Group A beta-hemolytic streptococcal pharyngitis, scarlet fever, and rarely asymptomatic carrier states are associated with a number of poststreptococcal suppurative and nonsuppurative complications. As in streptococcal pharyngitis, acute rheumatic fever, pediatric autoimmune neuropsychiatric disorders associated with streptococcal infection, and poststreptococcal glomerulonephritis most often occur in children. The hallmarks of rheumatic fever include arthritis, carditis, cutaneous disease, chorea, and subsequent acquired valvular disease. Pediatric autoimmune neuropsychiatric disorders encompass a subgroup of illnesses involving the basal ganglia in children with obsessive-compulsive disorders, tic disorders, dystonia, chorea encephalitis, and dystonic choreoathetosis. Poststreptococcal glomerulonephritis is most frequently encountered in children between two and six years of age with a recent history of pharyngitis and a rash in the setting of poor personal hygiene during the winter months. The clinical examination of a patient with possible poststreptococcal complications should begin with an evaluation for signs of inflammation (i.e., complete blood count, erythrocyte sedimentation rate, C-reactive protein) and evidence of a preceding streptococcal infection. Antistreptolysin O titers should be obtained to confirm a recent invasive streptococcal infection. Other important antibody markers include antihyaluronidase, antideoxyribonuclease B, and antistreptokinase antibodies.
Lancefield group A streptococci consist of a single species, Streptococcus pyogenes, that is associated with a variety of suppurative infections. Group A streptococci also have the capacity to trigger postinfectious syndromes of acute rheumatic fever and poststreptococ cal glomerulonephritis. Both entities are uniquely associated with these organisms. In addition, a variety of other nonsuppurative conditions are associated with infection with group A streptococci.1 Table 1 lists the suppurative and nonsuppurative complications of group A beta-hemolytic streptococcal illness.2–4 This article evaluates the most important complications.
Strength of Recommendations
| Key clinical recommendation | Label | References |
|---|---|---|
| To achieve clinical and bacteriologic cures, physicians should treat streptococcal pharyngitis with 10 days of oral penicillin, oral cephalosporins (other than cefaclor [Ceclor] or loracarbef [Lorabid]), or oral erythromycin in patients allergic to penicillin. | A | 1,9,15 |
| Initial laboratory testing for patients with suspected poststreptococcal disease should include a complete blood count, erythrocyte sedimentation rate or C-reactive protein, and an antistreptolysin O titer. | C | 11 |
| Antihyaluronidase, antideoxyribonuclease B, and antistreptokinase antibodies should be ordered if the diagnosis of a possible recent streptococcal infection remains in question. | C | 11,17 |
| Patients with suspected carditis should undergo echocardiography because electrocardiography can have false-negative results. | C | 11,20,32,33 |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, opinion, or case series. See page 1865 for more information.
TABLE 1 Complications of Group A Beta-Hemolytic Streptococcal Illness
| Nonsuppurative |
| Pediatric autoimmune neuropsychiatric disorders associated with streptococcal infection |
| Poststreptococcal autoimmune dystonia secondary to striatal necrosis |
| Poststreptococcal glomerulonephritis |
| Poststreptococcal reactive arthritis |
| Rheumatic fever |
| Sydenham’s chorea and other autoimmune movement disorders |
| Suppurative |
| Bacteremia |
| Cervical lymphadenitis |
| Endocarditis |
| Fasciitis/myositis syndrome |
| Mastoiditis |
| Meningitis |
| Otitis media |
| Perianal dermatitis in children |
| Peritonsillar or retropharyngeal abscess |
| Pneumonia |
| Sinusitis |
| Toxic shock |
Pathophysiology
Group A streptococci elaborate a number of cell surface components and extracellular products that are important in the pathogenesis of infection and in the immune response of the human host. Although the pathophysiology of poststreptococcal syndromes is not fully understood, antigenic mimicry may be the triggering factor leading to autoimmunity. In untreated persons with streptococcal pharyngitis, streptococcal breakdown products are thought to have molecular similarity to human tissues (e.g., heart valve tissue) and initiate the harmful immune response. The major surface protein of group A streptococci is M protein extending from the cell wall, which allows the bacteria to resist phagocytosis. The body is able to overcome this resistance with M protein–specific antibodies. This process also confers protection against subsequent infections with an organism of the same M type. However, the development of a vaccine that confers such immunity has been difficult because there are more than 80 antigenically distinct types of M protein.
In addition, group A streptococci produce extracellular products that may be important in local and systemic toxicity and in the spread of infection through tissues. These include streptolysins and toxins that damage cell membranes and account for the beta hemolysis produced by these organisms. The pyrogenic exotoxins, also known as erythrogenic toxins, are responsible for the rash of scarlet fever. Pyrogenic exotoxins also have been linked to unusually severe invasive infections including necrotizing fasciitis and streptococcal toxic shock syndrome.
Epidemiology
As in streptococcal pharyngitis, acute rheumatic fever and poststreptococcal glomerulonephritis most often occur in children. The peak incidence is between five and 15 years of age. Most initial attacks in adults take place in the late teens or early 20s. Rarely, initial attacks occur as late as the sixth decade. A recent epidemiologic survey5 of the Italian registry for renal biopsy, however, has suggested that poststreptococcal glomerulonephritis may be significantly underreported in patients 70 years and older.
Prevention and Recurrence
The complications of streptococcal infection arise predominantly from pharyngitis and scarlet fever, and rarely from asymptomatic carriers. Although antibiotics have been shown to reduce the severity of acute symptoms and shorten the duration of the illness by about one day, more than 90 percent of treated and untreated patients with acute pharyngitis are symptom-free by day 7.6 Therefore, the primary reason for treating uncomplicated streptococcal pharyngitis is to markedly reduce the incidence of subsequent rheumatic fever or its recurrence.7 It is unclear whether appropriate antibiotic treatment of group A streptococci infection reduces the risk of post-streptococcal glomerulonephritis, but one recent review8 did note a trend showing some protection.
Although oral or intramuscular penicillin and oral erythromycin are recommended for initial treatment of group A streptococcal pharyngitis, a recent meta-analysis9 found improved microbiologic and clinical outcomes among patients treated with most cephalosporins. The review identified 35 clinical trials comparing a cephalosporin with penicillin, and found significantly improved rates of clinical cure with cephalosporins (93.6 versus 85.8 percent). This was true even when the analysis was limited to the highest quality studies, although it did not apply to cefaclor (Ceclor) and loracarbef (Lorabid).
Rheumatic Fever
Although there has been a dramatic decline in severity and mortality of acute rheumatic fever since the 1940s, outbreaks still occur in the United States,10,11 closely following epidemics of streptococcal pharyngitis or scarlet fever. The disease continues to be an illness of the poor and underprivileged. Developing countries have an estimated 10 to 20 million new cases per year.12 Documentation of pharyngitis in older children and adults approaches 70 percent, whereas in younger children the rate is about 20 percent.13 Having hindsight suspicion of rheumatic fever in children presenting with signs of arthritis or carditis in the absence of a documented pharyngitis is important. Of note, outbreaks of impetigo never result in rheumatic fever.11,14,15
CLINICAL FEATURES
Rheumatic fever is a late inflammatory complication of acute group A streptococcal pharyngitis that manifests as an acute systemic febrile illness; it can include migratory arthritis involving the large joints, signs and symptoms of carditis and valvulitis, the erythema marginatum rash, subcutaneous nodules (Figure 1), and choreoathetotic movements of Sydenham’s chorea. These problems can appear in different degrees of severity and in various combinations. The clinical episodes may be time limited, but damage to the heart valves may be chronic and progressive, resulting in significant cardiac dysfunction. Although the Modified Jones Criteria provide a guide for clinical diagnosis,16 no specific symptoms, clinical signs, or laboratory tests are pathognomonic for rheumatic fever, nor do all patients with rheumatic fever fulfill the Modified Jones Criteria.11,16
Figure 1.
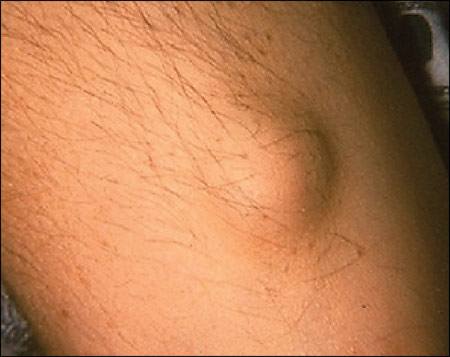
Subcutaneous nodule.
The criteria list major manifestations that include carditis, erythema marginatum, polyarthritis, subcutaneous nodules, and Sydenham’s chorea. Minor manifestations include clinical (e.g., arthralgia, fever) and laboratory (i.e., elevated C-reactive protein and erythrocyte sedimentation rate, prolonged PR interval on electrocardiograph) parameters. A diagnosis of rheumatic fever is supported by evidence of preceding group A streptococcal infection (i.e., positive throat culture or rapid streptoccocal antigen test, elevated or rising antistreptolysin titer), and the presence of two major manifestations or of one major and two minor manifestations.16
Arthritis
Arthritis is the most frequent and least specific manifestation of rheumatic fever. It usually affects the large joints and may be the first sign of illness. The lower extremities generally are affected first, followed by the upper extremities—hence the term migratory arthritis. Joint involvement occurs early in the illness and is more common and severe in adolescents and young adults. The polyarthritis may be painful, but it is transient; the inflammation lasts about two to three days in each joint and two to three weeks total. Radiographic examination can identify slight effusions but usually the results are normal. The arthritis is self-limited, resolves without sequelae, and responds well to salicylates and nonsteroidal anti-inflammatory drugs (NSAIDs).17
Carditis
Carditis associated with rheumatic fever manifests as pericarditis, myocarditis, and most commonly, endocarditis.11 Pericarditis can present with chest discomfort, pleuritic chest pain, pericardial friction rubs, and distant heart sounds. Myocarditis is rare in isolation and can present with signs and symptoms of heart failure. Endocarditis may be asymptomatic or present with a new heart murmur. Note that heart murmurs do not necessarily indicate valvular involvement, and they may be transient. If valvular disease occurs, it is most likely in the mitral, aortic, tricuspid, or pulmonary valve, in that order.18 Electrocardiograph and radiograph abnormalities may be present in as few as 30 percent of patients with carditis.19 In symptomatic and asymptomatic patients with a high clinical suspicion of rheumatic fever, echocardiography is recommended.
Rheumatic heart disease is an important long-term consequence of rheumatic fever and is the major cause of acquired valvular disease in the world.20 It usually occurs 10 to 20 years after the original rheumatic fever attack and is likely to occur in almost one half of patients with evidence of carditis at the initial diagnosis. Mitral stenosis caused by calcification is a classic finding and frequently needs to be treated surgically.
Sydenham’s Chorea
Sydenham’s chorea is characterized by abrupt, purposeless, nonrhythmic, involuntary movements; muscular weakness; and emotional disturbances. It usually is more marked on one side of the body than the other and may be completely unilateral. Outbursts of atypical behavior such as crying and restlessness are observed and, in rare cases, manifestations of a transient psychosis may be noted. There is no sensory loss or involvement of the pyramidal tracts. Sydenham’s chorea usually has a longer latency period and is a late isolated manifestation of the disease, thus acute-phase reactants and laboratory identification of streptococcal infection are absent. Sydenham’s chorea is self-limited, occurs in no more than 5 percent of patients, and typically lasts two to three months.17
Other Findings
Erythema marginatum is a rash consisting of erythematous rings found largely on the trunk that may appear, disappear, and reappear over several weeks or months. They typically are not pruritic and spare the face. Subcutaneous nodules are small, painless nodules on the extensor surfaces. Although subcutaneous nodules were thought to indicate carditis, this has been challenged, and on careful examination nodules are found in more than 10 percent of patients.21
Poststreptococcal Reactive Arthritis
Poststreptococcal reactive arthritis is a distinct clinical entity and is different from the typical arthritis associated with rheumatic fever. There is at least one to two weeks between the acute streptococcal infection and the onset of poststreptococcal reactive arthritis. In addition, the response to aspirin and other NSAIDs is poor. The modified Jones criteria usually are not met, and there appears to be no evidence of carditis. The duration is prolonged or recurrent and of high severity, and can be accompanied by tenosynovitis and renal abnormalities.22–24 There are no clear definitions or treatment guidelines to increase consistency in treatment.25,26
PANDAS
Pediatric autoimmune neuropsychiatric disorders associated with streptococcal infections (PANDAS) are indicative of a subgroup of children with obsessive-compulsive disorder or tic disorders occurring in association with streptococcal infections.27 Autoantibodies are proposed as the basis of this basal ganglia autoimmune illness, similar to the mechanism suggested for rheumatic fever and poststreptococcal glomerulonephritis.28 Included in this general category is Sydenham’s chorea, but it does not lead to carditis. There are five clinical criteria proposed for diagnosing PANDAS: the presence of obsessive-compulsive disorder or a tic disorder; prepubertal symptom onset; an episodic course characterized by acute, severe onset and dramatic symptom exacerbation; the presence of neurologic abnormalities such as choreiform movements during symptom exacerbations; and a temporal relationship between group A beta-hemolytic streptococcal infections and symptom exacerbation.29
Poststreptococcal Glomerulonephritis
Poststreptococcal glomerulonephritis is the leading cause of acute nephritic syndrome. Most cases are sporadic, although the disease has been known to occur in epidemic form. The illness is most frequently encountered in children between two and six years of age with a recent history of pharyngitis in a winter setting or a rash in a warmer climate. The incidence of poststreptococcal glomerulonephritis appears to be decreasing.30 It develops about 10 days after pharyngitis or two weeks after a skin infection with a nephritogenic strain of group A hemolytic streptococcus. Immunity is type-specific and long lasting; repeat infections are rare.30 It is unclear whether antibiotic treatment of the primary skin infection affords protection from the development of poststreptococcal glomerulonephritis. Studies31 addressing this question have been too small to answer this question definitely.
CLINICAL FEATURES
The classic presentation of poststreptococcal glomerulonephritis is a full-blown nephritic syndrome with oliguric acute renal failure. Most patients have milder disease, and subclinical cases are common. Patients with overt disease demonstrate gross hematuria characterized by red or smoky urine, headache, and generalized symptoms such as anorexia, nausea, vomiting, and malaise.32 Swelling of the renal capsule can cause flank or back pain. Physical examination may reveal hypervolemia, edema, or hypertension. Coexisting rheumatic fever is highly unsual.33 Acute poststreptococcal glomerulonephritis usually is diagnosed on clinical and serologic grounds without the need for biopsy, especially in children with a typical history.
Prognosis
The overall prognosis in classic poststreptococcal acute proliferative glomerulonephritis is good. More than 95 percent of patients recover spontaneously and return to baseline renal function within three to four weeks with no long-term sequelae.34 When there is delayed resolution, severe renal failure at onset, progressive renal failure, or systemic features such as skin rashes, joint pain, hepatosplenomegaly, and persistent fever, then biopsy is indicated to ensure that there is not a more serious cause for the illness.35 Isolated cases of hypertensive encephalopathy have been caused by asymptomatic poststreptococcal glomerulonephritis, attesting to the wide spectrum of poststreptococcal illnesses.36
Laboratory Evaluation
In patients with suspected poststreptococcal illness, laboratory evidence of inflammation and a preceding streptococcal infection should be documented. However, throat cultures often are negative by the time rheumatic fever, PANDAS, or poststreptococcal glomerulonephritis appears.37 Acute-phase reactants such as erythrocyte sedimentation rate and C-reactive protein levels are elevated early in the process and are effective in monitoring the disease. If either level returns to normal after discontinuation of antirheumatic therapy, the attack usually is over. A complete blood count should be ordered. Leukocytosis with neutrophilia and a mild normochromic normocytic anemia also may be present. Although synovial fluid analysis may reveal aseptic leukocytosis, it is not recommended routinely unless the patient has neurologic symptoms.
Several specific antibody responses are useful for the detection of preceding streptococcal infection because they reach a peak titer at about the time of onset of poststreptococcal disease and they indicate true infection rather than transient carriage. Initially, antistreptolysin O (ASO) titers should be obtained. If the test result is positive, it confirms a recent invasive streptococcal infection. The antibody response usually peaks at about four to five weeks and is first noted during the second or third week of an acute episode. However, ASO titers may be negative in up to 20 percent of patients who develop acute rheumatic fever.17
Elevated ASO titers may support but not definitely diagnose a poststreptococcal complication. The more specific and expensive antibody tests may be warranted, including antihyaluronidase, antideoxyribonuclease B, and antistreptokinase antibodies. Physicians should be aware that antibiotic therapy may prevent the development of an antibody response. If initial serologies are negative, but the clinical picture persists, it may be useful to repeat serial serum specimens two weeks later for comparison.17

