When used with a spermicide, the diaphragm can be a more effective barrier contraceptive than the male condom. The diaphragm allows female-controlled contraception. It also provides moderate protection against sexually transmitted diseases and is less expensive than some contraceptive methods (e.g., oral contraceptive pills). However, diaphragm use is associated with more frequent urinary tract infections. Contraindications to use of a diaphragm include known hypersensitivity to latex (unless the wide seal rim diaphragm is used) or a history of toxic shock syndrome. A diaphragm is fitted properly if the posterior rim rests comfortably in the posterior fornix, the anterior rim rests snugly behind the pubic bone, and the cervix can be felt through the dome of the device. The diaphragm should not be left in the vagina for longer than 24 hours. When the diaphragm is the chosen method of contraception, patient education is key to compliance and effectiveness. An extended visit with the physician or a nurse may be required for a woman to learn proper insertion, removal, and care of the diaphragm.
Methods of covering the cervix to prevent entry of sperm have been used for centuries. The Chinese employed disks made of oiled silk paper, while Europeans tried linen cloths and wax wafers.1 Half of a lemon rind, inserted into the vagina, was popular for a short time in Europe. Honey, pepper, and even animal dung were tried as spermicides or diaphragms.2 In the United States, Margaret Sanger was arrested in 1918 for distributing the first form of female-controlled contraception—a diaphragm made of galvanized rubber.
With the availability of oral contraceptive pills, use of the diaphragm has declined steadily over the past 40 years.2 However, when fitted properly and used correctly, the diaphragm remains an effective barrier contraceptive. The diaphragm may be the preferred method of contraception in women with contraindications to the use of oral contraceptive pills and women who do not wish to use an intrauterine device.
Effectiveness
Because the diaphragm is such a user-dependent form of contraception, reported rates for its effectiveness in preventing pregnancy range from 70 to 99 percent.3 The typical failure rate is 18 percent.3 Proper diaphragm fitting and patient education and motivation are key to compliance and success. When patients are properly trained and use the diaphragm consistently (i.e., perfect use), the failure rate is 6 percent.1–3 In other words, six pregnancies will occur in 100 women who use a diaphragm correctly with each act of sexual intercourse for one year. Over the same period, approximately 85 pregnancies will occur if no form of contraception is used.
Clinical practice has established the need for concurrent use of a spermicide with the diaphragm, although a recent Cochrane review noted that studies have failed to prove or disprove the contraceptive contribution of the spermicide.4 A recent trial5 found that nonoxynol–9 offers no protection against sexually transmitted diseases (STDs) and may increase the risk of human immunodeficiency virus (HIV) infection. The Centers for Disease Control and Prevention currently recommends that women at high risk for HIV infection not use nonoxynol–9 spermicides.6
Diaphragm users can combine contraceptive methods and theoretically achieve higher rates of effectiveness. For example, additional protection may be achieved through concurrent use of male condoms and a diaphragm with spermicide throughout the month.
Advantages and Disadvantages
Some advantages of the diaphragm over other forms of contraception are listed in Table 1.1,7–9 The prevention of STDs has been of particular interest in the past decade,7–9 and recent evidence indicates a lower rate of cervical neoplasia in some diaphragm users.10,11 [Reference 11—Evidence level B, case-control study] However, studies have not confirmed exact rates of STD prevention with diaphragm use. Notably, women who use the diaphragm typically are older, monogamous, more highly educated, and generally at lower risk for STDs compared with women who use other contraceptive methods or no form of contraception.
TABLE 1 Advantages of the Diaphragm
| Compared with OCPs and IUDs, the diaphragm provides moderate protection against sexually transmitted diseases.1,7–9 |
| Diaphragm use allows greater sexual spontaneity and more sexual sensation than male condom use.1 |
| Compared with OCPs, the diaphragm is safer for use in women with medical conditions and in women older than 35 years who smoke. |
| The diaphragm is less expensive than OCPs, IUDs, or sterilization. |
| The diaphragm is an easily reversible method of contraception, unlike tubal ligation or vasectomy. |
| Unlike the male condom, the diaphragm allows female-controlled contraception. |
| Diaphragm use requires one annual physician visit, compared with multiple visits for injectable hormonal contraceptive use. |
OCPs = oral contraceptive pills; IUDs = intrauterine devices.
One reported disadvantage of diaphragm use is an increased rate of urinary tract infections.12,13 The risk of toxic shock syndrome is 2.4 cases per 100,000 women using diaphragms, with the syndrome occurring almost exclusively when the device is left in place longer than 24 hours.14 Anatomic considerations sometimes prevent proper use of a diaphragm. Women with poor vaginal tone, a shallow vaginal shelf, rectocele, or cystocele may not be able to use the device effectively. A weight change of more than 6.8 kg (15 lb), pregnancy, or pelvic surgery may necessitate refitting.
Use of a diaphragm is contraindicated in women with hypersensitivity to latex (unless the wide seal rim diaphragm is used) or a history of toxic shock syndrome.
Diaphragm Types
Diaphragms are made of latex or silicone. Types of diaphragms include the latex arcing spring, coil spring, and flat spring, and the silicone wide seal rim15 (Figure 1).
The firmer rim of the arching spring diaphragm makes it the easiest type to insert. The diaphragm folds at two hinged points, forming an arc for insertion. The firm rim is easier to place in the posterior fornix. This diaphragm may be the best one to use in women with decreased pelvic tone, rectocele, or cystocele.
The coil spring diaphragm has a soft flexible rim that does not form an arc when folded. This diaphragm may be used in women with average vaginal muscle tone. It can be inserted with a diaphragm introducer tool.
The flat spring diaphragm is similar to the coil spring device but has a thinner, more delicate rim. Like the coil spring diaphragm, it can be inserted with a diaphragm introducer tool. The flat spring diaphragm may be used in women with firm vaginal muscle tone.
For women with hypersensitivity to latex, a silicone option is available. The wide seal rim diaphragm comes in an arcing spring style or a coil spring style. This silicone diaphragm is not available in pharmacies but must be ordered from the manufacturer.
Diaphragm Fitting
Diaphragm fitting rings (60 to 90 mm in size, with no dome) usually can be obtained from the manufacturer. However, the fitting procedure also may be performed using a series of fitting diaphragms, each with a hole in the middle of the dome. Examination gloves and vaginal lubricating gel are used during the fitting, but a sample of a spermicidal jelly may be shown to the patient as part of the instruction process.
FIGURE 1.
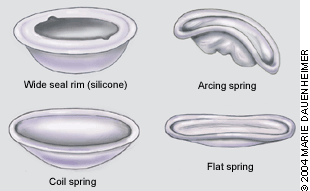
Types of diaphragms.
With the patient in the dorsal lithotomy position, the physician inserts the gloved index and middle fingers into the vagina, aiming for the posterior fornix. With the middle finger in the posterior fornix, the physician notes the spot where the bony inferior pubic arch touches the index finger. Before the fingers are removed from the vagina, some physicians mark this spot with their thumb or an instrument16 (Figure 2, top).
The fingers are kept in position as they are withdrawn from the vagina. The fitting ring or diaphragm then is placed with the rim over the end of the middle finger and the opposite side of the rim extended to the marked position on the index finger (Figure 2, bottom). Note that 75 mm is a common size.16
The physician lubricates the rim of the ring or fitting diaphragm and then folds the device in half so that the two sides of the rim are touching. Holding the vulva open with one hand, the physician uses the other hand to insert the folded diaphragm into the vagina and to direct placement toward the posterior fornix (Figure 3, left). To confirm posterior placement, the physician should check the location of the cervix through the dome of the fitting diaphragm or in the middle of the fitting ring (Figure 3, center). Finally, the anterior rim should be nudged so that it fits directly behind the pubic bone16 (Figure 3, right).
FIGURE 2.
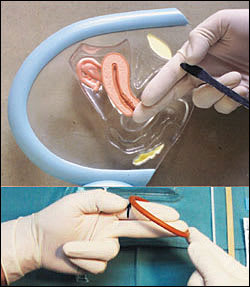
Diaphragm fitting using a fitting ring. (Top) The index and middle fingers are inserted into the vagina until the middle finger reaches the posterior fornix. The physician notes the spot where the bony inferior pubic arch touches the index finger. Before the fingers are removed from the vagina, this spot is marked with the thumb or an instrument. (Bottom) The diaphragm fitting ring is placed on the tip of the middle finger, and the opposite side of the rim is placed at the mark on the index finger. The diaphragm size is then determined (75 mm is a common size).
A diaphragm that is too small will not sit snugly behind the pubic bone. Consequently, the device will fall out with a Valsalva's maneuver or when the patient sits on the toilet or ambulates. If the diaphragm does not sit snugly, a larger size should be tried. If a diaphragm that is 5 to 10 mm larger does not fit, another fitting examination should be performed.
FIGURE 3.
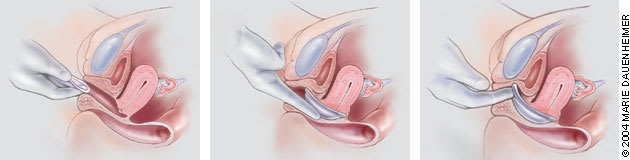
Insertion of fitting diaphragm. (Left) The lubricated diaphragm is folded and inserted at the introitus. (Center) The cervix should be palpable through the dome of the diaphragm. (Right) The anterior rim of the diaphragm is nudged so that it fits directly behind the pubic bone.
A diaphragm that is too big will rest improperly in front of the pubic bone. The patient generally will have discomfort, and the device may interfere with urination.
With a properly fitted diaphragm, the patient should not be able to feel anything inside her vagina, even when she walks or strains during defecation. The diaphragm is removed by “hooking” the anterior rim with the index finger and pulling the device down and out of the vagina.
Patient Education
Once proper fit is determined, the patient should practice diaphragm insertion and removal. The patient also should be instructed in the use of spermicidal jelly, preferably the placement of about one teaspoon of spermicide inside the cup of the diaphragm. For added efficacy and as a lubricant, spermicidal jelly also should be applied around the rim of the diaphragm. A well-instructed patient should be able to feel her cervix through the dome of the diaphragm and confirm anterior placement of the rim behind the pubic arch.
While proper size and fit of a diaphragm are important for effectiveness, correct use is even more important. The diaphragm can be inserted up to six hours to immediately before sexual intercourse. The diaphragm must remain in place for at least six hours after intercourse, but not for more than 24 hours (to reduce the risk of toxic shock syndrome). Douching should not be performed while the diaphragm is in place. If intercourse occurs more than once during the six hours that the diaphragm must remain in place, additional spermicidal jelly first should be inserted into the vagina. The diaphragm must be left in place while the spermicidal jelly is inserted.
When the diaphragm is removed, it should be washed with soap and water, rinsed and dried, and placed in its storage container. The diaphragm should be checked frequently for tears or holes. With good care and average use, diaphragms made of latex should last several years.

