Impaired glucose tolerance and impaired fasting glucose form an intermediate stage in the natural history of diabetes mellitus. From 10 to 15 percent of adults in the United States have one of these conditions. Impaired glucose tolerance is defined as two-hour glucose levels of 140 to 199 mg per dL (7.8 to 11.0 mmol) on the 75-g oral glucose tolerance test, and impaired fasting glucose is defined as glucose levels of 100 to 125 mg per dL (5.6 to 6.9 mmol per L) in fasting patients. These glucose levels are above normal but below the level that is diagnostic for diabetes. Patients with impaired glucose tolerance or impaired fasting glucose have a significant risk of developing diabetes and thus are an important target group for primary prevention. Risk factors for diabetes include family history of diabetes, body mass index greater than 25 kg per m2, sedentary lifestyle, hypertension, dyslipidemia, history of gestational diabetes or large-for-gestational-age infant, and polycystic ovary syndrome. Blacks, Latin Americans, Native Americans, and Asian-Pacific Islanders also are at increased risk for diabetes. Patients at higher risk should be screened with a fasting plasma glucose level. When the diagnosis of impaired glucose tolerance or impaired fasting glucose is made, physicians should counsel patients to lose 5 to 7 percent of their body weight and engage in moderate physical activity for at least 150 minutes per week. Drug therapy with metformin or acarbose has been shown to delay or prevent the onset of diabetes. However, medications are not as effective as lifestyle changes, and it is not known if treatment with these drugs is cost effective in the management of impaired glucose tolerance.
Type 2 diabetes mellitus is a major health problem that is associated with significant mortality and morbidity. Diabetes can be prevented or delayed through lifestyle interventions.1–3 Information that has been learned about the natural history and pathogenesis of diabetes indicates that this disease has a prolonged prediabetic phase.4 Consequently, family physicians have an opportunity to identify patients at high risk for developing diabetes and institute primary prevention strategies. Patients with impaired glucose tolerance (IGT) or impaired fasting glucose (IFG) are at significant risk for diabetes.5,6 Other risk factors for diabetes include family history of diabetes, body mass index (BMI) greater than 25 kg per m2, sedentary lifestyle, hypertension, dyslipidemia, history of gestational diabetes or large-for-gestational-age infant, and polycystic ovary syndrome. Blacks, Latin Americans, Native Americans, and Asian-Pacific Islanders also are at increased risk for diabetes.
Definitions and Epidemiology
An expert committee sponsored by the American Diabetes Association (ADA) has identified an intermediate group of patients who have blood glucose values that are higher than the defined normal level but not high enough to meet the diagnostic criteria for diabetes.7,8 This group includes patients with IGT or IFG. IGT is defined as two-hour 75-g oral glucose tolerance test values of 140 to 199 mg per dL (7.8 to 11.0 mmol per L); normal values on this test are below 140 mg per dL. IFG is defined as fasting plasma glucose values of 100 to 125 mg per dL (5.6 to 6.9 mmol per L); normal fasting glucose values are below 100 mg per dL (Table 1).8
Among U.S. adults 40 to 74 years of age, 15.6 percent (14.9 million) have IGT, and 9.7 percent (9.6 million) have IFG.9–11 IFG and IGT are metabolically distinct disorders, with limited overlap. Among those who had IFG and/or IGT, 16 percent had both IFG and IGT, 23 percent had IFG alone, and 60 percent had IGT alone.8,9
In an analysis of six prospective studies,6 the risk of developing diabetes was found to be approximately 3.6 to 8.7 percent per year in patients with IGT. Elevated fasting glucose levels, elevated two-hour postchallenge glucose values, and BMI greater than 27 kg per m2 were associated with the development of diabetes in these patients.5,6 Compared with normoglycemic persons, patients with IGT are at substantially greater risk of developing cardiovascular disease.12
IFG and IGT frequently are associated with metabolic syndrome. The Adult Treatment Panel III of the National Cholesterol Education Program13 has identified metabolic syndrome as a constellation of lipid and nonlipid risk factors for coronary artery disease. The syndrome is characterized by insulin resistance, atherogenic dyslipidemia (high triglyceride level, low high-density lipoprotein cholesterol level, and small, dense low-density lipoprotein cholesterol particles), hypertension, abdominal obesity, and prothrombotic and proinflammatory states. Metabolic syndrome is diagnosed when three or more of the risk factors in Table 213 are present.
It is important for family physicians to identify patients with metabolic syndrome and to intervene aggressively to reduce the risk of diabetes and macrovascular disease. In a prospective study14 conducted in Finland, cardiovascular disease and all-cause mortality were increased in men with metabolic syndrome even in the absence of cardiovascular disease and diabetes.
Women who have been diagnosed with gestational diabetes mellitus constitute another group at high risk for progression to diabetes. In a study of 1,636 women with recent gestational diabetes tested at one to four months postpartum, 14 percent were diagnosed with type 2 diabetes. It is recommended that women diagnosed with gestational diabetes undergo postpartum glucose testing at six weeks and every three years thereafter.15
Screening Recommendations
The American Academy of Family Physicians policy16 on screening for type 2 diabetes is consistent with the U.S. Preventive Services Task Force (USPSTF), which concludes that there is insufficient evidence to recommend for or against routine screening of asymptomatic adults for type 2 diabetes, IGT, or IFG.17 It has not been demonstrated that control of early diabetes as a result of screening provides additional benefit over waiting to treat until a clinical diagnosis has been made. The USPSTF does recommend screening for type 2 diabetes in adults with hypertension or hyperlipidemia. When these patients are specifically targeted for screening, the number needed to screen to prevent a cardiovascular event is lower than screening of the general population.17
The ADA acknowledges the lack of data from prospective studies on the benefits of screening for diabetes and therefore recommends using clinical judgment and patient preference as a guide. Based on expert opinion, selective screening is recommended (Table 3).18,19
The ADA also prefers to use the fasting plasma glucose level for screening, because it is faster, more acceptable to patients, and less expensive than other screening tests. The fasting plasma glucose level also is more reproducible than the 75-g oral glucose tolerance test and varies less between patients. However, the 75-g oral glucose tolerance test is more sensitive.18 The ADA recommends a screening interval of every three years in the absence of risk factors beyond age alone, whereas a shorter screening interval is recommended for persons at higher risk.18
No data are available to assess possible harms of diabetes screening. Potential harms include labeling patients as having a chronic illness, which may cause anxiety and make it difficult for them to obtain life or health insurance, and subjecting patients to the risks of long-term treatment with uncertain benefits.17
Pathogenesis
The progression from normal glucose tolerance to type 2 diabetes is characterized by dual defects that include insulin resistance and an insulin secretory defect caused by beta-cell dysfunction (Figure 1).4 Insulin resistance is characterized by decreased tissue sensitivity to insulin and marked compensatory hyperinsulinemia. Initially, plasma glucose levels are maintained in the normal range. In patients who will eventually develop diabetes, there is a decline in beta-cell secretory capacity.
The first glucose abnormality that is detected is a rise in the postprandial glucose levels because of reduced first-phase insulin secretion. With time, further decline in beta-cell function leads to elevation of the fasting glucose levels. Eventually, diabetes occurs, with more insulin secretory loss.20
FIGURE 1. Progression to Type 2 Diabetes
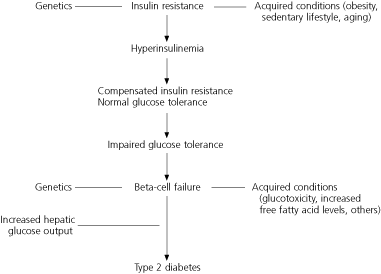
Etiologic sequence for the development of type 2 diabetes.
Adapted with permission from Ramlo-Halsted BA, Edelman SV. The natural history of type 2 diabetes. Implications for clinical practice. Prim Care 1999;26:773.
Lifestyle Interventions
Recent studies of patients with IGT have shown success for lifestyle interventions in delaying or preventing the development of diabetes (Table 4).1–3,21–23 There is strong evidence that a structured program of diet and exercise can reduce the risk of progression to type 2 diabetes in patients with IGT. Patients with IFG and IGT should be advised on the benefits of modest weight loss, good dietary habits, and regular physical activity.
DIABETES PREVENTION PROGRAM
In the Diabetes Prevention Program,1 3,234 patients with IGT and a BMI greater than 24 kg per m2 were randomly assigned to one of the following groups: placebo, metformin (Glucophage), or intensive lifestyle modification. The lifestyle modification group was offered a 16-lesson curriculum aimed at achieving a weight loss goal of more than 7 percent of their initial body weight through a low-calorie, low-fat diet and moderate-intensity exercise for 150 minutes per week.
TABLE 4 Summary of Major Diabetes Prevention Trials
| Study | Study population | Mean BMI (kg per m2) | Type of intervention | Relative risk reduction (%) | Approximate number needed to treat23 |
|---|---|---|---|---|---|
| Diabetes Prevention Program1 | 3,234 patients with IGT | 34 | Lifestyle modification | 58 | 7 for three years |
| Metformin (Glucophage), 850 mg twice daily | 31 | 14 for three years | |||
| Finnish Diabetes Prevention Study2 | 522 patients with IGT | 31 | Lifestyle modification | 58 | 22 for one year or 5 for five years |
| Da Qing IGT and Diabetes Study3 | 577 patients with IGT | 25.8 | Diet | 31 | 17 for six years |
| Exercise | 46 | 14 for six years | |||
| Diet plus exercise | 42 | 16 for six years | |||
| STOP-NIDDM trial21 | 1,429 patients with IGT | 31 | Acarbose (Precose), 100 mg three times daily | 24 | 10 for 3.3 years |
| TRIPOD study22 | 236 Hispanic women with a history of gestational diabetes | 30 | Troglitazone (Rezulin), 400 mg daily* | 55 | 15 for 2.5 years |
BMI = body mass index; IGT = impaired glucose tolerance; BMI = body mass index; STOP-NIDDM = Study to Prevent Non-Insulin-Dependent Diabetes Mellitus; TRIPOD = Troglitazone in the Prevention of Diabetes; FDA = U.S. Food and Drug Administration.
*—The U.S. Food and Drug Administration has withdrawn troglitazone from the market because of liver toxicity.
After an average follow-up of 2.8 years, there was a 58 percent relative risk reduction in the progression to diabetes in the lifestyle intervention group compared with the placebo group. The relative risk reduction was 31 percent in the metformin group compared with the placebo group. In this study, lifestyle intervention was effective in men and women, and in all ethnic groups. It was most beneficial in patients older than 60 years; in this group, it reduced the incidence of diabetes by about 71 percent.1
TABLE 5 Recommended Lifestyle Changes
| Daily calories | |
| Fat: <25 to 30 percent2,3 | |
| Saturated fat: <10 percent2 | |
| Carbohydrates: 50 to 60 percent2 | |
| Protein: 15 to 20 percent3 | |
| Daily fiber intake: >15 g for every 1,000 calories consumed2 | |
| Foods: salad,3 vegetables,3 fruits, whole grains,24 fish high in omega-3 fatty acids, legumes, lean meat; minimal intake of refined sugars3 | |
| Exercise*: moderate-intensity physical activity, such as brisk walking or biking, for 150 minutes per week1,2 | |
| Weight loss goal*: 5 to 7 percent of body weight1,2 | |
| Counseling by professionals on weight reduction and exercise with regular follow-up and reinforcement1,2 | |
*—The literature varies as to specific recommendations for weight loss and exercise.
FINNISH DIABETES PREVENTION STUDY
In the Finnish Diabetes Prevention Study,2 522 obese patients with a mean BMI of 31 kg per m2 and IGT were randomly assigned to a control group or an intervention group. Patients in the intervention group were instructed to lose 5 percent of their body weight, limit fat intake to less than 30 percent of daily calories, limit saturated fat intake to less than 10 percent of daily calories, increase fiber intake to at least 15 g per 1,000 calories, and exercise moderately for at least 150 minutes a week.
After 3.2 years of follow-up, there was a 58 percent relative risk reduction in the incidence of diabetes in the intervention group compared with the control group. There also was a significant positive correlation between the ability to achieve lifestyle modifications and preventing progression to diabetes.2
DA QING IGT AND DIABETES STUDY
In the Da Qing IGT and Diabetes Study in China, 577 patients with IGT were randomly assigned to a control group or to one of three treatment groups: diet alone, exercise alone, or diet plus exercise. Over six years of follow-up, the relative risk reduction in progression to diabetes was 31 percent in the diet group, 46 percent in the exercise group, and 42 percent in the combined group.3
Pharmacologic Agents
Although not as effective as lifestyle interventions, drug therapy with metformin and acarbose (Precose) has been shown to prevent the progression of IGT to diabetes.1,21 Success also has been achieved with troglitazone (Rezulin),22 although the U.S. Food and Drug Administration has withdrawn this drug from the market because of liver toxicity.
In the Diabetes Prevention Program, treatment with metformin was associated with 31 percent relative reduction in the progression of diabetes in patients with IGT.1 Metformin was more effective in younger patients with a higher BMI and higher fasting plasma glucose levels, and was least effective in patients older than 60 years.
In the Study to Prevent Non-Insulin-Dependent Diabetes Mellitus (STOP-NIDDM), patients with IGT who were treated with acarbose showed 25 percent relative reduction in progression to diabetes.21 This risk reduction did not persist when acarbose therapy was discontinued at the end of the study. The incidence of diabetes increased in the group that originally had been treated with acarbose, indicating that this drug therapy must be continued to maintain preventive effects.
Final Comments
Lifestyle interventions (Table 5)1–3,24 can be difficult to implement because it is impractical for the usual family practice systems to provide intensive dietary and exercise interventions similar to those used in clinical trials. However, lifestyle interventions are highly effective and superior to drug therapy, and should be the first choice in treating patients with IGT or IFG.
Although pharmacologic agents have been shown to be successful in preventing or delaying the onset of diabetes, whether these agents can prevent complications of diabetes or protect against cardiovascular disease remains unknown. Routine use of pharmacologic agents as a substitute for lifestyle modification should be discouraged until more studies have been conducted and the cost-effectiveness of drug therapy has been assessed. A healthier lifestyle can modify other risk factors for cardiovascular disease such as obesity, hypertension, and dyslipidemia. Drug therapy can be considered when aggressive lifestyle interventions are unsuccessful.9
Strength of Recommendations
| Key clinical recommendation | Strength of recommendation | References |
|---|---|---|
| It is recommended that women diagnosed with gestational diabetes undergo postpartum glucose testing at six weeks and every three years thereafter. | C | 15 |
| The U.S. Preventive Services Task Force recommends screening fortype 2 diabetes in adults with hypertension or hyperlipidemia. | B | 17 |
| There is strong evidence that a structured program of diet and exercisecan reduce the risk of progression to type 2 diabetes in patients with impaired glucose tolerance. | A | 2,3,4 |

