
Am Fam Physician. 2005;72(5):845-848
Author disclosure: Nothing to disclose.
Mohs micrographic surgery is an approach to skin cancer removal that aims to achieve the highest possible rates of cure and to minimize the size of the wound and consequent distortions at critical sites such as the eyes, ears, nose, and lips. Mohs micrographic surgery is a two-step, same-day procedure performed with local anesthetic. It involves removing the tumor in stages by histologically confirming clear margins on frozen sections and by addressing the resultant defect. Options for healing include second intent, primary closure, local flaps, interpolation flaps, and grafts. Larger tumors may require referral for reconstructive surgery. Mohs micrographic surgery is the treatment of choice for skin tumors in critical sites, large or recurrent tumors, tumors in sites of radiation therapy, and tumors with aggressive histologic features.
Mohs micrographic surgery is an important advance in the treatment of selected skin cancers, in particular high-risk basal cell and squamous cell carcinomas, but many other types of cutaneous tumors as well. In 1941, Frederick Mohs described a surgical technique he had developed for the staged removal of skin cancer using in situ fixation of cutaneous tissue.1 After fixation, Mohs excised the cancer and cut horizontal sections from the undersurface of the tissue sample for microscopic viewing. The horizontal sectioning allowed for complete examination of the peripheral tumor margins, as opposed to the standard vertical sectioning, which did not produce continuity of a 360-degree circumferential margin.
Although many refinements have been made to Mohs’ original technique, the two main objectives are the same: to remove all of the tumor roots as accurately as possible by histologically confirming negative margins, and to create the smallest possible defect by sparing tissue uninvolved by the tumor. Mohs micrographic surgery minimizes tumor recurrence rates; reduces the size of surgical defects; allows for cosmesis; and preserves the function of the eyelids, the nares, the commissure of the mouth, and the ears.
Procedure
Most patients who are referred for Mohs micrographic surgery are treated on an out-patient basis with local anesthetic. Occasionally, oral sedation is added. The process begins with curettage of any residual tumor that may have persisted after the biopsy. Residual tumor generally is soft and evokes little resistance to the curette. Preoperative borders can be better predicted with this simple initial step.
The visible tumor is then excised, usually with 2-mm margins of normal skin. Any specimens are marked with colored dye to specify orientation. By convention, the 12 o’clock position usually is cephalad and the 6 o’clock position is caudad (Figure 1a). Hemostasis is obtained with electrocautery, and a temporary dressing is applied.
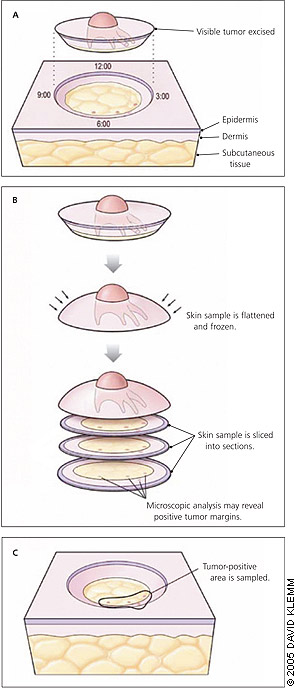
The tissue is made into frozen horizontal sections in an intraoffice laboratory. The Mohs surgeon examines the specimens microscopically (Figure 1b) and marks any positive tumor margins on a map drawn to approximate the scale and shape of the gross specimen. The histologic processing and microscopic analysis take about 45 minutes, after which more tissue in the tumor-positive areas is sampled, if needed (Figure 1c). The procedure is repeated until negative margins are confirmed. On average, two stages are required to completely remove the majority of tumors.
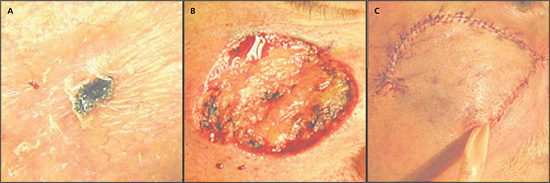
Once negative margins are verified, surgeons analyze the defect to select an optimal repair technique. Some lesions are small, shallow, or in convexities and heal by second intent, often with excellent results. Many defects are larger and deep enough that, without repair, functional impairment may result (e.g., retraction of an eyelid, elevation of a nasal ala, distortion of a commissure of the mouth). In these cases, surgeons have many reconstructive options from which to choose, including primary closure, local flaps (Figures 2a and 2b), or grafts. In some cases, the defects are severe enough that the patient must be referred for reconstructive surgery performed under general anesthesia.
Advantages
There are several advantages to taking a micrographic surgical approach to the treatment of skin cancer. First, Mohs micrographic surgery is the most effective method of eradicating the most common type of skin cancer, basal cell carcinoma,2 with a five-year cure rate of 99 percent.3 Having the same physician perform the dual function of surgeon and pathologist increases the accuracy of tumor localization and intraoperative interpretation of risk based on histologic subtypes encountered during surgery. Indications of more aggressive tumor behavior, such as perineural invasion, may lead the surgeon to take more generous margins at specific sites.
Another benefit is that Mohs micrographic surgery spares tissue. Because the majority of skin cancers occur on the head and neck, preserving uninvolved tissue is of paramount importance, especially around the eyes, nose, ears, and mouth.
Finally, compared with other surgical techniques involving postoperative repair, the cost of Mohs micrographic surgery is similar to that of simple excision in the office with permanent section postoperative margin control. Mohs micrographic surgery is less expensive than excisions with intraoperative margin control with frozen sections performed in a private office or in an outpatient surgical facility.4
Complications
Potential complications from Mohs micrographic surgery are similar to those expected in the outpatient surgical setting (i.e., scarring, postoperative pain, bleeding, hematoma, flap or graft necrosis, and wound infection). Scarring can be minimized by selecting a repair technique appropriate for the size of the wound and the anatomic site. Most pain after Mohs micrographic surgery is minimal and can be controlled with oral analgesics.
Bleeding and hematoma can occur, particularly with local flaps and grafts. Bleeding complications are minimized by assiduous intraoperative hemostasis and occasionally by the placement of a temporary drain under larger flaps (Figure 2c). For patients receiving anticoagulants, perioperative discontinuation of these medications is not indicated,5 but patients must be counseled to restrict physical activity during the first 48 hours after surgery to minimize the risk of hematoma.
Flap necrosis is rare but can occur with poor flap design, with excessive wound tension, or as a consequence of bleeding and hematoma. Flap necrosis also can occur in persons who smoke heavily. Skin grafts often are delayed for a few weeks in smokers to allow the formation of granulation tissue at the wound bed before repair. Wound infection develops in less than 3 percent of patients,6 generally occurs about 48 hours after surgery, and usually can be managed with oral antibiotics.
Types of Skin Cancers Treated with Mohs Micrographic Surgery
Mohs micrographic surgery is not commonly used to treat melanoma because melanocytic atypia is difficult to assess with frozen sections. Permanent paraffin-embedded sections yield superior histologic resolution for assessment of melanocytic atypia.
In 2001, nonmelanoma skin cancers accounted for an estimated 1 million cancers in the United States.7 Skin cancer accounts for more than 50 percent of all cancers in the United States.8 Patients with basal cell carcinomas receive the most referrals for Mohs micrographic surgery, followed by persons with squamous cell carcinomas. Mohs micrographic surgery is an excellent approach to the management of a variety of other less common skin tumors, including Merkel cell carcinoma and neoplasms arising from the sebaceous gland and hair follicle unit or from the sweat coils and ducts.
After a biopsy has been performed and the presence of a carcinoma has been confirmed, the physician needs to classify the tumor as low risk or high risk. Low-risk tumors usually can be treated with simple techniques such as electrodesiccation and curettage, cryosurgery, or fusiform excision. Features of high-risk basal cell and squamous cell carcinomas that qualify for Mohs micrographic surgery can be grouped into five broad categories: (1) tumors in critical sites, (2) tumors of large size, (3) tumors with aggressive histology, (4) tumors in the immunosuppressed patient, and (5) tumors with postoperative involved margins or clinically ambiguous borders (Table 19 ).10–15
| High-risk anatomic location (eyelids, nose, ears, lips, genitalia, fingers) |
| Large tumors (20 mm or more in diameter) on the torso and extremities |
| Recurrent tumors after previous excision or destruction |
| Tumors occurring in previous sites of radiation therapy |
| Tumors with aggressive histologic patterns (small-strand, infiltrative, or morphea-like growth in basal cell carcinomas; perineural invasion; or poorly differentiated histology or deep invasion in squamous cell carcinomas) |
| Tumors in immunosuppressed patients |
| Tumors with involved borders or vague clinical margins, or incompletely excised tumors (positive histologic margins after resection) |
In critical sites such as the eyelids, nose, ears, lips, fingers, and genitalia, where tissue sparing is of paramount importance, tumors generally are treated best with Mohs micrographic surgery. The risk of recurrence and perineural invasion increases with tumor size,16 and the more simple therapeutic approaches often are ineffective for larger tumors. The size at which a tumor is considered at high risk of recurrence is relative to its location: a tumor of 6 mm or more in diameter on the central face is considered high risk, as is a tumor measuring 20 mm or more on the back (Table 19 ).17 Tumors in some locations, such as the eyelids, nose, ears, or lips, are candidates for Mohs micrographic surgery regardless of size.
Tumors with aggressive histology include sclerosing patterned basal cell carcinomas, which are characterized by thin tumor strands disseminated throughout a fibrous stroma. A sclerosing pattern arises in basal cell carcinomas from three causes: spontaneous evolution, a previous surgical or destructive procedure, and radiation fibrosis. Other features of aggressive histology are perineural or perivascular invasion, areas of squamous differentiation within a basal cell carcinoma, and poor differentiation in a squamous cell carcinoma. Immunosuppressed patients tend to develop more aggressive tumors with higher recurrence rates, particularly squamous cell carcinomas.18 Mohs micrographic surgery is always a consideration in this population.
Inadequate margin control, another feature of high-risk tumors, implies that the physician is having trouble defining the tumor borders clinically or that the tumor has been excised and the margins are histologically positive.
Referrals
A referral to a surgeon who performs Mohs micrographic surgery should include a pathology report, precise information about the tumor location, and a brief, pertinent medical history. Sometimes the surgeon has trouble accurately locating the tumor site after significant wound healing has occurred following biopsy. Many patients are older adults who have scars from previous treatments and poor vision that makes it difficult for them to identify the original biopsy site. A photograph or drawing pinpointing the anatomic site can help avoid this problem and the resultant delays in treatment.
To find a local surgeon who can perform Mohs micrographic surgery, family physicians can consult the “Find a Surgeon” section on the Web site of the American College of Mohs Micrographic Surgery and Cutaneous Oncology athttp://www.acmmsco.org.