Clostridium difficile infection is responsible for approximately 3 million cases of diarrhea and colitis annually in the United States. The mortality rate is 1 to 2.5 percent. Early diagnosis and prompt aggressive treatment are critical in managing C. difficile–associated diarrhea. Major predisposing factors for symptomatic C. difficile colitis include antibiotic therapy; advanced age; multiple, severe underlying diseases; and a faulty immune response to C. difficile toxins. The most common confirmatory study is an enzyme immunoassay for C. difficile toxins A and B. The test is easy to perform, and results are available in two to four hours. Specificity of the assay is high (93 to 100 percent), but sensitivity ranges from 63 to 99 percent. In severe cases, flexible sigmoidoscopy can provide an immediate diagnosis. Treatment of C. difficile–associated diarrhea includes discontinuation of the precipitating antibiotic (if possible) and the administration of metronidazole or vancomycin. Preventive measures include the judicious use of antibiotics, thorough hand washing between patient contacts, use of precautions when handling an infected patient or items in the patient’s immediate environment, proper disinfection of objects, education of staff members, and isolation of the patient.
Clostridium difficile is a gram-positive, spore-forming rod that is responsible for 15 to 20 percent of antibiotic-related cases of diarrhea and nearly all cases of pseudomembranous colitis.1 The species was named “difficile” because initially it was hard to culture.2 Early studies showed that C. difficile could be isolated from the gastrointestinal tracts of most neonates; thus, it was believed to be a commensal organism. In the late 1970s, however, C. difficile was found to be the primary cause of pseudomembranous colitis.3 Because of the frequent use of broad-spectrum antibiotics, the incidence of C. difficile diarrhea has risen dramatically in recent decades.4,5
C difficile–associated diarrhea often is perceived to be an occasional and easily treated side effect of antibiotic therapy. Research has shown, however, that C. difficile infection accounts for considerable increases in the length of hospital stays and more than $1.1 billion in health care costs each year in the United States.5 The condition is a common cause of significant morbidity and even death in elderly or debilitated patients.
Family physicians should stress preventive measures for C. difficile–associated diarrhea (especially the judicious use of antibiotics) and should maintain a high index of suspicion for C. difficile infection in their patients.
Strength of Recommendations
| Key clinical recommendation | Label | References |
|---|---|---|
| Test for Clostridium difficile toxin in patients with community-acquired or traveler’s diarrhea who have had antibiotics or chemotherapy in recent weeks. | B | 20 |
| Test for C. difficile toxin in patients with nosocomial diarrhea beginning three or more days after admission to the hospital. | B | 20 |
| If necessary, rapid diagnosis of C. difficile–associated diarrhea can be made by flexible sigmoidoscopy or abdominal computed tomography. | C | 4 |
| In patients with confirmed C. difficile infection, the offending antibiotic should be withdrawn. | B | 20 |
| The recommended antibiotic is metronidazole (Flagyl) in a dosage of 250 mg orally four times per day or 500 mg orally three times per day for 10 to 14 days. | A | 20 |
A = consistent, good-quality, patient-oriented evidence; B = inconsistent or limited-quality, patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, opinion, or case series. See page 835 for more information.
Illustrative Case
An 87-year-old white woman was readmitted to the hospital because of recurrent pneumonia. Ten days earlier, she had been treated for right lower lobe pneumonia at another institution. She was discharged on moxifloxacin (Avelox) and doxycycline (Vibramycin). She returned home, where she was recovering, but then she became weak, short of breath, and constipated.
On readmission, chest radiography revealed right lower lobe pneumonia. The patient’s white blood cell (WBC) count was 16,300 per mm3 (16.3 × 109 per L), compared with 7,500 per mm3 (7.5 × 109 per L) during her previous hospitalization.
At the time of readmission, the patient was afebrile, and her vital signs were stable. Moxifloxacin and doxycycline were continued. Overnight, however, the patient became febrile, and her WBC count rose to 36,000 per mm3 (36 × 109 per L). She also passed several loose stools. In light of the patient’s recent history of antibiotic use, the sudden leukocytosis, her age and frail condition, and her recent hospital admission, C. difficile–associated diarrhea was considered, and a stool sample was obtained for analysis.
Empiric treatment with oral metronidazole (Flagyl) and famotidine (Pepcid) was initiated. Shortly thereafter, the patient developed marked hypotension. Fluid boluses produced no improvement, and a dopamine (Intropin) drip was started. Moxifloxacin and doxycycline were discontinued, and treatment with oral vancomycin (Vancocin), intravenous metronidazole, and intravenous ceftizoxime (Cefizox); (anti–C. difficile–associated diarrhea therapy) was started.
Sigmoidoscopy revealed diffuse pseudomembranes throughout the patient’s distal colon, confirming C. difficile infection (Figure 1). An abdominal computed tomographic (CT) scan was consistent with this diagnosis. Ceftizoxime was discontinued, and total parenteral nutrition was initiated.
Figure 1.
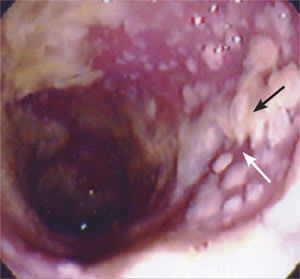
Irregular yellow plaques of necrotic debris (black arrow) with intervening edematous bowel mucosa (white arrow) in an 87-year-old woman. These findings are consistent with pseudomembranes caused by Clostridium difficile infection.
Used with permission from Kaiser Permanente, Fontana, Calif.
Over the next eight days, the patient’s condition continued to decline as she became more acidotic, her urine output diminished, her mental status fluctuated, and her abdomen became grossly distended and tympanic. Subtotal colectomy and ileostomy were considered, but were refused by the patient and her husband.
After more than a week of severe illness, the patient began to improve. Eventually, she was transferred to a skilled nursing facility for rehabilitation.
Epidemiology
Each year, C. difficile infection results in approximately 3 million cases of diarrhea and colitis in the United States. The case mortality rate is approximately 1 to 2.5 percent. Until recently, C. difficile infection was thought to result from an overgrowth of commensal organisms in the colon; however, studies have shown that fewer than 3 percent of adults carry this pathogen.1
Acquisition of C. difficile occurs primarily in the hospital setting, where the organism has been cultured from bed rails, floors, windowsills, and toilets, as well as the hands of hospital workers who provide care for patients with C. difficile infection (Table 1).4,6 The organism can persist in hospital rooms for up to 40 days after infected patients have been discharged.1
TABLE 1 Risk Factors for Clostridium difficile–Associated Diarrhea
| Admission to intensive care unit |
| Advanced age |
| Antibiotic therapy |
| Immunosuppressive therapy |
| Multiple and severe underlying diseases |
| Placement of a nasogastric tube |
| Prolonged hospital stay |
| Recent surgical procedure |
| Residing in a nursing home |
| Sharing a hospital room with a |
| C. difficile–infected patient |
| Use of antacids |
The rate of C. difficile acquisition is estimated to be 13 percent in patients with hospital stays of up to two weeks and 50 percent in those with hospital stays longer than four weeks.7 Patients who share a room with a C. difficile–positive patient acquire the organism after an estimated hospital stay of 3.2 days, compared with a hospital stay of 18.9 days for other patients.2
Pathophysiology
The precipitating event for C. difficile colitis is disruption of the normal colonic microflora. This disruption usually is caused by broad-spectrum antibiotics (Figure 2),1,5,8–11 with clindamycin (Cleocin) and broad-spectrum penicillins and cephalosporins most commonly implicated.8 Antibiotics with a reduced propensity to induce infection include aminoglycosides, metronidazole, antipseudomonals, and vancomycin.8 The risk of developing antibiotic-associated diarrhea more than doubles with longer than three days of antibiotic therapy (risk ratio: 2.28).12
Figure 2. Pathogenesis of Clostridium difficile Infection.
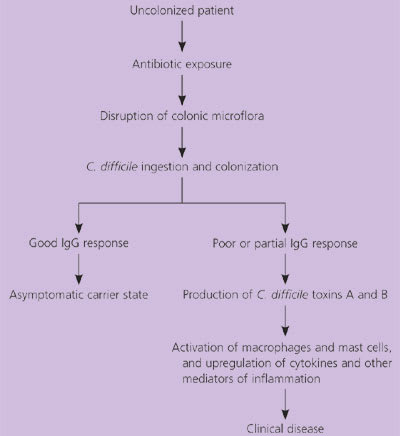
Pathogenesis of Clostridium difficile infection.
Information from references 1, 5, and 8 through 11.
After disruption of the colonic microflora, colonization of C. difficile generally occurs through the ingestion of heat-resistant spores, which convert to vegetative forms in the colon.9 Depending on host factors, an asymptomatic carrier state or clinical manifestations of C. difficile colitis develop.9 Manifestations of the disease range from mild diarrhea to life-threatening C. difficile colitis. C. difficile–associated diarrhea can occur up to eight weeks after the discontinuation of antibiotics.1 Most cases of C. difficile infection occur on days 4 through 9 of antibiotic therapy.9
The major host factors predisposing patients to the development of symptomatic C.difficile–associated diarrhea include antibiotic therapy, advanced age, number and severity of underlying diseases, and faulty immune response to C. difficile toxins (Table 1).4,6 Patients at highest risk for fulminant disease include those who recently received immunosuppressive therapy or recently underwent surgical procedures, and those with a history of C. difficile–associated diarrhea.4 The increased risk may be due partly to the debilitated patient’s inability to mount an IgG antibody immune response against C. difficile toxin A.5 The ability to mount an immune response is not protective against C. difficile colonization, but it is associated with decreased morbidity, mortality, and recurrence of C. difficile–associated diarrhea.10,13
C. difficile causes toxin-mediated colitis. Pathogenic strains of C. difficile produce two protein exotoxins: toxin A and toxin B.1 Toxin A activates macrophages and mast cells. Activation of these cells causes the production of inflammatory mediators, which leads to fluid secretion and increased mucosal permeability.1 Toxin B has little enterotoxic activity but is extremely cytotoxic in vitro. C. difficile toxins also cause leukocyte chemotaxis and the upregulation of cytokines and other inflammatory mediators. Consequently, there is a profound colonic inflammatory response, which is evidenced clinically by a high WBC count.1 As colitis worsens, focal ulcerations occur, and the accumulation of purulent and necrotic debris forms the typical pseudomembrane.9
Diagnosis
The diagnosis of C. difficile–associated diarrhea requires a careful history, with particular emphasis on antibiotic use during the preceding three months (Figure 3).18 The clinical presentation ranges from no symptoms to fulminant pseudomembranous colitis. A detailed description of the patient’s diarrhea, including color, consistency, and frequency, is important in differentiating other causes of diarrhea from C. difficile–associated diarrhea. Other important factors include a history of fever, immunosuppression, a recent surgical procedure, previous infection with C. difficile, recent change in bowel habits, and the presence of abdominal symptoms (Table 1).4,6 A selected differential diagnosis is provided in Table 2.6
Figure 3. Diagnosis and Treatment of Clostridium difficile–Associated Diarrhea
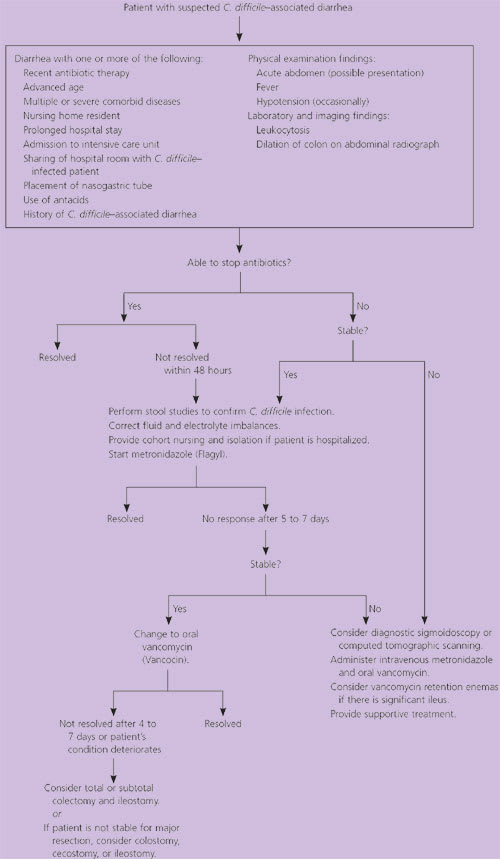
Suggested algorithm for the diagnosis and treatment of Clostridium difficile–associated diarrhea.
Adapted with permission from Viswanath YK, Griffiths CD. The role of surgery in pseudomembranous enterocolitis. Postgrad Med J 1998;74:216–9.
TABLE 2 Selected Differential Diagnosis of Clostridium difficile–Associated Diarrhea
| Disorder | Typical presentation |
|---|---|
| C. difficile–associated diarrhea | Recent history of antibiotic use, evidence of colitis, fecal leukocytes, fever; may not resolve with discontinuation of antibiotics; diarrhea typically watery, may be florid |
| Antibiotic intolerance | History of antibiotic intolerance, no evidence of colitis; resolves with antibiotic withdrawal |
| Infectious enteritis or colitis (diarrhea not associated with C. difficile): bacterial gastroenteritis, viral gastroenteritis, amebic dysentery | History of travel, camping, infectious contacts, or day care attendance; associated with nausea and vomiting |
| Inflammatory bowel disease: Crohn’s disease, ulcerative colitis | Bloody diarrhea, abdominal pain, nausea, vomiting, loss of appetite, family history |
| Ischemic colitis | History of vascular disease; pain associated with eating |
Information in part from reference 6.
The most common laboratory test for diagnosing C. difficile–mediated disease is an enzyme immunoassay that detects toxins A and B. This test provides results within two to six hours and has a specificity of 93 to 100 percent. The sensitivity is 63 to 99 percent, which means that false-negative results can occur.13–16 However, the majority of combination enzyme immunoassays have a sensitivity of 85 to 95 percent.8 The immunoassay should not be used as an indicator of response to therapy because results remain positive for extended periods in 25 percent of successfully treated patients.8
The gold standard for the diagnosis of C. difficile–mediated disease is a cytotoxin assay. Although this test is highly sensitive and specific, it is difficult to perform, and results are not available for 24 to 48 hours.
C. difficile can be cultured. However, culture is not specific for pathogenic toxin-producing C. difficile strains and, therefore, is not clinically helpful except for strain typing in outbreaks of nosocomial infection.9
A combination immunoassay that is currently in development tests for C. difficile–specific glutamate dehydrogenase (sensitivity: 97 percent) and toxins A and B (specificity: 97 to 99 percent). The advantages of this test are same-day turnaround and a high negative predictive value.16
An important indicator of impending fulminant colitis is a sudden rise in the peripheral WBC count to between 30,000 and 50,000 per mm3 (30 to 50 × 109 per L), often accompanied by significant bandemia.4,17 Because progression to shock can occur even in patients who have had relatively benign symptoms for weeks, early warning signs such as the leukemoid reaction are invaluable.4
In patients with fulminant C. difficile–associated diarrhea, flexible sigmoidoscopy can provide an immediate diagnosis.4,13 The finding of pseudomembranes is pathognomonic for C. difficile colitis (Figure 1). CT scanning also can diagnose fulminant disease quickly. When considered with the clinical history, the presence of ascites, colon wall thickening, or dilation can help categorize the severity of the colitis.4
Treatment
The treatment of C. difficile–associated diarrhea depends on the clinical presentation (Figure 3).18 In otherwise healthy adults, the first step is to discontinue the precipitating antibiotic, if possible, and administer fluids and electrolytes to maintain hydration. With this conservative therapy, diarrhea can be expected to resolve in 15 to 23 percent of patients.19
Specific pharmacotherapy for C. difficile–associated diarrhea should be initiated in older patients, patients with multiple medical problems, and patients in whom antibiotics need to be continued. Specific treatment also should be initiated if diarrhea persists despite discontinuation of the precipitating antibiotic or if there is evidence of colitis (i.e., fever, leukocytosis, characteristic findings of colitis on CT scanning or endoscopy).6 Use of opiates and antidiarrheal medications should be avoided or minimized because decreased intestinal motility can exacerbate toxin-mediated disease.
First-line therapy consists of metronidazole, 500 mg orally three or four times daily for 10 to 14 days.20,21 Metronidazole is an inexpensive drug with a greater than 90 percent positive response rate21 (Table 3).13,19–21
TABLE 3 Treatment of Clostridium difficile Colitis
| Drug | Dosage | Mode of administration | Efficacy | Side effects | Price* | Advantages and disadvantages |
|---|---|---|---|---|---|---|
| Metronidazole (Flagyl) | 500 mg orally every six to eight hours for 10 to 14 days Alternatives: 250 mg every six hours for 10 to 14 days and 500 mg IV every eight hours for 10 to 14 days | Oral and IV | > 90% | Nausea, vomiting Metallic taste Potentiation of warfarin (Coumadin) Disulfiram-like reaction | $256 (36 to 40): 500 mg orally every six hours for 14 days $678 (103 to 644): 500 mg IV every eight hours for 14 days | Effective by IV administration Less expensive than vancomycin More side effects than vancomycin |
| Vancomycin (Vancocin) | 125 to 500 mg orally every six hours for 10 to 14 days | Oral only† Nasogastric tube Retention enema | > 90% | Minimal; can include unpleasant taste, mouth irritation, nausea or vomiting; rarely, rash | $1,724: 500 mg orally every six hours for 14 days | Safe for use in pregnant women High cost Use may lead to resistance |
| Vancomycin retention enemas | Uncertain | Variable |
IV = intravenous.
*—Estimated cost to the pharmacist based on average wholesale prices in Red book. Montvale, N.J.: Medical Economics Data, 2004. Cost to the patient will be higher, depending on prescription filling fee.
†—Vancomycin is not secreted into the bowel; therefore, IV administration is not effective.
Vancomycin also is an effective treatment, with a response rate of greater than 90 percent.21 If a patient is pregnant or does not respond to or tolerate metronidazole, vancomycin should be initiated in a dosage of 125 to 500 mg orally four times daily for 10 to 14 days.6
Response to therapy can be assessed by the resolution of fever, usually within the first two days. Diarrhea should resolve within two to four days. Treatment is continued for 10 to 14 days. Therapeutic failure is not determined until treatment has been given for at least five days.19
Twenty to 25 percent of patients with C. difficile infection will have recurrent infection.6,9 Recurrence seldom is caused by treatment-resistant strains; usually, it is due to the germination of persistent C. difficile spores in the colon after treatment or to reinfection because of reingestion of the pathogen.9 Management of recurrent C. difficile infections remains controversial, although most relapses respond to another course of antibiotics given in standard dosages for 10 to 14 days. Up to 5 percent of patients have more than six recurrences.6
In patients with recurrent C. difficile infection, enemas containing human stool have been used to restore normal microflora in the colon. This approach has had good response rates; however, the enemas are unwieldy to perform, and there is a risk of transmitting retroviruses or other infectious agents.22 One potential benefit of enemas is a decrease in the use of vancomycin for recurrent C. difficile infections and therefore a theoretical decrease in the emergence of vancomycin-resistant bacteria.
Approximately 3 percent of patients develop severe C. difficile–associated diarrhea. The mortality rate in these patients ranges from 30 to 85 percent.19 Initial treatment of severe cases must be aggressive, with intravenous metronidazole and oral vancomycin used in combination.11 If ileus occurs, vancomycin can be administered by nasogastric tube with intermittent clamping, retention enemas, or both.23 If medical therapy fails or perforation or toxic megacolon develops, surgical intervention with colectomy and ileostomy is indicated but carries a high mortality rate.4,9,18
Prevention
Prevention of C. difficile infection is challenging. Established guidelines should be followed to minimize exposure to the pathogen, particularly in debilitated patients. Preventive measures include the judicious use of antibiotics, hand washing between patient contacts, rapid detection of C. difficile by immunoassays for toxins A and B, isolation of patients who have C. difficile–associated diarrhea, use of precautions when in contact with the patient and surrounding environment, proper disinfection of objects (e.g., sodium hypochlorite, alkaline glutaraldehyde, ethylene oxide), education of staff members, and use of continued precautions until the diarrhea ceases.1,6
A multidiscipline antibiotic management program to restrict the inappropriate use of antibiotics (e.g., third-generation cephalosporins) can lead to a significant decrease in nosocomial infections caused by C. difficile.24 In particular, restriction of clindamycin use has been shown to decrease the incidence of C. difficile–associated diarrhea.25 Family physicians can do much to decrease the occurrence of C. difficile–mediated disease by restricting the use of broad-spectrum antibiotics in their patients.
Probiotic use is a more controversial mode of prevention. Lactobacilli have been shown to reduce the incidence of antibiotic-associated diarrhea, but have not been proven to decrease the incidence of C. difficile–associated diarrhea.26 Anecdotally, many physicians report success with lactobacilli and use this preventive measure routinely, especially in patients at higher risk for severe disease.

