
Am Fam Physician. 2005;72(8):1527-1534
Patient information:See related handout on genital herpes, written by the author of this article.
Author disclosure: Nothing to disclose.
Genital herpes simplex virus infection is a recurrent, lifelong disease with no cure. The strongest predictor for infection is a person’s number of lifetime sex partners. The natural history includes first-episode mucocutaneous infection, establishment of latency in the dorsal root ganglion, and subsequent reactivation. Most infections are transmitted via asymptomatic viral shedding. Classic outbreaks consist of a skin prodrome and possible constitutional symptoms such as headache, fever, and inguinal lymphadenopathy. As the infection progresses, papules, vesicles on an erythematous base, and erosions appear over hours to days. These lesions usually crust, re-epithelialize, and heal without scarring. First-episode infections are more extensive: primary lesions last two to six weeks versus approximately one week for lesions in recurrent disease. Atypical manifestations are common. Infected persons experience a median of four recurrences per year after their first episode, but rates vary greatly. Genital herpes simplex virus type 2 recurs six times more frequently than type 1. Viral culture is preferred over polymerase chain reaction testing for diagnosis. Serologic testing can be useful in persons with a questionable history. Effective oral antiviral medications are available for initial, episodic, and suppressive therapy but are not a cure. There is some evidence that alternative therapies such as L-lysine, zinc, and some herbal preparations may offer some benefit. Counseling patients about the risk of transmission is crucial and helps prevent the spread of disease and neonatal complications.
| Clinical recommendation | Evidence rating | References |
|---|---|---|
| A test for isolation of HSV in cell culture should be obtained in patients who present with genital lesions. | C | 2 |
| Although type-specific assays can be used to confirm HSV infection, screening for HSV-1 or HSV-2 infection in the general population is not indicated. | C | 2,18 |
| Acyclovir (Zovirax), valacyclovir (Valtrex), or famciclovir (Famvir) should be offered to patients with genital herpes to reduce symptoms, but these agents are not a cure for HSV infection. | A | 2 |
| Episodic therapy for three days should be offered to patients who have mild and infrequent HSV outbreaks. | B | 2,23 |
| Because the frequency of outbreaks often decreases, interruption of suppressive therapy for HSV infection should be considered at yearly intervals. | C | 2 |
Genital herpes simplex virus (HSV) infection is a recurrent, lifelong disease with no cure. HSV is an enveloped, linear, double-stranded DNA virus whose only known hosts are humans.1 The two types of HSV (i.e., HSV-1 and HSV-2) are distinguished by antigenic differences in their envelope proteins. HSV-1 normally is associated with oral infections and HSV-2 with genital infections, but either type can infect a person anywhere on the skin. At least 50 million persons in the United States have genital HSV infection,2 and an estimated 500,000 to 700,000 cases of symptomatic first-episode genital HSV infections occur annually.3 There is a concerning relationship between human immunodeficiency virus (HIV) and genital HSV infection because the interaction of HSV-2 and HIV-1 may result in more efficient transmission of HIV-1 and an increased rate of HIV replication during HSV reactivation.4 The strongest predictor for genital HSV infection is a person’s number of lifetime sex partners (Table 1).5
| Advent of sexual activity at or before 17 years of age |
| History of sexually transmitted diseases |
| History of undiagnosed genital lesions or discharge |
| Human immunodeficiency virus infection |
| Multiple sex partners |
| Multiple lifetime sex partners |
| Partner diagnosed with genital HSV infection |
The natural history of HSV infection includes acute or subclinical first-episode mucocutaneous infection, establishment of viral latency, and subsequent reactivation. The herpes virus enters the body through the skin or mucous membranes by direct sexual contact with the secretions or mucosal surfaces of an infected person. The virus multiplies at the epithelial layer and then ascends along the sensory nerve roots to the dorsal root ganglion, where it becomes latent. With reactivation, the virus travels from the dorsal root ganglion back down the nerve root to create a mucocutaneous outbreak, or it may produce no detectable symptoms.1 Subclinical viral shedding has been documented in more than 80 percent of HSV-2–seropositive persons who report no lesions.6 Only 10 to 25 percent of persons who are HSV-2 seropositive report a history of genital herpes, which suggests that most infected persons have unrecognized symptomatic or completely asymptomatic infections.6 However, once patients are told of their positive antibody status, more than 50 percent identify clinically symptomatic recurrences that previously were ascribed to other conditions.6 It is thought that viral shedding in persons who are unaware that they are infected is responsible for at least 70 percent of HSV transmission.6
Clinical Manifestations
“Classic” outbreaks of primary genital HSV infection begin with a prodrome lasting two to 24 hours that is characterized by localized or regional pain, tingling, and burning. Patients also may have constitutional symptoms such as headache, fever, inguinal lymphadenopathy, anorexia, and malaise.7 As the disease progresses, papules, vesicles on an erythematous base, and erosions appear over hours to days. Patterns of HSV-1 and HSV-2 infection appear identical: vesicles usually are uniform in size, and the tense center umbilicates to form a depressed center. These lesions usually crust and then re-epithelialize and heal without scarring.8 In women, ulcers can occur on the introitus, urethral meatus, labia, and perineum. In men, ulcers often appear on the shaft or glans of the penis. In both men and women, lesions may appear on the perianal area, thighs, or buttocks7 (Figure 1).
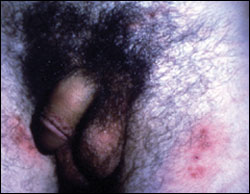
There are two types of first-episode, clinically apparent eruptions. The first is a nonprimary clinical eruption in a patient who has been infected previously with any type of HSV. The second type is a true primary infection, which is the first HSV infection in a seronegative patient. First-episode infections have more numerous and scattered vesicles and more systemic symptoms. Approximately 80 percent of primarily infected persons develop constitutional symptoms.7
Primary lesions last two to six weeks and can be extremely painful, containing large quantities of infectious HSV particles.8 Vesicles appear approximately six days after sexual contact.8 Viral shedding lasts longer in first-episode infections, usually 15 to 16 days, and new lesions will continue to form for about 10 days after the initial infection.9 For unknown reasons, women have more severe disease, constitutional symptoms, and complications than do men.8,10 This may be a result of the larger affected surface area in women and the ability of the virus to spread more easily over moist surfaces. Cervical ulcerative lesions are common and are almost always associated with first-episode disease. This type of lesion can present with intermittent bleeding and vaginal discharge. Dysuria and urinary retention syndromes may occur from urethral lesions and urine contact with vulvar lesions. Up to 25 percent of women may develop aseptic meningitis.7,10
Systemic complications are rare in men, although a similar urinary retention pattern may occur in men with severe disease, especially if they are uncircumcised.8 Crusts usually will not form on the moist surface under the foreskin. Perianal infection with associated proctitis is common in homosexual men.11
Recurrent HSV outbreaks usually are milder than the initial episode: there typically are fewer grouped lesions (Figures 2 and 3), and viral shedding occurs at a lower concentration and for a shorter duration (i.e., about three days). However, the prodromal symptoms are similar. Crusts are shed in approximately eight days to reveal a pink, re-epithelialized surface8 (Figure 4). Atypical manifestations are common: vesicles may not form, and HSV infection may be misdiagnosed as another disease.7 Review of a patient’s chart may be useful in identifying multiple, nonspecific complaints that serve as red flags. Patients who previously tested positive for HSV-1 and are newly infected with HSV-2 are less likely to present with classic signs and symptoms.8 Antibodies for one type of HSV seem to provide partial protection against infection with the other serotype: women infected with HSV-1 have a 5 to 20 percent lower annual rate of seroconversion to HSV-2 compared with uninfected women.12
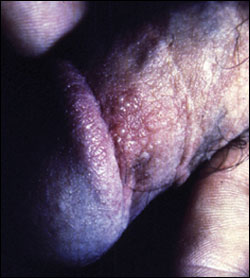

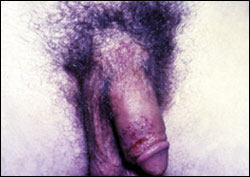
Recurrence rates for HSV-2 vary greatly, but the median is four recurrences per year, and the median time to first recurrence is 50 days.13 Approximately 90 percent of patients have at least one recurrence during the first year (Table 2).13 Patients who experience more severe primary infections (i.e., lasting 35 days or more) have recurrent episodes twice as often and with a shorter time to first recurrence.13 Men have more recurrences than women.13 Although HSV-1 can cause up to 33 percent of initial outbreaks, HSV-2 infections recur six times more frequently.8 Recurrent outbreaks occur in 50 percent of patients with HSV-1, and the median time to the first recurrence is one year following primary infection.8 Recurrences seem to continue at these rates for at least three years8 and may become less frequent over time. Recurrences are spontaneous, but various factors such as fever; nerve or tissue damage; physical or emotional stress; exposure to heat, cold, and ultraviolet light; immunosuppression; menses; concurrent infection; fatigue; and sexual intercourse have been associated with recurrences.7
| Recurrences* | Incidence (%) |
|---|---|
| Zero | 11 |
| 1 or more | 89 |
| 6 or more | 38 |
| More than 10 | Overall: 20 |
| Men: 26 | |
| Women: 14 |
Autoinoculation to different areas of the body can occur (e.g., by touching an open lesion on the mouth and scratching the genital area), but this process usually is prevented by circulating antibodies.14 Auto-inoculation is most likely to occur soon after primary infection, when antibody titers are still increasing. The use of condoms has been shown to reduce transmission rates of HSV-2 significantly in susceptible women but not in men. Using condoms during 25 percent or more instances of sexual intercourse was associated with reduced rates of HSV transmission, which suggests that even occasional condom use can protect women from acquisition of HSV-2.15 To be effective, the condom must completely cover lesions on an infected man.
Diagnosis
VIRAL CULTURE
Viral culture is an insensitive test for detection of HSV from genital ulcers but is highly sensitive for differentiating between HSV-1 and HSV-2.8 It is a widely available test and is the preferred method for identifying HSV infection.2 Cultures are much more productive during the vesicular or early ulcerative stages and generally are not productive more than five days after the patient becomes symptomatic. A prepackaged viral collection system—not standard cotton swabs—should be used. The vesicle, if present, should be punctured and the fluid absorbed onto the swab. The base of the lesion should be swabbed vigorously because the virus is cell-associated. The swab is then placed into the a viral system vial (one brand: Culturette) for transport. At the laboratory, specimens are inoculated into cell cultures and monitored microscopically for five to seven days to obtain maximum sensitivity.
POLYMERASE CHAIN REACTION TESTING
Polymerase chain reaction testing for HSV DNA has greater sensitivity than the traditional viral culture (sensitivity of more than 95 percent, compared with 75 percent for culture).16,17 Its role in the diagnosis of HSV infection has not yet been well defined, most likely because of the high cost. It is used for the diagnosis of HSV encephalitis because the results are more rapid than viral culture.2
SEROLOGIC TESTING
HSV antibodies form during the first several weeks after infection and remain indefinitely.2 Fifty to 90 percent of adults have antibodies to HSV, but only about 30 percent have antibodies specific to HSV-2.18 Type-specific serologic assays can be used to confirm HSV infection in persons with a questionable history or in those who have unrecognized or subclinical infections. Serologic testing also is helpful in the presence of a false-negative culture, which is common in patients with recurrent infection or healing lesions.18 Because HSV-2 infection is almost exclusively sexually acquired, HSV-2 antibodies are consistent with an anogenital infection. However, HSV-1 antibodies may be present in anogenital and orolabial infections; they cannot be used to differentiate between infections.2 If HSV antibodies are present, testing for other causes of genital ulcers (e.g., syphilis, chancroid) should be considered, especially in high-risk populations. It is important to ask laboratories for glycoprotein G–specific HSV serology, because some still use older assays that do not accurately distinguish HSV-1 from HSV-2. Universal screening for HSV antibodies is not recommended.2
Treatment
Effective oral antiviral medications are available for the treatment of genital herpes (Table 3).2,19 These agents offer clinical benefit but do not cure the disease,2 despite early animal models20,21 suggesting that antiviral medications may affect the long-term natural history of HSV infection. These drugs can be used for episodic treatment or long-term suppressive therapy. Acyclovir (Zovirax), a guanosine analog that inhibits viral DNA synthesis, is the oldest and most-studied medication. It has poor bioavailability and a short half-life, which necessitates frequent dosing. Valacyclovir (Valtrex), a prodrug that metabolizes to acyclovir, has better bioavailability and requires less frequent dosing than acyclovir. Famciclovir (Famvir) is a purine analog that has high bioavailability. All have rare, similar side effects, which include nausea, vomiting, headache, and diarrhea.19
| Drug | Side effects | Indication | Oral dosage | Cost (generic)* |
|---|---|---|---|---|
| Acyclovir (Zovirax) | Nausea, vomiting, headache, diarrhea, vertigo, myalgia, rash | Initial infection | 400 mg three times daily for 7 to 10 days | $63 to 93 ($45 to 65) |
| 200 mg five times daily for 7 to 10 days | 56 to 80 (36 to 51) | |||
| Recurrences | 400 mg three times daily for 5 days | 47 (32 to 34) | ||
| 200 mg five times daily for 5 days | 40 (25 to 28) | |||
| 800 mg twice daily for 5 days | 60 (40 to 42) | |||
| Suppressive therapy | 400 mg twice daily | 187 (129) | ||
| Famciclovir (Famvir) | Headache, nausea, vomiting, fatigue, paresthesias, pruritus | Initial infection | 250 mg three times daily for 7 to 10 days | 92 to 132 |
| Recurrences | 125 mg twice daily for 5 days | 40 | ||
| Suppressive therapy | 250 mg twice daily | 263 | ||
| Valacyclovir (Valtrex) | Headache, nausea, vomiting, dizziness | Initial infection | 1 g twice daily for 7 to 10 days | 112 to 160 |
| Recurrences | 500 mg twice daily for 3 to 5 days | 26 to 44 | ||
| 1 g daily for 5 days | 40 | |||
| Suppressive therapy | 500 mg daily | 131 | ||
| 1 g daily (in patients with more than nine episodes per year) | 239 |
FIRST CLINICAL EPISODE
Most patients with initial genital HSV infection should receive antiviral therapy.2 The main goals of initial treatment are to improve symptoms and speed recovery. One study22 of acyclovir showed that treatment decreased constitutional symptoms by three days, local pain by two days, viral shedding by seven days, time until all lesions were crusted by three days, and time until all lesions were healed by six days. All of the antiviral medications currently used to treat HSV infection have been shown to decrease by several days the time until all lesions are crusted and healed, as well as localized pain, constitutional symptoms, and viral shedding.19
EPISODIC THERAPY
Episodic therapy is best for HSV-infected patients who have mild and infrequent recurrences.23 The treatment goal is to diminish symptoms and infectivity during recurrences, rather than reduce the frequency of recurrences. To be effective, treatment should be started during the prodromal phase or within one day of lesion onset; therefore, it is important that a prescription is readily available.2 Early therapy provides a small but statistically significant benefit.23
SUPPRESSIVE THERAPY
Patients with frequent recurrences overwhelmingly choose suppressive therapy.24 It is recommended for patients with more than six episodes per year, but patient preferences also should be considered.25 Suppressive therapy reduces recurrences by 70 to 80 percent in patients who have frequent outbreaks (i.e., more than six per year), and it probably is effective in patients with fewer recurrences.26 Long-term safety and effectiveness have been well documented in patients receiving daily therapy with acyclovir for as long as six years, and in patients who have taken valacyclovir or famciclovir for one year.24,27,28 The development of drug resistance is rare in immunocompetent patients.29 For the most part, treatment is well tolerated and effective and is not associated with significant side effects. Suppressive therapy has been shown to reduce the risk of transmission significantly in heterosexual, HSV-2–discordant couples, but therapy has not been shown to fully eliminate asymptomatic viral shedding.30,31
Few studies have compared valacyclovir or famciclovir with acyclovir for suppressive therapy, but the existing studies suggest that all have comparable clinical outcomes.24,27 Therefore, cost and dosing frequency are important considerations in selecting a drug for prolonged treatment.2 Because the number of outbreaks may decrease over time, interruption of therapy should be discussed at yearly intervals to assess the need for continued therapy.2
SEVERE DISEASE
Intravenous acyclovir should be considered in patients with severe or disseminated disease (e.g., pneumonitis, hepatitis, central nervous system involvement).2 The recommended intravenous dosage is 5 to 10 mg per kg every eight hours for two to seven days, or until obvious clinical improvement occurs. The patient then can be switched to oral acyclovir to complete at least 10 days of total therapy.2
OTHER THERAPIES
During HSV outbreaks, patients should keep the affected area clean and dry to prevent secondary infections. Loose-fitting clothing and cotton underwear are helpful. Patients should avoid touching the lesions and should wash their hands after any contact with the sores. An ice pack or baking soda compress may be soothing. Topical acyclovir is less effective than oral acyclovir, and its use should be discouraged.32
Small studies have shown some benefit with the use of 1,000 mg of L-lysine given orally three times daily,33 125 mg per day of aspirin given orally,34 creams or gels containing licorice root (Glycyrrhiza glabra) applied three times daily,35 lemon balm (Melissa officinalis) applied four times daily,36 topical zinc applied daily,37 and 0.5 percent Aloe vera cream applied three times daily.38 However, studies of all of these remedies must be considered preliminary until randomized controlled trials are conducted. Future treatments may include immunomodulators such as imiquimod (Aldara) and a variety of microbicides and vaccines to prevent primary infection.39,40
Psychosocial Implications and Counseling
Although HSV infection is a self-limited disorder, it has a tremendous impact on affected patients, and counseling is crucial to management. The principal goals of counseling are to help patients cope with the infection and to prevent sexual and perinatal transmission.2 Informing the patient of the diagnosis can be a delicate matter; because many patients are in considerable disbelief, it may be beneficial to wait until after the initial outbreak resolves to discuss the chronic aspects of the disease.2 Numerous resources for patients are available (Table 4). Many patients may experience a sense of loneliness and isolation, as well as anxiety, diminished self-esteem, reluctance to initiate close relationships, and fear of initiating sexual relationships and sexual expression. A minority of patients may experience a deepening depression with each recurrence, and all aspects of their lives, including job performance, may be affected.8
| American Social Health Association | Web site:http://www.ashastd.org |
| Centers for Disease Control and Prevention | Web site:http://www.cdc.gov/std/Herpes/STDFact-Herpes.htm |
| Herpes Web | Web site:http://www.herpesweb.net |
| International Herpes Management Forum | Web site:http://www.ihmf.org/Patient/PatientResources.asp |
| National Herpes Hotline | Telephone: (919) 361–8488 (9 a.m. to 6 p.m. EST, Monday through Friday) |
| National STD & AIDS Hotline | Telephone: (800) 227-8922 or (800) 342-2437 (24 hours per day, 7 days per week) |
| En Español: (800) 344-7432 (8 a.m. to 2 a.m. EST, 7 days per week) |
By educating the patient about the disease process, the physician can help empower the patient to manage the disease. Patient concerns usually include the severity and frequency of clinical outbreaks, sexual relationships and transmission of the disease to others, and whether the disease will affect future childbearing.2 The Centers for Disease Control and Prevention has recommended specific messages to be addressed when counseling patients (Table 5).2
| Abstain from sexual activity when lesions or prodromal symptoms are present. |
| Antiviral therapy is available and effective but does not cure the infection. |
| Asymptomatic patients diagnosed by serologic testing should receive the same counseling messages as those with symptomatic infections. |
| Patients should inform current and future sex partners that they have genital herpes. |
| Patients should be informed about the natural history of the disease, including recurrence of episodes. |
| Sex partners may be infected even without symptoms, and serologic testing can determine whether they are at risk. |
| Sexual transmission can occur during asymptomatic periods. |
| When they cover infected areas, latex condoms can reduce the risk of transmission and should be used consistently. |