Controversy surrounds the management options for localized prostate cancer—conservative management, prostatectomy, and radiation. Choosing among these options is difficult because of long-term side effects that include sexual, urinary, and bowel dysfunction. Some recent studies suggest that patients who have chosen treatment (i.e., radical prostatectomy or radiation) have longer disease-free survival compared with patients who have chosen conservative management (i.e., watchful waiting). However, several biases may artificially enhance the perceived value of treatment and make the interpretation of studies on treatment outcomes difficult. Sources of bias include lead time, length time, and patient selection. Because of the uncertain efficacy of management options and the risk of long-term treatment complications, family physicians need to engage their patients in the decision-making process.
Although prostate cancer is a commonly diagnosed malignancy, its management remains controversial. The majority of patients with prostate cancer are older than 65 years (median age of diagnosis is 71 years for white American men and 69 years for black American men).1 Approximately 220,000 American men were diagnosed in 2003, but because of the long natural history of prostate cancer, there were only 28,900 deaths in that year.2 Conservative management, or watchful waiting, has been suggested as an alternative to more aggressive therapies such as radical prostatectomy or radiation because many patients with prostate cancer will die from other causes (most commonly heart disease). Conservative management also may be favorable for older men with a life expectancy of less than 10 years because they are unlikely to benefit from, and perhaps less able to tolerate, aggressive interventions.3
Strength of Recommendations
| Key clinical recommendation | Label | References | Comments |
|---|---|---|---|
| Radical prostatectomy should be considered for patients with moderately and poorly differentiated localized prostate cancer. | B | 4,5,17 | Better disease-free survival rates and fewer deaths from prostate cancer after radical prostatectomy compared with conservative management. Probable small difference in overall survival. |
| External radiation therapy and radical prostatectomy are equally recommended. | B | 6,7,21 | Treatments appear to be similar with respect to disease-free survival rates. Radiation therapy probably has a lower risk of erectile dysfunction compared with radical prostatectomy but carries a higher risk of chronic bowel symptoms. |
| Conservative management should be considered for patients with well-differentiated, localized prostate cancer. | B | 12,13 | No difference in overall survival in patients with well-differentiated cancer (i.e., Gleason score of 2 to 4) compared with patients without cancer. Shorter life expectancies and higher risk of death in patients with poorly differentiated cancer. Intermediate outcomes in patients with moderately differentiated cancer (i.e., Gleason score of 5 to 6). |
| Brachytherapy and external radiation therapy are equally recommended. | B | 22,23,35 | Treatments appear to be similar with respect to disease-free survival rates. Risk of erectile dysfunction similar to conformal radiation therapy; risk of irritative urinary symptoms following brachytherapy may be higher. Chronic bowel symptoms appear to be rare. |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, opinion, or case series. See page 1865 for more information.
Recent studies4–7 have suggested that patients with clinically localized prostate cancer have a longer disease-free survival following radical prostatectomy or radiation. Direct comparisons of treatment options with conservative management generally have favored treatment, partly because of study design issues often found in observational prostate cancer studies: lead-time, length-time, and selection bias. With these biases in mind, this analysis discusses outcomes and long-term side effects associated with the primary management options for clinically localized prostate cancer, including external radiation and interstitial seed radiation (or brachytherapy).
Sources of Bias
LEAD-TIME BIAS
Screening generally detects early disease in asymptomatic patients. Survival length typically is calculated from the date that disease is diagnosed until death. Therefore, the interval between cancer detection and death is longer in screened patients than in unscreened patients (Figure 1). Prostate-specific antigen (PSA) screening has shifted the diagnosis of prostate cancer toward clinically localized (i.e., T1 and T2) disease.5–8 The lead time resulting from PSA screening has been estimated to be between three and five years.9 Increased survival rates after treatment in the post-PSA era, when compared with the pre-PSA era, may be the result of earlier diagnosis and not necessarily early detection and treatment. A large randomized trial comparing treatment with conservative management for patients diagnosed by PSA screening has not been completed.10
Figure 1. Lead-Time Bias
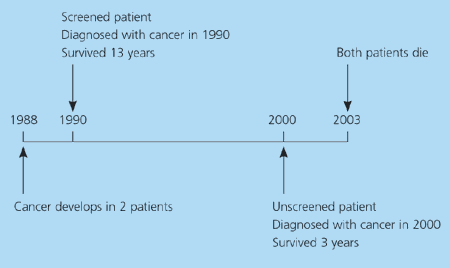
Two men develop prostate cancer in 1988 and die in 2003. One patient was not screened; he developed symptoms and was diagnosed with prostate cancer in 2000 and he died three years later. A second patient was screened and diagnosed with early prostate cancer in 1990; after diagnosis, he lived 13 additional years. The screened patient has a longer interval between the diagnosis of prostate cancer and death, a longer disease-free survival. This may be the result of early diagnosis and not necessarily the result of screening and early treatment.
LENGTH-TIME BIAS
Diseases with long preclinical phases are more likely to be detected by screening (Figure 2).11 For example, screening may not be able to detect rapidly progressing disease because the window of opportunity to detect asymptomatic disease is small. Because of differences in tumor differentiation (Gleason score), prostate cancer tumors progress at different rates (Table 1).12 A greater proportion of cancers diagnosed as a result of PSA screening are moderately differentiated (i.e., Gleason score of 5 to 6) than are poorly differentiated.5–8 It is not clear whether all patients with Gleason 5 to 6 cancer benefit from aggressive management.13 Therefore, better disease-free survival rates after treatment in the post-PSA era may be the result of treatment for less aggressive tumors (length-time bias) and not necessarily because of early detection and treatment.
Figure 2. Length-Time Bias

Differences in tumor differentiation may result in length-time bias. In this example, the broken line represents the rapid growth of a poorly differentiated tumor. These tumors are less likely to be diagnosed by screening and (because of the rapid growth) are more likely to be diagnosed as a result of clinical symptoms. Slow-growing tumors(solid line) are more likely to be diagnosed by screening. Differences in survival that are observed in screened and unscreened populations may be caused in part by different cancer types (and not by early screening and treatment).
SELECTION BIAS
Observational studies may be biased because of patient selection. Outcomes in patients from tertiary care institutions may not be directly comparable with those in patients from community settings. For example, urinary complication rates following surgery have been shown to be lower in high-volume hospitals.14 Surgical studies tend to select only patients who have had a radical prostatectomy; patients with advanced disease generally are excluded because surgery often is stopped upon the discovery of extensive disease.15 More importantly, surgically managed patients will have pathologic evaluation of resected tissue and pelvic lymph nodes. Only 40 to 60 percent of patients with clinically localized disease (T1 and T2) remain classified with localized disease because of capsular (T3) or nodal (T4) involvement.4–5 Thus, patients with localized cancer based on surgical staging (after radical prostatectomy) generally will have better outcomes compared with patients with clinically localized cancer (during conservative management or after radiation) because patients with advanced disease have been excluded from the surgical cohort.
Methods
A structured review was used to evaluate treatment outcomes based on literature published in the past 15 years. Search terms included “prostate cancer and prostatectomy,” “radiation,” or “watchful waiting,” and “prostate cancer and quality of life” or “treatment complications.” Titles and abstracts were scanned to select papers that considered treatment outcomes or treatment complications. Papers considering hormonal, combination, or salvage therapies were not included.
Management Options
CONSERVATIVE MANAGEMENT
Conservative management, with or without androgen ablation reserved for symptomatic disease, is one option for prostate cancer patients.3 One study,16 based on the Connecticut Tumor Registry, documented outcomes for 451 patients (between 65 and 75 years of age) who were diagnosed with clinically localized prostate cancer from 1971 to 1976 and managed conservatively. Compared with an age-matched cohort of men without prostate cancer, patients with well-differentiated prostate cancer (i.e., Gleason score of 2 to 4) did not have an increased risk of death; patients with Gleason scores of 8 to 10, however, had about a two- and threefold increase in the risk of death, respectively. A more recent analysis12 of 767 patients from the same registry has shown that patients with Gleason 2 to 4 and Gleason 5 to 6 cancers under conservative management had a relatively small risk of dying from prostate cancer; patients with Gleason 7 to 10 cancers under conservative management, however, were more likely to die from prostate cancer than other causes (Table 1).12
PROSTATECTOMY
Surgical removal of the prostate is an option for patients with clinically localized cancer. A large randomized trial in the post-PSA era comparing radical prostatectomy with conservative management is under way, but will not be completed for several years.10 A recently published Scandinavian trial17 randomized 695 patients with clinically localized prostate cancer (median age, 65 ± 5 years) to radical prostatectomy or conservative management (median follow-up, 6.2 years). The majority of patients had Gleason 5 to 6 cancer. More than one third of these men were diagnosed with prostate cancer because of symptoms; only about 5 percent were diagnosed by PSA screening. At eight years, about 14 percent of patients whose disease was conservatively managed (95 percent confidence interval [CI], 7 to 15 percent) and 7 percent of prostatectomy patients (95 percent CI, 3 to 11 percent) developed metastases. The relative (radical prostatectomy compared with conservative management) hazard rate of death from prostate cancer (adjusted for age, Gleason score, and cancer stage) was 0.45 (95 percent CI, 0.25 to 0.84).17 However, differences in overall survival between the two treatment groups were not statistically significant. It is important to note that the study may not have been adequately powered to detect differences in overall survival. Furthermore, it remains to be determined whether an overall survival difference after radical prostatectomy may occur with a longer follow-up period.
Several case series have suggested that patients with early prostate cancer have a good disease-free survival rate following radical prostatectomy. The Mayo Clinic followed 2,518 patients with prostate cancer (mean age, 63 years) who underwent radical prostatectomy between 1990 and 1993 (mean follow-up, 5.6 years). The majority of patients had clinically localized Gleason 5 to 6 cancer. Less than 20 percent of these men were diagnosed as a result of PSA screening. After radical prostatectomy, 73 to 83 percent of patients diagnosed with clinically localized (T1 and T2) cancer were alive and free of disease at five years. For comparison, 63 percent of patients with locally advanced T3 cancer were alive and free of disease at five years.4
Another large series5 followed 2,091 patients (average age, 58.1 ± 6.6 years) with clinically localized prostate cancer who had a radical prostatectomy between 1982 and 1999. The majority of patients had Gleason 5 to 6 cancer, and 40 percent of these patients were diagnosed as a result of PSA screening. Biochemical failure (i.e., a PSA level higher than 0.2 ng per mL) was one indicator of recurrence. The five-, 10-, and 15-year probabilities of biochemical recurrence-free survival were 84, 72, and 63 percent, respectively. The corresponding metastases-free survival probabilities were 96, 89, and 81 percent.5 Another related study18 found that the median time to biochemical failure was about eight years, and the median time to metastases after biochemical failure was about five years.
Observations from these two studies suggest that patients have good disease-free survival rates following radical prostatectomy for clinically localized disease. However, to draw inferences about the efficacy of surgery, these results would have to be compared with older studies that followed men during conservative management in the pre-PSA era. This comparison may be biased in favor of surgery because some of the patients in the more recent surgical studies were diagnosed by PSA screening; thus, more recent studies have a greater proportion of patients with localized disease (a downward stage migration or lead-time bias). It also is not clear whether the type of cancer detected early or by screening is the same as cancer detected based on clinical symptoms (length-time bias). Finally, these surgical studies have included only patients who have had a radical prostatectomy and are from tertiary care institutions, and may not be applicable to patients in the community setting (selection bias). Therefore, management outcomes in the post-PSA era based on large community databases (i.e., Surveillance, Epidemiology, and End Results), may be more applicable to the average patient.8 However, these databases are just beginning to mature because of the long natural history of prostate cancer and the time required for updating these databases.19
RADIATION TREATMENT
Another treatment for patients with localized prostate cancer is external or interstitial radiation. Conformal external beam radiation generally has replaced conventional external radiation because higher doses of radiation can be directed to the prostate with less radiation to the surrounding tissues.20 Interstitial radiation or brachytherapy involves the placement of radioactive pellets (i.e., iodine-125 or palladium-103) into the prostate gland.
No large trials comparing radiation with other forms of treatment in the post-PSA era have been conducted. Some outcomes that are not specific to prostate cancer, such as overall survival, may be worse after radiation because patients who choose radiation tend to be older and have more comorbid illnesses than patients who choose radical prostatectomy (selection bias). To compare treatment outcomes, it is preferable to compare studies with similar clinical stages and grade distributions to minimize lead and length-time biases. A recent retrospective cohort study21 found similar biochemical failure (a rising PSA after treatment) rates after radical prostatectomy and higher-dose radiation (≥ 72 Gy). Differences between radiation and radical prostatectomy were attributable to pretreatment PSA and T-stage (lead time) and biopsy Gleason score (length time).21 Several case series also have shown that disease-free survival following radiation is comparable with radical prostatectomy.6–7 The efficacy of brachytherapy is comparable with external radiation.22–23
Long-Term Side Effects
Radical prostatectomy has the highest risk of immediate serious complications, including a low risk of death.24 Although all of these management options have long-term side effects, patients are more likely to experience symptoms as a result of cancer progression (i.e., urinary obstruction and bone pain) during conservative management.25 Because approximately twice as many patients develop metastatic disease under conservative management, these patients are probably more likely to require hormone ablation therapy, which causes side effects such as fatigue and hot flashes.17–26
Erectile dysfunction and urinary side effects are the most common complications following radical prostatectomy and radiation (Table 2).20,27–47 The risk of developing these symptoms after treatment increases with patient age and comorbid illnesses.48–49 Lower complication rates occur in hospitals that perform a large number of prostatectomies. Fewer men will have postsurgical erectile dysfunction after unilateral or bilateral nervesparing surgery.42–50 Urinary side effects may be lower with sparing of the bladder neck51 and muscles of the distal urethral sphincter.49 Estimates for the risks of post-treatment complications also depend on how side effects are described in a study and tend to be higher if patients, instead of physicians, report the symptoms.42–43
TABLE 2 Side Effects of Prostate Cancer Treatment
| Side effect | Estimated risk after radical prostatectomy (%) | Estimated risk after external radiation therapy (%) | Estimated risk after brachytherapy (%) |
|---|---|---|---|
| Bowel dysfunction | 2 to 17 | 0 to 30 | 1 to 10 |
| Erectile dysfunction | 50 to 90 | 30 to 85 | ∼ 20 at 2 to 3 years, |
| ∼ 50 at 5 to 6 years | |||
| Urinary dysfunction | 15 to 60 | 2 to 30 | 12 to 30 |
In general, more than 50 percent of patients are expected to have erectile dysfunction as a result of radical prostatectomy.27,28,37,38 Generally, the risk is less following conformal radiation and brachytherapy.35 The risk of frank urinary incontinence is low, but milder urinary symptoms (i.e., urine leakage) are common after radical prostatectomy and external radiation.27,32,49 More patients will have irritative urinary symptoms after brachytherapy than after external radiation.35 The risk of long-term bowel symptoms (i.e., tenesmus and stool leakage) is highest following external radiation.27–29 The risk of long-term bowel symptoms following brachytherapy is low.36–52
Patient Recommendations
Until the completion of randomized trials in the post-PSA era, it is difficult to recommend a best management option for localized prostate cancer with certainty.10 Because of long-term side effects, the integration of survival outcomes with quality of life is important. One recent study13 used decision modeling to estimate quality-adjusted life years (QALYs) for patients with clinically localized prostate cancer. Patients with Gleason 2 to 4 cancer had a decreased number of QALYs following treatment; conversely, patients with Gleason 7 to 10 cancer had an increased number of QALYs following treatment. Outcomes for patients with Gleason 5 to 6 cancer were less clear; treatment was marginally beneficial depending on the underlying assumptions used in the model.13
This review focuses on management options for localized prostate cancer; patients with advanced disease (i.e., extracapsular or nodal involvement) generally are not candidates for curative management. Based on the recent literature, the following suggestions can be made for patients with clinically localized prostate cancer. Conservative management is reasonable for patients with Gleason 2 to 4 cancer because these patients do not have a shortened life expectancy, and treatment is associated with long-term side effects. In contrast, patients with Gleason 7 to 10 cancer should consider treatment (i.e., radical prostatectomy or radiation). These patients have a high risk of dying from prostate cancer, and disease-free survival appears to be better after treatment. The majority of patients have moderately differentiated (Gleason score of 5 to 6) cancer. Because there is no convincing evidence for or against treatment, younger patients who are tolerant of long-term treatment side effects should consider radical prostatectomy or radiation therapy.13 However, older patients and patients with a life expectancy of less than 10 years with moderately differentiated cancer probably will not benefit from more aggressive management of their disease.

