
Am Fam Physician. 2003;68(7):1343-1350
With the rising popularity of international travel to exotic locations, family physicians are encountering more febrile patients who recently have visited tropical countries. In the majority of cases, the fever is caused by a common illness such as tracheobronchitis, pneumonia, or urinary tract infection. However, fever in returned travelers always should raise suspicion for a severe or potentially life-threatening tropical infection. In addition to the usual medical history, physicians should obtain a careful travel history, a description of accommodations, information about pretravel immunizations or chemoprophylaxis during travel, a sexual history, and a list of exposures and risk factors. The extent and type of lymphadenopathy are important diagnostic clues. Altered mental status with fever is an alarm symptom and requires urgent evaluation and treatment. Malaria must be considered in patients who traveled even briefly within an endemic area. Enteric fever is treated with fluoroquinolones, dengue fever with supportive measures only, leptospirosis with penicillin or doxycycline, and rickettsial infections with doxycycline.
Estimates indicate that 15 to 37 percent of short-term travelers experience a health problem during an international trip, and up to 11 percent of returned travelers have a febrile illness.1 The majority of travelers with fever have infections that are common in nontravelers, such as upper respiratory tract infection, urinary tract infection, or community-acquired pneumonia.2,3
Once routine infections have been considered, the differential diagnosis should be expanded to include travel-related infections. The most serious cause of fever in travelers returning from the tropics is Plasmodium falciparum malaria, which can be rapidly fatal.2,3 Other important causes of fever in returned travelers include typhoidal and non-typhoidal salmonellosis, dengue fever, viral hepatitis, and rickettsial infections.2,3
The family physician also should considerthat the trip may be temporally, but not causally, related to a fever. In rare instances, noninfectious diseases such as malignancies or collagen vascular diseases present coincidentally during travel.
Approach to the Diagnosis
A systematic approach to the evaluation of fever in the returned traveler includes identification of special risk factors, exposures, or physical findings that will help focus the work-up (Table 1). Consultation with an infectious diseases subspecialist may assist in arriving at a diagnosis.
| 1. Always consider mundane causes such as urinary tract and upper respiratory tract infections. |
| 2. Do not forget to consider nontravel-related causes: the fever may have nothing to do with the trip. |
| 3. If the incubation period appears to be short (less than 21 days), the majority of patients will have malaria, typhoid fever, or dengue fever. Rickettsial diseases (also a short incubation period) are becoming more frequent causes of fever in returned travelers. |
| 4. If the incubation period appears to be long (more than 21 days), the majority of patients will have malaria or tuberculosis. In addition, consider hepatitis A in unimmunized patients. |
| 5. Consult with an infectious diseases subspecialist early if the patient is particularly ill or has altered mental status. Although meningococcemia and viral hemorrhagic fevers are highly uncommon, consider these diagnoses, because they are medical emergencies. |
| 6. If attention to these guidelines still does not establish the diagnosis, consider uncommon causes. In this situation, consult with an infectious diseases subspecialist. |
PRETRAVEL PREPARATION
Pretravel immunizations and chemoprophylaxis taken during travel must be determined, because these will influence the probability of acquiring infections.4,5 Proper administration of vaccines against hepatitis A, hepatitis B, and yellow fever effectively rules out these infections.6 However, immune globulin as a preventive for hepatitis A and vaccines against typhoid fever are only about 70 percent effective; therefore, hepatitis A and typhoid fever still should be considered in patients who have been immunized with these agents.4
Childhood vaccination against diseases such as polio, diphtheria, or measles may not provide adequate protection in adults unless a booster dose has been administered or natural disease has been reported.6 Immigrants from developing countries may not have received routine immunizations.
If a patient recently has traveled to an area where malaria is endemic, the physician should determine whether personal protective measures (e.g., insect repellents, bed nets) and chemoprophylaxis were used.4,6 Although these measures clearly decrease the risk of acquiring malaria, no antimalarial chemoprophylactic regimen is completely protective. Poor adherence with antimalarial drug regimens is well documented in travelers who contract malaria.4
The health of the patient before travel also is important. The presence of underlying medical conditions (e.g., cardiopulmonary disease, immunosuppression, asplenia) may increase susceptibility to various infections. Furthermore, medications taken for treatment of an underlying condition may alter the presentation of certain diseases.
TRAVEL HISTORY
Questions about the travel history should focus on the patient's exact itinerary, reason for travel, and accommodations.
Travel Itinerary
The risk of acquiring a travel-related infection depends on the precise geographic location and the length of stay at each destination.4,5 Specific regions visited within each country should be determined, because some infections are focally transmitted, and risk is only present when traveling in endemic areas.4,6 For example, malaria may be a risk only in rural areas of a country. The Centers for Disease Control and Prevention (CDC) publishes a reference, Health Information for International Travel, 2003–2004,7 detailing specific infections that are found in different locations. A more up-to-date version of this reference is available on the CDC Web site (www.cdc.gov/travel). Infections can be acquired en route, so layovers and intermediate stops should be identified. The type of transportation also is relevant, because outbreaks of many types of infections have been linked specifically to airplanes, trains, and cruise ships.
Purpose of Travel
Determining the reason for travel can assist in assessing the risk for certain infections. The purpose of the trip may affect the duration of travel, the likelihood of travel in isolated or rural areas, and the likelihood of sexual contact with local inhabitants.8
Accommodations
Travelers who stay in modern hotels in major urban centers generally have fewer exposures than backpackers or volunteer workers who spend significant time in rural settings with the local population.9 Persons who visit family and friends while abroad also are at increased risk of becoming ill because they often stay in homes away from usual tourist routes.10
EXPOSURE HISTORY
The risk of acquiring a tropical infection is affected by the patient's activities during travel.11 Because many tropical illnesses have nonspecific signs and symptoms, identification of a unique exposure may provide the only clue to the correct diagnosis. Activities in remote areas increase the chance of exposure to insect vectors and fresh-water lakes and streams that may harbor schistosomes or leptospires.5 In addition, eating certain foods increases the risk for food-borne illnesses.
Sexual contact with new partners can occur during travel and has been reported by up to one half of young persons who visit tropical regions.12 This contact can result in exposure to sexually transmitted diseases (STDs). Although STDs usually present with genital lesions, occasionally only fever and nonspecific systemic symptoms may be noted.4,5 The sexual history should include the number of partners, types of sexual activities, and protection used.
| Exposure | Diseases | |
|---|---|---|
| Undercooked food | Cholera, nontyphoidal salmonellosis, trichinosis, typhoid fever | |
| Untreated water | Cholera, hepatitis A, nontyphoidal salmonellosis, typhoid fever | |
| Unpasteurized dairy products | Brucellosis, tuberculosis | |
| Fresh water contact | Leptospirosis, schistosomiasis | |
| Sexual contact | Chancroid, gonorrhea, hepatitis B, human immunodeficiency virus infection, syphilis | |
| Animals | Brucellosis, plague, Q fever, rabies, tularemia | |
| Insects | ||
| Mosquitoes | Dengue fever, malaria | |
| Ticks | Rickettsial diseases, tularemia | |
| Reduviids | American trypanosomiasis | |
| Tsetse flies | African trypanosomiasis | |
| Sick contacts | Meningococcal disease, tuberculosis, viral hemorrhagic fevers | |
INCUBATION PERIOD
The physician may be able to estimate the incubation period for a patient's illness based on the sequence of symptoms, travel history, and exposures. This step can help narrow the differential diagnosis by prompting consideration or elimination of certain infections. Symptoms of dengue fever, typhus, and viral hemorrhagic fever usually begin within 10 days of exposure.4,6 Typhoid fever may present up to 21 days after exposure to contaminated food or drink. The usual incubation period for P. falciparum malaria ranges from eight to 40 days, but infection with the other three Plasmodium species that cause malaria (i.e., Plasmodium vivax, Plasmodium ovale, and Plasmodium malariae) may not become clinically apparent for several months or even years after exposure. In addition, the usual incubation period for malaria can be lengthened if the patient has taken antimalarial medications.13 [Evidence level B, observational study] The incubation periods for various tropical diseases are provided in Table 3.4
| Fewer than 21 days | More than 21 days |
|---|---|
| East African trypanosomiasis | Acute human immunodeficiency virus infection |
| Dengue fever | Acute systemic schistosomiasis (Katayama fever) |
| Japanese encephalitis | Amebic liver abscess |
| Leptospirosis | Borreliosis (relapsing fever) |
| Malaria | Brucellosis |
| Meningococcemia | Leishmaniasis |
| Nontyphoidal salmonellosis | Malaria (especially after ineffective prophylaxis) |
| Plague | Rabies |
| Typhoid fever | Tuberculosis |
| Typhus | Viral hepatitis (A, B, C, D, E) |
| Viral hemorrhagic fevers | West African trypanosomiasis |
| Yellow fever |
PHYSICAL FINDINGS
A thorough physical examination of the patient can help guide the physician toward the correct diagnosis. Key findings and their implications for the returned traveler with fever are summarized in Table 4.4–6,9 The presence of localized or generalized lymphadenopathy is especially helpful diagnostically (Table 5).6 Fever and altered mental status in the returned traveler represent a medical emergency and should be managed in consultation with an infectious diseases subspecialist (Table 6).6
| Area of physical examination | Diagnostic value |
|---|---|
| Vital signs | A pulse rate that is slow for the degree of fever (pulse-temperature dissociation) may suggest typhoid fever or a rickettsial disease. |
| Skin | A maculopapular rash may be present in many travel-related infections, notably dengue fever, leptospirosis, and typhus, as well as in acute human immunodeficiency virus infection and acute hepatitis B.4,6 Drug eruption also should be considered. Rose spots (evanescent crops of pink macules, 2 to 3 mm in diameter, on the chest or abdomen) suggest typhoid fever. An eschar (black necrotic ulcer with erythematous margins) may be present in many rickettsial diseases. Patients with dengue fever, meningococcemia, and viral hemorrhagic fevers may present with petechiae, ecchymoses, or hemorrhagic lesions. |
| Eyes | The eyes should be examined for evidence of conjunctivitis (leptospirosis). |
| Sinuses, ears, teeth | Sinuses, ears, and teeth are common sites of occult infection (sinusitis, otitis media, tooth abscess); attention to these areas can help avoid unnecessary testing for travel-related causes of infections.9 |
| Heart, lungs | Auscultation of the lungs should focus on the detection of inspiratory crackles and wheezes, whereas auscultation of the heart should focus on detection of a murmur (subacute bacterial endocarditis).5 |
| Abdomen | Splenomegaly is associated with mononucleosis, malaria, visceral leishmaniasis, typhoid fever, and brucellosis. |
| Lymph nodes | The presence of localized or generalized lymphadenopathy may be diagnostically helpful (see Table 5). |
| Neurologic system | Fever and altered mental status in the returned traveler represent a medical emergency (see Table 6). |
| Localized lymphadenopathy |
| Bacterial infection: bartonellosis (cat-scratch disease), plague, staphylococcal infection, streptococcal infection, tuberculosis (scrofula), tularemia, typhus |
| Parasitic infection: African trypanosomiasis, American trypanosomiasis, filariasis, toxoplasmosis |
| Generalized lymphadenopathy |
| Bacterial infection: brucellosis, leptospirosis, melioidosis, secondary syphilis, tuberculosis, enteric fever |
| Viral infection: acute human immunodeficiency virus infection, dengue fever, hepatitis B, Lassa fever, measles, mononucleosis (Epstein-Barr virus, cytomegalovirus), rubella |
| Fungal infection: blastomycosis, coccidioidomycosis, histoplasmosis |
| Parasitic infection: visceral leishmaniasis |
| Noninfectious causes: malignancy (lymphoma, melanoma, metastatic carcinoma), rheumatoid arthritis, sarcoidosis, systemic lupus erythematosus, medications (phenytoin [Dilantin], carbamazepine [Tegretol], allopurinol [Zyloprim], sulfonamides) |
| Bacterial infection: acute bacterial meningitis, enteric fever, meningococcemia |
| Viral infection: Japanese encephalitis, rabies, yellow fever, viral hemorrhagic fevers (Ebola, Marburg, Lassa) |
| Parasitic infection: African trypanosomiasis, Plasmodium falciparum malaria |
LABORATORY TESTS
The initial laboratory evaluation should focus on diseases that are life-threatening, most importantly P. falciparum malaria and typhoid fever.9,10 Thick and thin blood films for malaria and blood cultures for typhoid fever are important initial tests in the evaluation of febrile travelers. If the first blood films are negative and malaria is still suspected, smears should be repeated every eight to 12 hours for several days.4–6,8,9,11
Other useful screening tests include a complete blood count with differential (paying close attention to eosinophilia), blood chemistries, liver-associated enzymes, and urinalysis with urine culture. Because most viral and rickettsial infections are diagnosed by demonstrating an antibody response, storing a tube of serum drawn when a patient is first evaluated (acute-phase sample) may provide the diagnosis when a sample is obtained at a later date (convalescent-phase sample) and the samples are compared.4,5,9
Major Tropical Causes of Fever
MALARIA
Malaria should be the first consideration in a febrile traveler who has returned from an endemic area. The disease is caused by a blood parasite that is transmitted by night-biting Anopheles mosquitoes. The worldwide distribution of malaria is shown in Figure 1. Updated information on malarious areas is available in the CDC's travel resource7 or on its travel Web site. Even brief exposures in endemic areas can put travelers at risk for malaria, as in cases of runway or airport malaria.14
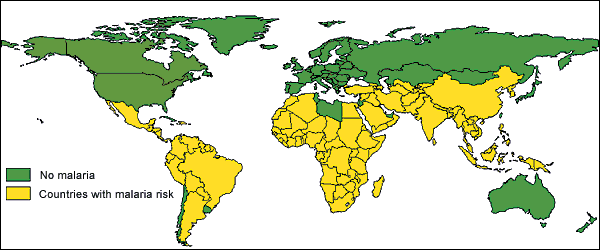
Of the four Plasmodium species that cause malaria in humans, P. falciparum results in the most serious illness. Approximately 90 percent of malaria cases originate in Africa, and up to 90 percent of travelers with P. falciparum infection become ill within two months of departure from a malarious area.4 Symptoms resulting from infection with other Plasmodium species may be delayed and can present months to years after international travel.4
A typical presentation consists of the abrupt onset of rigors followed by high fevers and diaphoresis.15,16 The patient also may have profound malaise, severe headaches, myalgias, and vague abdominal pain.15 Gastrointestinal symptoms of nausea, vomiting, and diarrhea may occur in up to 25 percent of patients and potentially can result in a delay of the diagnosis of malaria. Jaundice and hepatosplenomegaly may be noted on physical examination. Anemia, thrombocytopenia, leukopenia, and abnormal liver-associated enzymes often accompany clinical illness.15 Untreated P. falciparum infection can cause hypoglycemia, renal failure, pulmonary edema, and neurologic deterioration, leading to death.4
The physician must aggressively pursue the confirmation or exclusion of malaria by performing serial blood smears.11 A total of three smears should be obtained eight to 12 hours apart over the course of two days.4,9,11 Hospital admission is required for any patient with P. falciparum malaria or any patient in whom identification to the species level cannot be obtained. Consultation with an infectious diseases subspecialist is recommended to ensure proper antimalarial treatment.
ENTERIC FEVER
Enteric fever refers to a clinical syndrome caused by Salmonella typhi (typhoid fever) or, less commonly, Salmonella paratyphi (paratyphoid fever).11,17 Disease usually is acquired by direct fecal-oral spread or through fecal contamination of food or water. Typhoid fever is common in many developing nations, and travel to Mexico, India, the Philippines, Pakistan, El Salvador, and Haiti accounts for the majority of cases.
Following an incubation period of five to 21 days, patients with enteric fever usually present with sustained fever, anorexia, malaise, and vague abdominal discomfort. Although diarrhea may occur early, it often resolves before fever develops, and constipation is a usual complaint on presentation.4,17 Apulse-temperature dissociation and hepatosplenomegaly may be noted on physical examination.4 Rose spots are found in 30 to 50 percent of patients, but they are subtle and fleeting and must be searched for carefully. Laboratory findings are nonspecific.
Because current typhoid vaccines have an efficacy of approximately 70 percent, enteric fever is still a possibility in patients who have been immunized.18 [Evidence level C, consensus/expert guidelines] Diagnosis is achieved by isolation of the organism in cultures of blood, stool, urine, bone marrow, and duodenal aspirates.11
Resistance to chloramphenicol (Chloromycetin), ampicillin, and trimethoprim-sul-famethoxazole (Bactrim, Septra) is relatively common. Multidrug-resistant Salmonella strains usually are found in travelers who have been to the Indian subcontinent. Fluoroquinolone antibiotics are the treatment of choice.17 [Evidence level C, consensus/expert guidelines]
DENGUE FEVER
After an incubation period of three to 10 days, patients with dengue fever typically present with abrupt onset of fever, frontal headaches, and severe myalgias.19,22,23 Retroorbital pain, exacerbated by movement of the eyes, is a usual complaint.23 The typical rash associated with dengue fever is a macular or maculopapular erythroderma that blanches under light pressure.11,22,23 Leukopenia and mild thrombocytopenia are frequent but non-specific findings.23
The diagnosis of dengue fever is made clinically and can be confirmed by a fourfold rise in antibody titer between acute-and convalescent-phase serum samples obtained at least four weeks apart. Treatment consists of supportive measures.
LEPTOSPIROSIS
Leptospirosis, caused mainly by the spirochete Leptospira interrogans, infects animals in tropical and subtropical climates; the organism is then excreted in their urine.24 Transmission to humans occurs when leptospires enter the body through abraded skin, mucous membranes, or conjunctiva following contact with urine-contaminated soil or water.25 Exposure to the organism is occupational (e.g., gardening, farming) or recreational (e.g., swimming, rafting, crossing streams).24
Leptospirosis may occur as two clinically distinguishable syndromes. In anicteric leptospirosis (90 percent of cases), high fever, headaches, and myalgias follow a usual incubation period of seven to 12 days. Conjunctivitis, a maculopapular rash, and hepatosplenomegaly also may be noted. Later in the course of the illness, immune-mediated aseptic meningitis, uveitis, or chorioretinitis may occur.25 Icteric leptospirosis, or Weil's syndrome, is less common (10 percent of cases) and much more severe. It is characterized by fever, jaundice, azotemia, and hypotension.
The diagnosis of leptospirosis usually is established retrospectively by serologic tests. Blood, urine, and cerebrospinal fluid can be obtained for culture. Empiric therapy with penicillin or doxycycline (Vibramycin) should be initiated if the diagnosis is considered.24–26 Doxycycline is an effective prophylaxis for travelers to endemic areas who have a high risk of exposure.27 [Evidence level B, systematic review of lower quality controlled trials]
RICKETTSIAL INFECTIONS
Rickettsial diseases are vector-borne illnesses usually carried by ticks, lice, fleas, or mites, and are widely distributed throughout the world. The many agents that cause these diseases produce similar syndromes.
Mediterranean spotted fever, or boutonneuse fever, is the most common imported rickettsial disease in returning travelers.28,29 This disease is endemic in southern Europe and the Middle East, where it is caused by Rickettsia conorii. In sub-Saharan Africa, a similar illness, African tick typhus, is caused by Rickettsia africae.4,26
After a five- to seven-day incubation period, fever, headaches, and myalgias may occur. A maculopapular rash beginning on the trunk and spreading outward (as opposed to distal to proximal spread in Rocky Mountain spotted fever, the main rickettsial disease in the United States) to involve the palms, soles, and face may accompany the illness. There is usually an eschar, called a tâche noire (black spot), at the site of the tick bite4,11,26 (Figure 2).
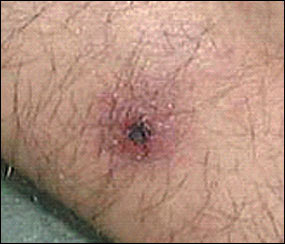
The diagnosis of rickettsial disease usually is confirmed with serologic testing. Doxycycline is the treatment of choice.