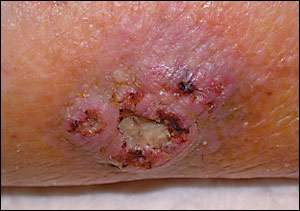
An 83-year-old woman presented with a non-healing ulcer on her left lower extremity. According to the woman’s caregiver, she had had the ulcer for more than 20 years. She reported no history of diabetes, coronary artery disease, or peripheral vascular disease, and she was taking no medications. On physical examination, there was a 3 × 3 cm ulcer, mildly tender to palpation, with a central necrotic area and irregular borders (see accompanying figure). The patient had no lower extremity edema and arterial pulses were palpable on both legs. A biopsy was performed.
Question
Based on the patient’s history and physical examination, which one of the following is the correct diagnosis?
A. Venous stasis ulcer.
B. Arterial ulcer.
C. Marjolin’s ulcer.
D. Pyoderma gangrenosum.
E. Diabetic ulcer.
Discussion
The answer is C: Marjolin’s ulcer. This entity was first described in 1828 by Marjolin, who diagnosed non-healing ulcers developing in burn scars. Later, Dupuytren found these ulcers to be malignant in nature.1 Today, the term Marjolin’s ulcer is used to describe a cancer arising from any site of chronic inflammation. Ninety percent of Marjolin’s ulcers develop from burn scars. Other sites include stasis ulcers, decubitus ulcers, granuloma inguinale, syphilitic lesions, and smallpox vaccination scars. They also may occur in sinus tracts from hidradenitis suppurativa, acne conglobata, or osteomyelitis.1
Marjolin’s ulcers occur most often on the extremities and in wounds that have been present for 30 years or more.2 Clinically, they exhibit exuberant granulation tissue that spills over their well-defined margins onto the surrounding tissue. Histologic examination is consistent with squamous cell carcinoma in 95 percent of cases, although basal cell carcinoma, melanoma, and sarcomas also may be present.3
Marjolin’s ulcers have a particularly aggressive course with higher rates of recurrence and overall metastasis compared with other forms of squamous cell carcinoma. The five-year survival rate is 30 percent.4 There is limited evidence on what percentage of treatment-resistant, non-healing ulcers reveal malignancy. Thus, experts recommend biopsy (i.e., wedge-shaped biopsy of the borders or multiple punch biopsies) of any non-healing ulcer resistant to standard intervention to rule out malignancy.5–7 Once diagnosed, wide excision followed by skin grafting is recommended. Although autologous skin grafting remains the gold standard, bioengineered skin substitutes such as cultured autologous and allogeneic keratinocyte grafts, composites, cellular and acellular matrices, and living skin substitutes also have been shown to stimulate healing by acting as an occlusive dressing while releasing tissue growth factors and cytokines.8 Elective lymph node dissection has been suggested because of the high rate of metastasis.9 Finally, amputation may be recommended for recurrent disease, or when a Marjolin’s ulcer is associated with underlying osteomyelitis.
The etiology for the malignant transformation is unclear. One suggestion is that previously traumatized skin may be more susceptible to mutation, and that fibrotic and avascular scar tissue may interfere with immune surveillance, resulting in the inability of circulating lymphocytes to reach and destroy neoplastic cells.1–9
The differential diagnosis is broad and may include any non-healing ulcer such as venous stasis ulcers, diabetic ulcers, arterial ulcers, or pyoderma gangrenosum.
Venous stasis ulcers account for 70 to 80 percent of all lower extremity ulcers. They usually are located in the gaiter area between the gastrocnemius muscle and a point 2.5 cm below the ankle. Venous stasis ulcers often are shallow with irregular borders and are surrounded by indurated, hyperpigmented skin. As the etiology suggests, venous stasis ulcers are found invariably on edematous legs, which may be leaking serous fluid.
Because diabetic ulcers usually are painless, they often go undetected. Usually developing on the feet, diabetic ulcers result from several factors associated with longstanding diabetes. First, peripheral neuropathy impairs the normal sensory protective mechanism of the feet, leaving them vulnerable to repeated trauma and subsequent ulcer formation. Secondly, these ulcers worsen and heal poorly as a result of microvascular and atherosclerotic derangements. Finally, diabetic ulcers have a high incidence of infection, which further slows the healing process. Arterial ulcers are the result of atherosclerosis and peripheral vascular disease. Narrowed vessels limit blood supply to the distal extremity, which results in ischemic necrosis. Arterial ulcers develop on the distal extremities and are sharply demarcated and painful with little granulation tissue. They may be accompanied by other signs of poor perfusion such as weak pulses and cool, hairless skin.
Pyoderma gangrenosum also may develop in surgical scars, fistulas, and ostomy sites. However, pyoderma gangrenosum is an immune-mediated, inflammatory condition commonly associated with inflammatory bowel disease and immunodeficient states. These ulcers are painful with well-demarcated, undermined edges. They have a purulent, dusky purple base, surrounding erythema, and satellite pustules.10
Selected Differential Diagnosis of Non-healing Ulcers
| Condition | Characteristics |
|---|---|
| Venous stasis leg ulcer | Located in the gaiter area; shallow with irregular borders; surrounded by indurated, hyperpigmented skin |
| Arterial leg ulcer | Develops on the distal extremities, sharply demarcated, painful with little granulation tissue |
| Marjolin’s ulcer | Cancer arising from any site of chronic inflammation, most often found in burn scars; squamous cell carcinoma most common |
| Pyoderma gangrenosum | Painful; well-demarcated, undermined edges and purulent, dusky purple base; surrounding erythema and satellite pustules |
| Diabetic leg ulcer | Often painless, may be deep, surrounded by thick callus, and may be malodorous because of secondary infection |

