
Am Fam Physician. 2005;71(8):1563-1570
Each year, pacemaker therapy is prescribed to approximately 900,000 persons worldwide. Current pacemaker devices treat bradyarrhythmias and tachyarrhythmias and, in some cases, are combined with implantable defibrillators. In older patients, devices that maintain synchrony between atria and ventricles are preferred because they maintain the increased contribution of atrial contraction to ventricular filling necessary in this age group. In general, rate-responsive devices are preferred because they more closely simulate the physiologic function of the sinus node. Permanent pacemakers are implanted in adults primarily for the treatment of sinus node dysfunction, acquired atrioventricular block, and certain fascicular blocks. They also are effective in the prevention and treatment of certain tachyarrhythmias and forms of neurocardiogenic syncope. Biventricular pacing (resynchronization therapy) recently has been shown to be an effective treatment for advanced heart failure in patients with major intraventricular conduction effects, predominately left bundle branch block. Many studies have documented that pacemaker therapy can reduce symptoms, improve quality of life and, in certain patient populations, improve survival.
Since the first permanent pacemaker was implanted in 1958, device therapy has continued to grow. It is estimated that more than 300,000 patients in the United States receive a permanent pacemaker each year, and about 900,000 pacemakers are implanted worldwide. The indications for pacemaker therapy have expanded in the past 45 years and now include the treatment of bradyarrhythmias and the electrical therapy of tachyarrhythmias, certain types of syncope, and advanced heart failure. Device technology also has evolved from simple single-chamber, fixed-rate pacemakers to multichamber, rate-responsive (to meet physiologic needs) units capable of pacing, cardioversion, and defibrillation. In specific populations, clinical studies have demonstrated improvement of patient survival with the use of these implantable devices.
The decision to implant a pacemaker usually is based on symptoms of a bradyarrhythmia or tachyarrhythmia in the setting of heart disease. Symptomatic bradycardia is the most common indication. It has been defined as a “documented bradyarrhythmia that is directly responsible for the development of the clinical manifestations of frank syncope or near syncope, transient dizziness or light-headedness, and confusional states resulting from cerebral hypoperfusion and attributable to low heart rates.”1 Other symptoms that may result from severe bradycardia include fatigue, reduced exercise capacity, and frank congestive heart failure. Physiologic sinus bradycardia, which can occur in highly trained athletes, must be excluded and should not be confused with pathologic bradyarrhythmias.
ACC/AHA/NASPE Recommendations
The American College of Cardiology (ACC) and the American Heart Association (AHA) published the first clinical guideline for permanent pacemaker implantation in 1984. Three subsequent revisions have been published, the latest being the 2002 update in collaboration with the North American Society of Pacing and Electrophysiology (NASPE), on which this article is based.1 The selected recommendations are presented in the usual three-class ACC/AHA format (Table 1).1
ACQUIRED ATRIOVENTRICULAR AND FASCICULAR BLOCKS IN ADULTS
Atrioventricular (AV) block is classified as first-, second-, or third-degree. First-degree AV block is defined as an abnormally prolonged PR interval. Second-degree, Mobitz type I AV block (Wenckebach) is manifested by progressive prolongation of the PR interval eventuating in a dropped QRS complex. It usually is associated with a narrow QRS complex. Second-degree, Mobitz type II AV block demonstrates a constant PR interval before a dropped QRS and usually is associated with a wide QRS complex. Advanced AV block refers to blockage of two or more consecutive P waves, whereas complete (third-degree) AV block is defined as absence of all atrioventricular conduction.
First-degree and type I second-degree AV block usually are caused by delayed conduction in the AV node irrespective of QRS duration. Type II second-degree AV block usually is infra-nodal, especially when the QRS is wide. Third-degree AV block may occur at any anatomic level.
The decision to implant a pacemaker in a patient with abnormal AV conduction depends on the presence of symptoms related to bradycardia or ventricular arrhythmias and their prognostic implications. Observational studies over the years strongly suggest that permanent pacing improves survival in patients with complete AV block, especially if syncope has occurred.2–5 Therefore, symptomatic third-degree AV block is a class I indication for permanent pacing, whereas asymptomatic third-degree AV block is a class IIa indication. More recently, it has been recognized that type II second-degree AV block may be a precursor to complete AV block.6,7 Type II second-degree AV block should be treated with a permanent pacemaker even in an asymptomatic patient, particularly if it is associated with fascicular block, which also is a class IIa recommendation.
Marked first-degree AV block (PR greater than 300 ms) can produce symptoms even in the absence of higher degrees of block. In this situation, symptoms are caused by hemodynamic impairment resulting from the inappropriate timing of atrial systole, which may produce symptoms similar to those of the “pacemaker syndrome” (i.e., fatigue, syncope or presyncope, and malaise secondary to low cardiac output, hypotension, and elevated ventricular filling pressures that may develop when ventricular pacing results in improper timing of atrial and ventricular systole).8,9 Small, observational studies have suggested that patients with left ventricular dysfunction and markedly prolonged PR interval may benefit from pacing with a shorter AV conduction time.10
Fascicular block refers to electrocardiographic (ECG) evidence of impaired conduction below the AV node in the right bundle branch or in one or both fascicles of the left bundle branch. Bifascicular block refers to impaired conduction in two of the three fascicles; for example, right bundle branch block and a block in the left anterior-superior division of the left bundle branch. Trifascicular block is diagnosed when block is documented in all three fascicles, whether simultaneously or at different times. First-degree AV block in combination with bifascicular block fulfills the criteria of trifascicular block, as does “alternating bundle branch block.” Alternating (bilateral) bundle branch block is diagnosed when in different leads of the ECG or on successive ECG tracings there is clear evidence of block in all three fascicles; for example, right bundle branch block in one lead and left bundle branch block in another (Figure 1).
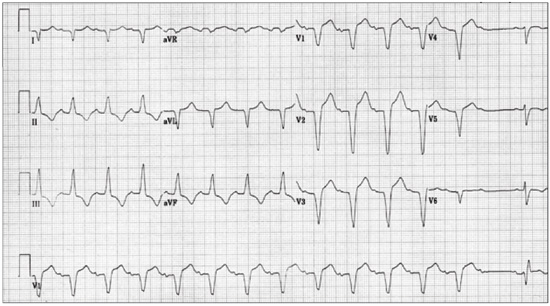
Recurrent syncope is common in patients with bifascicular block, but it is not associated with an increased incidence of sudden death.11,12 Pacing therapy in such patients often relieves the transient neurologic symptoms (syncope) but does not prevent sudden death.13 On the other hand, syncope in the presence of permanent or transient third-degree AV block is associated with an increased incidence of sudden death, and pacing therapy definitely is indicated. Similarly, if the cause of syncope in the presence of bifascicular or trifascicular block cannot be determined with certainty, prophylactic pacing is indicated.14 Pacing is not indicated for asymptomatic bifascicular block because the rate of progression to more advanced degrees of block is very slow.
Some investigators recommend pacing therapy for asymptomatic bifascicular block if there is evidence of severe intraventricular conduction delay on electrophysiologic study. Patients with alternating bundle branch block have a high mortality rate and a significant incidence of sudden death if pacing therapy is not provided.15
Before a decision is made to implant a pacemaker for AV block, it is important to exclude and correct reversible causes such as electrolyte abnormalities. Some types of AV block can be expected to resolve and do not require pacing therapy; for example, perioperative block caused by hypothermia or inflammation near the AV junction as a result of a surgical procedure in that area, or AV block developing during the acute phase of Lyme disease. Conversely, prophylactic pacing is recommended in certain conditions even if the AV block is transient, because of the possibility of disease progression (for example, in sarcoidosis, amyloidosis, and certain neuromuscular diseases). The principal recommendations for pacing in these conditions are listed in Tables 2 and 3.1
SINUS NODE DYSFUNCTION (SICK SINUS SYNDROME)
Sinus node dysfunction is a syndrome manifested by a variety of cardiac arrhythmias, including sinus bradycardia, sinoatrial block, and paroxysmal supraventricular tachycardia, which frequently alternates with periods of bradycardia or even asystole (the so-called bradycardia-tachycardia syndrome). Symptoms can result from the bradycardia, the paroxysmal supraventricular tachycardia, or both. It is important to correlate symptoms with one of the above-mentioned arrhythmias by means of ECG, ambulatory ECG monitoring (Holter), or an event recorder. At times, correlation can be difficult because of the intermittent nature of the arrhythmia. Electrophysiologic studies can be used to diagnose sinus node dysfunction, but their utility is limited by low sensitivity and specificity.
Trained athletes often manifest physiologic sinus bradycardia with resting heart rates as low as 40 to 50 beats per minute. During sleep, these athletes may have a heart rate as low as 30 beats per minute with associated pauses or type I second-degree AV block resulting in asystole as long as 2.8 seconds.16,17 These findings are caused by increased vagal tone and usually do not indicate the need for pacing. In the general population, monitoring during sleep may reveal sinus pauses of variable duration. The clinical significance of such pauses is uncertain. In patients with sleep apnea, treatment of the apnea frequently will reduce the occurrence of pauses. In turn, atrial overdrive pacing (pacing at a rate higher than the native sinus rate) may reduce episodes of obstructive sleep apnea in these patients.18 Another form of sinus node dysfunction is “chronotropic incompetence,” defined as an inadequate sinus rate response to stress or exercise. Clinical studies have shown that rate-responsive pacing clinically benefits patients by restoring physiologic heart rate during exercise.19
Although sinus node dysfunction is the most common reason for pacemaker implantation,19 permanent pacing in these patients may not improve survival, despite symptom reduction and improvement in quality of life.20,21 Atrial-based pacing may be preferable in patients with sinus node dysfunction,22 although conflicting data have been reported in various studies. A recent randomized trial23 reported reduced risk of atrial fibrillation recurrence with dual-chamber pacing compared with ventricular-based pacing in patients with sinus node dysfunction. The main pacing recommendations for this syndrome are listed in Table 4.1
PREVENTION AND TERMINATION OF TACHYARRHYTHMIAS
Permanent pacing may prevent or terminate certain ventricular and supraventricular arrhythmias in selected patients,24,25 although it usually is not considered as first-line therapy. Re-entrant rhythms (atrial flutter, reentrant paroxysmal supraventricular tachycardia, and ventricular tachycardia) may be terminated by specific pacing patterns available in special antitachyarrhythmia devices. These devices may detect the tachycardia and automatically initiate a pacing sequence to terminate it or may require that the patient initiate activation by application of an external magnet.
Atrial synchronous pacing may prevent recurrences of re-entrant paroxysmal supraventricular tachycardia,26 but catheter ablation is used more commonly and is more effective. In patients with sinus node dysfunction and paroxysmal atrial fibrillation, one randomized study23 demonstrated a 21 percent lower risk of atrial fibrillation recurrence with atrial-based pacing. Permanent pacing has been shown to prevent arrhythmias in other situations. In some patients with the long QT syndrome, ventricular tachycardia may be prevented by continuous pacing.27 A combination of beta blockade and pacing can shorten the QT interval and prevent sudden cardiac death28 in patients with the long QT syndrome. However, the insertion of an implantable defibrillator in combination with overdrive pacing should be considered in high-risk patients. The main recommendations for the prevention and termination of tachyarrhythmias by pacing are listed in Tables 5 and 6.1
SYNCOPE AND NEURALLY MEDIATED SYNDROMES
Syncope is commonly encountered in clinical practice, particularly in older patients. It usually results from a sudden, transient reduction of cerebral blood flow. Older patients are particularly prone to syncope because of age-related physiologic changes that reduce their ability to adapt to sudden blood pressure drops. Syncope may be caused by a variety of cardiac and noncardiac conditions, and the precise cause often is difficult to identify. In patients with heart disease, an arrhythmic cause should be excluded.
Neurocardiogenic syncope and neurally mediated syndromes account for 10 to 40 percent of syncopal episodes. Vasovagal syncope is one of the most common types of neurocardiogenic syncopal syndromes. Classically, it is preceded by a prodrome of nausea and diaphoresis and is associated with severe bradycardia. Episodes usually are triggered by pain, anxiety, or stress. A family history of syncope may be present.
Pacing therapy for patients with refractory neurocardiogenic syndromes associated with severe bradycardia or asystole is controversial. Approximately 25 percent of patients have a predominant vasodepressor response (loss of vascular tone and hypotension without significant bradycardia).29 These patients would not benefit from pacing. Another large subset have a mixed vasodepressor and cardioinhibitory response (i.e., sinus bradycardia, prolongation of the PR interval, or advanced AV block). Although some studies have shown significant reduction of syncopal episodes with pacing therapy,30 others have reported that pacing did not prevent syncope any better than drug therapy.31 In a recent randomized trial of symptomatic patients with bradycardia, permanent pacing decreased the one-year recurrence rate of syncope to 19 percent in patients treated with a pacemaker, compared with 60 percent in control patients.32 Head-up tilt-table testing may be diagnostic in this condition and help the physician select patients who may benefit by pacing therapy. Asymptomatic spontaneous or provoked prolonged sinus pauses in this group of patients have an excellent prognosis even without pacing therapy.33
An uncommon cause of syncope is the hypersensitive carotid sinus syndrome, which is defined as syncope or presyncope resulting from an extreme response to carotid sinus stimulation. As in vasovagal syncope, there usually are vasodepressor and cardioinhibitory components of this reflex. Therefore, before pacing therapy is recommended for carotid sinus hypersensitivity, the relative contribution of the two reflex components must be determined. The basic criterion for a diagnosis of hypersensitive carotid sinus syndrome is asystole in excess of 3 seconds caused by sinus arrest or advanced AV block as a result of carotid sinus stimulation.31 Pauses up to 3 seconds during carotid sinus stimulation are considered to be physiologic. Recent studies have reported that many older patients who sustain unexplained falls may have a hypersensitive carotid sinus,34 and that pacing therapy can reduce subsequent falls significantly.35 The main recommendations for pacing therapy in these syndromes are listed in Table 7.1
HEART FAILURE AND DILATED CARDIOMYOPATHY
Thirty to 50 percent of patients with advanced congestive heart failure have major intraventricular conduction defects.36 These defects and especially complete left bundle branch block have been shown to be independent predictors of mortality. Delayed activation of the left ventricle as a result of left bundle branch block has been shown to produce significant dyssynchrony in left ventricular contraction and relaxation. The dyssynchrony can be minimized by biventricular pacing. This pacing technique has been shown in several prospective randomized trials to reduce heart failure symptoms, and improve hemodynamics and left ventricular indices of systolic function.37–39 However, there is no convincing evidence that biventricular pacing improves survival. Many patients with severe left ventricular dysfunction who meet criteria for biventricular pacing currently are receiving devices that combine pacemaker and cardioverter-defibrillator modalities. It is fair to say that the indications for biventricular pacing are still evolving. Current pacing recommendations for dilated cardiomyopathy are listed in Table 8.1
Pacemaker Selection and Patient Follow-Up
Pacemakers that maintain synchrony between atria and ventricles are particularly advantageous in older adults because the hemodynamics of aging include a reduction of ventricular compliance and increased dependence of ventricular filling on atrial contraction. Similarly, rate-responsive pacemakers are preferred in patients with sinus node dysfunction and chronotropic incompetence. Although single-lead atrial-based pacing has the advantage of simplicity and lower cost, concerns about subsequent development of AV block limit its application. A detailed description of the types of pacemakers available and indications for implantation of a particular device are beyond the scope of this review.
Careful follow-up and monitoring of implanted device function are essential elements of care after a patient has received a pacemaker. The frequency and method of such follow-up depend on many factors, including the age of the patient, number and severity of comorbid conditions being treated, and age of the device. Telephone and in-person clinic follow-up can be equally effective when performed appropriately.