Pityriasis rosea is a common, acute exanthem of uncertain etiology. Viral and bacterial causes have been sought, but convincing answers have not yet been found. Pityriasis rosea typically affects children and young adults. It is characterized by an initial herald patch, followed by the development of a diffuse papulosquamous rash. The herald patch often is misdiagnosed as eczema. Pityriasis rosea is difficult to identify until the appearance of characteristic smaller secondary lesions that follow Langer's lines (cleavage lines). Several medications can cause a rash similar to pityriasis rosea, and several diseases, including secondary syphilis, are included in the differential diagnosis. One small controlled trial reported faster clearing of the exanthem with the use of erythromycin, but the mechanism of effect is unknown. Resolution of the rash may be hastened by ultraviolet light therapy but not without the risk of hyperpigmentation. Topical or systemic steroids and antihistamines often are used to relieve itching.
Pityriasis rosea is a common skin condition characterized by a herald patch and the later appearance of lesions arrayed along Langer's lines (cleavage lines). The condition is diagnosed most often in children and young adults. Several large case series from dermatology practices indicate that the incidence of pityriasis rosea peaks in persons 20 to 29 years of age, with no consistent gender predilection (Table 1).1–3
Etiology
Although the etiology of pityriasis rosea is unclear, several factors indicate an infectious cause. First, outbreaks of the condition occur in clusters, suggesting that an infectious agent is circulating within a community.4 Second, recurrence of pityriasis rosea outside the acute phase is rare, suggesting that there is long-lasting immunity after the infection. Third, up to 69 percent of patients with pityriasis rosea have a prodromal illness before the herald patch appears.5 Finally, some patients with pityriasis rosea show an increase in B lymphocytes, a decrease in T lymphocytes, and an elevated sedimentation rate.6
Unfortunately, even though electron microscopy shows some viral changes and possible viral particles, antibody and polymerase chain reaction tests for known viruses have failed to identify an offending virus. The results of one study7 showed elevated levels of human herpesvirus 7 in patients with pityriasis rosea. However, subsequent study results6,8,9 showed no consistent increase of human herpesvirus 7 levels in affected patients compared with control patients. Furthermore, human herpesvirus 7 infection is common in childhood, and the virus is prone to reactivation. Several other viruses have been examined, but none has been found to be causative.6
Chlamydia pneumoniae, Legionella pneumophila, and Mycoplasma pneumoniae also have been suggested as potential infectious agents in pityriasis rosea. However, the results of a small prospective case-control study10 did not show a significant rise in antibodies to these bacteria when affected patients were compared with matched control patients.
Diagnosis
Identification of pityriasis rosea can be challenging for a number of reasons. The diagnosis is unclear at the onset of symptoms, and there are no noninvasive tests that confirm the condition. In at least one half of patients, the first symptoms of pityriasis rosea are nonspecific and consistent with a viral upper respiratory infection.1,5 A herald patch then appears, typically on the trunk. This large lesion is commonly 2 to 10 cm in diameter, ovoid, erythematous, and slightly raised, with a typical collarette of scale at the margin (Figure 1). At this stage, however, the diagnosis usually remains unclear. Microscopic examination of potassium hydroxide preparations shows no fungal elements. The lesion cannot be differentiated from eczema and often is treated as such.
TABLE 1 Epidemiology of Pityriasis Rosea from Dermatology Practices
| Study | Site | Age range | Peak ages | Male-to-female ratio | Other |
|---|---|---|---|---|---|
| Cheong and Wong1 | Singapore | Not reported | 20 to 24 years | Male predominance | Peak incidence in March, April, and November |
| Harman, et al.2 | Turkey | 10 to 39 years (87 percent of affected patients) | 20 to 29 years | 1.0 to 1.2 | Peak incidence in rainy, snowy months |
| Tay and Goh3 | Singapore | Nine months to 82 years | 20 to 29 years | 1.2 to 1.0 | 17 percent had a herald patch; 6 percent had the inverse form (i.e., extremities affected but trunk spared)* |
*—It is likely that the percentage of patients with a herald patch is much higher in primary care settings, because patients with a herald patch and a clear diagnosis are unlikely to have been referred.
A few days to a few weeks after the appear ance of the herald patch, crops of smaller lesions, 5 to 10 mm in diameter, develop across the trunk and, less commonly, on the extremities. These lesions are salmon colored, ovoid, raised, and have the same collarette of scale as the herald patch (Figure 2). At this stage, the diagnosis usually is clear, particularly if the physician can observe or elicit a history of the herald patch.
FIGURE 1
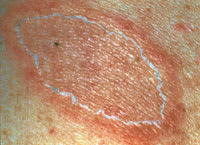
Herald patch with collarette of scale at the margin.
FIGURE 2
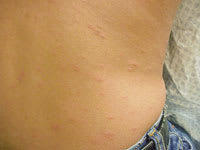
Classic pityriasis rosea of the lower abdomen with associated herald patch.
FIGURE 3
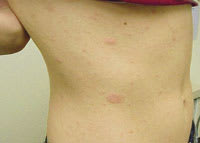
Typical oblong trunk lesions of pityriasis rosea.
If the diagnosis is uncertain, especially if the palms and soles are affected and the patient is sexually active, the physician should consider the possibility of secondary syphilis. Appropriate evaluation includes direct fluorescent antibody testing of lesion exudates, a VDRL test, or dark-field microscopy.11 Other conditions in the differential diagnosis include diffuse nummular eczema, tinea corporis, pityriasis lichenoides, guttate psoriasis, viral exanthem, lichen planus, and medication reaction.
The smaller secondary lesions of pityriasis rosea follow Langer's lines (Figure 3). When the lesions occur on the back, they align in a typical “Christmas tree” or “fir tree” pattern. Elsewhere on the body, the lesions follow the cleavage lines as follows: transversely across the lower abdomen and back, circumferentially around the shoulders, and in a V-shaped pattern on the upper chest12 (Figure 4). Pruritus is variable. Except for mild to severe itching in 25 percent of patients, no systemic symptoms typically are present during the rash phase of pityriasis rosea.
FIGURE 4
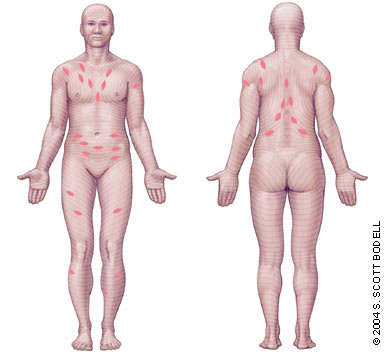
Lesions aligning along Langer's lines
Pityriasis rosea can occur in an inverse form in which the extremities are affected but the trunk is spared (Figure 5). Less commonly, pityriasis rosea occurs in a localized form, which makes the diagnosis more difficult. Gigantean (larger and fewer lesions), pustular, purpuric, or vesicular pityriasis rosea occurs in rare cases.13 At times, no herald patch is found. In one series,3 only 17 percent of patients referred to a dermatology clinic reported a herald patch; absence of a herald patch made the diagnosis more difficult and necessitated referral.
FIGURE 5
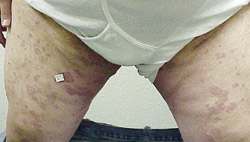
Inverse form of pityriasis rosea, with peripheral distribution.
Biopsy usually is not indicated in the evaluation of patients with suspected pityriasis rosea. Histology has shown that in addition to non-specific subacute and chronic inflammation, 55 percent of specimens contain epidermal cells that display dyskeratotic degeneration.14
Worsening of the rash or a second wave of lesions is not uncommon before eventual spontaneous resolution of the eruption. Recurrence of the condition later in life is rare.
Although no causal link has been established, multiple drugs have been associated with an extensive and often prolonged form of pityriasis rosea (Table 2).13,15–21 A review of the literature shows that single case reports account for most of the drug associations.
Treatment
The rash of pityriasis rosea typically lasts about five weeks and resolves by eight weeks in more than 80 percent of patients.1 An important goal of treatment is to control pruritus, which may be severe in 25 percent of patients.22 For patients with severe pruritus, experts have recommended treatment with zinc oxide, calamine lotion, topical steroids, oral antihistamines, and even oral steroids.13 [Evidence level C, expert opinion]
TABLE 2 Medications that May Cause Rashes Similar to Pityriasis Rosea
Few controlled trials on the treatment of pityriasis rosea have been conducted. The results of one prospective, blinded, placebo-controlled study of 90 patients showed complete clearance of the rash in 73 percent of patients who received two weeks of erythromycin therapy, compared with no clearance of the rash in patients who received placebo.5 [Evidence level B, single controlled trial] The study was not randomized, and allocation to groups was not concealed; therefore, the benefit may have been overestimated. Because studies have failed to identify an increase in antibody titers against Mycoplasma, Chlamydia, or Legionella species, the authors of the study speculate that the effect of erythromycin may be related to its anti-inflammatory properties.
Ultraviolet radiation, through artificial sources or intentional exposure to natural sunlight, has been recommended to decrease the duration of rash and intensity of itching in patients with pityriasis rosea. In a 1983 study,23 20 patients were treated unilaterally (one half of each patient's body served as the treatment side and the other half was left untreated) five times daily. Improvement of rash and itching was reported for 50 percent of the treated sides. However, a 1995 study24 that used the same unilateral approach in 17 patients found initial improvement in the appearance of the lesions, but no change in itching or overall patient status (severity of lesions or itching) on follow-up two and four weeks after the two-week treatment course. Therefore, the authors recommend treatment only for the purpose of achieving earlier lesion improvement in patients with extensive disease.24 [Evidence level B, inconsistent randomized controlled trials] Because post-inflammatory hyperpigmentation may occur with ultraviolet B radiation therapy, some experts recommend against its use.13
Patients should be advised about the self-limited nature of pityriasis rosea and about the need to contact their physician if the rash or pruritus lasts more than three months. Persistence beyond that time should prompt physicians to reconsider the original diagnosis, to consider biopsy to confirm the diagnosis, and to check for the use of medications that may cause a rash similar to that of pityriasis rosea.

