIDDLE EAR
Middle ear pathology may lead to conductive hearing loss. Perforations of the tympanic membrane cause hearing loss by reducing the surface area available for sound transmission to the ossicular chain (Figure 3). The main causes of tympanic membrane perforations are chronic otitis media and trauma. In patients who have had chronic otitis media with tympanic membrane perforation, otoscopic examination and debridement are essential. Ototopical antibiotics (ofloxacin [Floxin]) are necessary, and oral antibiotics may be helpful. An accurate assessment of the patient's tympanic membrane and hearing can be made only when the ear is dry.
FIGURE 3.
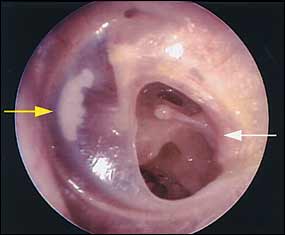
Otoscopic view of perforation of the tympanic membrane (white arrow) and myringosclerosis (yellow arrow).
Traumatic perforations of the tympanic membrane can occur because of water accidents, barotrauma, explosions, penetrating injury, or temporal bone fractures. Small perforations (less than 2 mm) often heal spontaneously.5 In the acute setting, blood may obstruct the ear canal and prevent visualization of the membrane. Ototopical antibiotics and precautions to keep the ear dry are recommended. If the perforation or hearing loss persists beyond two months, the patient should be referred for consideration of surgical correction. Trauma also can cause ossicular injury or hemotympanum presenting as hearing loss.
Otitis media is the most common cause of conductive hearing loss in children.6 Middle ear effusions decrease the mobility of the tympanic membrane and the ossicular chain. This loss of mobility results in an average hearing loss of 20 to 30 dB. The diagnosis of otitis media can be confirmed by tympanometry and audiometry, and resolution of the effusion restores hearing. Myringotomy tubes are recommended for use in children with recurrent acute otitis media (more than three episodes in six months or four episodes in one year), chronic middle ear effusions (more than three months in duration), or significant hearing impairment (greater than 30 dB along with an effusion).7 [Evidence level C, consensus opinion]
Cholesteatoma is an accumulation of squamous epithelium within the middle ear. This mass may be seen in patients with otitis media. Cholesteatomas are divided into two types: congenital and acquired. Congenital cholesteatoma presents as a pearly white mass located behind an intact tympanic membrane in a patient with unilateral conductive hearing loss. Acquired cholesteatoma results from a retracted or perforated tympanic membrane with an ingrowth of epithelium. Cholesteatomas are locally destructive and characterized by chronic drainage. Conductive hearing loss caused by ossicular erosion is present in 90 percent of patients with cholesteatomas.8 Longstanding cholesteatomas expand to involve the mastoid, inner ear, and facial nerve. Suspicion of cholesteatoma warrants surgical consultation.
Myringosclerosis of the tympanic membrane develops in response to infection or inflammation (Figure 3). Irregular white patches consisting of calcium are visible on the membrane.9 Isolated myringosclerosis of the tympanic membrane rarely causes significant conductive hearing loss. However, extensive myringosclerosis, referred to as tympanosclerosis, involves the tympanic membrane, ossicular chain, and middle ear mucosa, and causes significant conductive hearing loss by stiffening the entire system.
Otosclerosis is characterized by abnormal bone deposition at the footplate (base of stapes). This bone deposition leads to fixation of the stapes at the oval window, preventing normal vibration. Otosclerosis typically presents as progressive bilateral conductive hearing loss in middle-aged white women. It is the leading cause of conductive hearing loss in adults who do not have a middle ear effusion or a history of otitis media.10 There is usually a positive family history. Treatment consists of amplification with hearing aids or surgical repair by stapedectomy (Figure 4).
FIGURE 4.
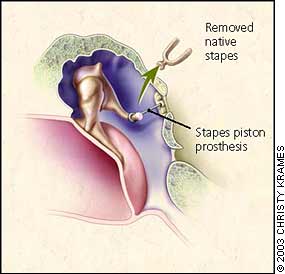
Stapedectomy: surgical repair of fixated stapes caused by otosclerosis. Working through an operation microscope, the surgeon lifts the ear drum, removes the nonfunctioning stapes, and positions the prosthesis.
FIGURE 5.
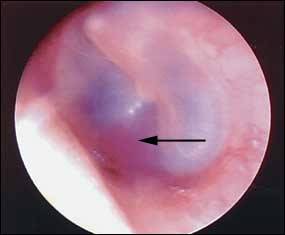
Otoscopic view of glomus tumor (arrow).
Glomus tumors are a rare cause of conductive hearing loss (Figure 5). These neuroendocrine tumors arise from the adventitia of the jugular bulb or the neural plexus within the middle ear space. Characteristically, patients presenting with glomus tumors are women 40 to 50 years of age who report pulsatile tinnitus and hearing loss. On examination, a pulsating reddish-blue mass may be seen behind an intact tympanic membrane. However, diagnosis of these tumors is difficult, and computed tomography of the temporal bones is required. An anomalous carotid artery or jugular bulb may present in a similar fashion. Clues to the diagnosis of conductive hearing loss are listed in Table 2.
TABLE 2 Clues to the Diagnosis of Conductive Hearing Loss
| History | Physical findings | Suggested cause of conductive hearing loss |
|---|---|---|
| Sudden painless loss of hearing | Cerumen | Complete canal occlusion |
| Sudden painful loss of hearing | Narrow canal with debris | Otitis externa |
| Normal canal with red, immobile tympanic membrane | Chronic otitis media | |
| Gradual painless loss of hearing | Immobile tympanic membrane | Middle ear effusion |
| Normal mobile tympanic membrane | Otosclerosis | |
| Reddish-blue pulsating mass behind intact tympanic membrane | Glomus tumor or vascular anomaly | |
| Retracted or perforated tympanic membrane, with chronic drainage | Cholesteatoma |
Sensorineural Hearing Loss
Sensorineural hearing loss encompasses disorders that affect the inner ear and the neural pathways to the auditory cortex. Although most patients with this type of hearing loss are adults, children also can be affected. Hereditary and non-hereditary congenital hearing loss are the two major pediatric classifications. The majority of hereditary losses are autosomal recessive and are frequently associated with other systemic findings. More than 100 congenital syndromes are associated with sensorineural hearing loss.
The consequences of delayed detection can be significant. Neonates considered at high risk for congenital hearing loss (Table 3)11 traditionally have been screened, and 30 states now require universal newborn auditory screening.12 The American Academy of Pediatrics and several other organizations endorse universal auditory screening. However, in 2001, the U.S. Preventive Services Task Force concluded that current “evidence is insufficient to recommend for or against routine screening of newborns for hearing loss during the postpartum hospitalization.”13
TABLE 3 High-Risk Indicators* of Hearing Loss in Infants and Young Children
| Birth to 28 days |
| Family history of permanent sensorineural hearing loss during childhood |
| In utero infection (e.g., toxoplasmosis, rubella, cytomegalovirus infection, herpes) |
| Ear or other craniofacial abnormalities |
| Illness or condition requiring admission to neonatal intensive care unit for at least 48 hours |
| Physical features or other stigmata associated with a syndrome known to include sensorineural or conductive hearing loss |
| 29 days to 24 months |
| Parental or caregiver concern about hearing, speech, language, or developmental delay |
| Family history of permanent hearing loss during childhood |
| Physical features or other stigmata associated with a syndrome known to include sensorineural or conductive hearing loss or eustachian tube dysfunction |
| Head trauma |
| Postnatal infection associated with sensorineural hearing loss (e.g., meningitis) |
| In utero infection (e.g., toxoplasmosis, rubella, cytomegalovirus infection, herpes, syphilis) |
| Neonatal indicators: hyperbilirubinemia requiring exchange transfusion, persistent pulmonary hypertension associated with mechanical ventilation, conditions requiring extracorporeal membrane oxygenation |
| Syndromes associated with progressive hearing loss (e.g., neurofibromatosis, osteopetrosis, Usher's syndrome) |
| Neurodegenerative disorders (e.g., Hunter's syndrome) or sensory motor neuropathies (e.g., Friedreich's ataxia, Charcot-Marie-Tooth disease) |
| Head trauma |
| Recurrent or persistent otitis media with effusion for at least three months |
*—These indicators are red flags and may help physicians in referring children for audiologic testing.
Information from Joint Committee on Infant Hearing. Year 2000 position statement: principles and guidelines for early hearing detection and intervention programs. Pediatrics 2000;106:809–10.
BILATERAL HEARING LOSS
Presbycusis is a symmetric, progressive deterioration of hearing in elderly patients, and is a diagnosis of exclusion (Table 4). The etiology is a combination of inherited and environmental factors, including lifetime noise exposure and tobacco use. High-frequency hearing and speech discrimination ability are affected. Audiology consultation is recommended for accurate testing and consideration of amplification, if appropriate.
TABLE 4 Clues to the Diagnosis of Sensorineural Hearing Loss
| History | Physical findings | Audiogram | Suggested cause of sensorineural hearing loss |
|---|---|---|---|
| Gradual hearing loss, noise exposure, tobacco use | Elderly patients with normal tympanic membrane | Bilateral, symmetric high-frequency loss | Presbycusis |
| Gradual hearing loss, tinnitus, noise exposure | Normal tympanic membrane | Bilateral, symmetric loss centered at 4,000 Hz | Noise-induced traumatic loss |
| Rapidly progressive hearing loss, possibly fluctuating, bilateral loss | Normal tympanic membrane, with possible vertigo or disequilibrium | Any abnormal configuration with poor speech discrimination | Autoimmune hearing loss |
| Sudden unilateral hearing loss, tinnitus, vertigo, head trauma, straining | Normal tympanic membrane; vertigo and nystagmus, with positive pneumatic pressure | Any unilateral abnormal configuration | Perilymph fistula |
| Sudden, fluctuating, unilateral hearing loss, tinnitus, episodic vertigo | Normal tympanic membrane | Unilateral low-frequency loss | Meniere's disese |
| Gradual unilateral hearing loss, tinnitus | Normal tympanic membrane, possible facial nerve weakness and unsteadiness | Any unilateral abnormal configuration | Acoustic neuroma |
Noise trauma is the most common preventable cause of sensorineural hearing loss. The noise source may be occupational, recreational, or accidental. Gunfire, explosions, and loud music can cause irreversible hearing impairment. High frequencies are affected first, typically at 4,000 Hz, followed by middle and lower frequencies. The hearing loss is accompanied by high-pitched tinnitus. Aggressive use of noise protection is recommended to prevent this form of hearing loss. The use of foam-insert earplugs decreases noise exposure by 30 dB.
A less common cause of hearing loss is ototoxin exposure, typically from diuretics, salicylates, aminoglycosides, and many chemotheraupetic agents. These medications must be administered carefully in patients who are elderly, have poor renal function, require a prolonged course of medication, or require simultaneous administration of multiple ototoxic agents. Patients with ototoxin exposure may experience hearing loss or dizziness.
Autoimmune hearing loss has been diagnosed with increasing frequency since the 1980s. Patients present with rapidly progressive bilateral sensorineural hearing loss and poor speech discrimination scores, and they also may have vertigo or disequilibrium. Hearing loss progresses over three to four months, and an associated autoimmune disorder may be present. Symptoms usually improve with the administration of oral prednisone, and response to this steroid is currently the best way to make the diagnosis. Low-dose methotrexate therapy is becoming an accepted alternative to long-term prednisone therapy.14
UNILATERAL HEARING LOSS
Temporal bone fractures can cause unilateral sensorineural and conductive hearing loss. When the fracture line involves the bony labyrinth (cochlea or vestibule), sensorineural hearing loss occurs. Temporal bone injuries are associated with facial nerve paralysis, cerebrospinal fluid leakage, and other intracranial injuries. Early consultation is essential, and prompt surgical intervention may be required.
Trauma may cause rupture of the round or oval window membranes, with perilymph leaking into the middle ear (fistula). Patients experience abrupt loss of hearing, along with vertigo and tinnitus. Perilymph fistulas also may occur after straining, lifting, coughing, or sneezing, and are managed with three to six weeks of bed rest, followed by surgical repair if symptoms do not improve.15
Meniere's disease is another cause of sensorineural hearing loss. Patients report unilateral fluctuating hearing loss with aural fullness, tinnitus, and episodic vertigo. Initially, the hearing loss is in the low frequencies, but higher frequencies are affected as the disease progresses. The etiology of Meniere's disease remains unclear, but endolymphatic hydrops (increased fluid pressure within the inner ear) has been identified. The work-up consists of serial audiometry to document a fluctuating loss, vestibular testing to verify the diseased ear, and radiographic imaging to rule out an acoustic tumor. Treatment includes a low-salt diet, diuretics, and vestibular suppressants. Hearing aids are often ineffective because patients suffer from poor speech discrimination, as well as diminished tolerance to amplified sound. Chemical labyrinthectomy with gentamicin is now a common nonsurgical option for control of vertigo if medical management fails.16
Idiopathic unilateral sudden sensorineural hearing loss, defined as hearing loss of 30 dB within a three-day period, is an urgent situation.17 Associated symptoms include tinnitus, vertigo, and aural fullness. Speculative etiologies include viral infections and vascular insults. Rarely, perilymph fistulas and acoustic neuromas are implicated. A history of upper respiratory infection within a month of the hearing loss is often described in patients who have a viral etiology. The work-up includes audiometry, followed by radiographic imaging to rule out an acoustic tumor. Patients should begin treatment with oral steroids within three weeks. One study found that patients with minimal hearing loss, no vestibular symptoms, and early treatment have better outcomes.18
Patients with acoustic neuromas present with unilateral sensorineural hearing loss approximately 10 to 22 percent of the time19 (Figure 6). Patients with asymmetric sensorineural hearing loss require evaluation for a retrocochlear tumor. Acoustic neuromas and other cerebellopontine-angle tumors need to be ruled out. Magnetic resonance imaging of the brain with gadolinium continues to be the gold standard for diagnosing these masses.
FIGURE 6.
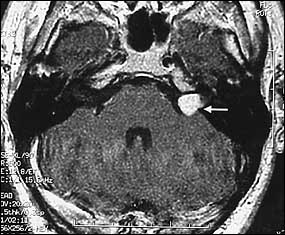
Magnetic resonance image showing an acoustic neuroma (arrow).

