In the 1980s, after a steady decline during preceding decades, there was a resurgence in the rate of tuberculosis in the United States that coincided with the acquired immunodeficiency syndrome epidemic. Disease patterns since have changed, with a higher incidence of disseminated and extrapulmonary disease now found. Extrapulmonary sites of infection commonly include lymph nodes, pleura, and osteoarticular areas, although any organ can be involved. The diagnosis of extrapulmonary tuberculosis can be elusive, necessitating a high index of suspicion. Physicians should obtain a thorough history focusing on risk behaviors for human immunodeficiency virus (HIV) infection and tuberculosis. Antituberculous therapy can minimize morbidity and mortality but may need to be initiated empirically. A negative smear for acid-fast bacillus, a lack of granulomas on histopathology, and failure to culture Mycobacterium tuberculosis do not exclude the diagnosis. Novel diagnostic modalities such as adenosine deaminase levels and polymerase chain reaction can be useful in certain forms of extrapulmonary tuberculosis. In general, the same regimens are used to treat pulmonary and extrapulmonary tuberculosis, and responses to antituberculous therapy are similar in patients with HIV infection and in those without. Treatment duration may need to be extended for central nervous system and skeletal tuberculosis, depending on drug resistance, and in patients who have a delayed or incomplete response. Adjunctive corticosteroids may be beneficial in patients with tuberculous meningitis, tuberculous pericarditis, or miliary tuberculosis with refractory hypoxemia.
From 1985 until 1992 there was a resurgence of tuberculosis in the United States that coincided with the epidemic of acquired immunodeficiency syndrome (AIDS). Although the U.S. incidence of tuberculosis has since been in decline, this disease remains a major problem for much of the world, with a global prevalence of infection estimated at 32 percent.2 Thus, the percentage of U.S. cases that occur among foreign-born persons is increasing (53 percent in 2003).1 Extrapulmonary tuberculosis has become more common since the advent of human immunodeficiency virus (HIV) infection.3
SORT: KEY RECOMMENDATIONS FOR PRACTICE
| Clinical recommendation | Evidence rating | References |
|---|---|---|
| All patients with tuberculosis should have counseling and testing for HIV infection. | C | 7,8 |
| All confirmed cases of active tuberculosis should be reported to the local health department. | C | 7 |
| Adjunctive corticosteroid therapy is recommended, based on limited evidence, in patients with tuberculous meningitis or pericarditis, and in miliary tuberculosis with refractory hypoxemia. | B | 7,9,10 |
| Patients should be monitored using directly observed therapy whenever feasible to ensure compliance and prevent emergence of drug resistance. | C | 12 |
| Patients should be screened for latent tuberculosis infection or active disease before initiation of therapy with a TNF-α inhibitor. | C | 42,43 |
| Patients with pulmonary or laryngeal tuberculosis should be placed in respiratory isolation until they are no longer infectious. | B | 7 |
HIV = human immunodeficiency virus; TNF = tumor necrosis factor.
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, see page 1639 or https://www.aafp.org/afpsort.xml.
Extrapulmonary Tuberculosis and HIV Infection
Extrapulmonary involvement can be seen in more than 50 percent of patients with concurrent AIDS and tuberculosis.3–5 The risk of extrapulmonary tuberculosis and mycobacteremia increases with advancing immunosuppression.6 Unique features of AIDS-associated tuberculosis include extra-pulmonary disease, disseminated disease, rapid progression, visceral lymphadenopathy, tissue abscesses, and negative tuberculin skin test. Response to antituberculous therapy is favorable and similar to that of patients without HIV infection, although adverse drug reactions occur more commonly in those with HIV infection. It is unclear whether patients with HIV infection have a higher risk of relapse. Infectious disease consultation is advisable given complex drug-drug interactions and the risk of paradoxical response or immune reconstitution.
Principles of Management
Clinical clues that should prompt suspicion of extrapulmonary tuberculosis are listed in Table 1. Patients with suspected tuberculosis should have appropriate specimens sent for acid-fast bacillus (AFB) staining, mycobacterial culture, and histology. Hospitalization is not necessary for tuberculosis to be diagnosed unless clinically indicated. Hospitalized patients in whom infectious (i.e., pulmonary or laryngeal) tuberculosis is suspected should be placed in an airborne-infection isolation room and should wear a surgical mask during transport and in waiting areas.7 Health care workers and visitors entering the isolation room should wear at least N95 disposable respirators, as should health care workers performing procedures such as sputum induction, bronchoscopy, jet irrigation of abscesses, and autopsies. All patients with tuberculosis should have counseling and testing for HIV infection. The local health department should be notified of all confirmed cases of tuberculosis.7
TABLE 1 Clinical Clues to Prompt Suspicion of Extrapulmonary Tuberculosis
| Ascites with lymphocyte predominance and negative bacterial cultures |
| Chronic lymphadenopathy (especially cervical) |
| CSF lymphocytic pleocytosis with elevated protein and low glucose |
| Differential diagnosis of Crohn’s disease and amebiasis |
| Exudative pleural effusion with lymphocyte predominance, negative bacterial cultures, and pleural thickening |
| HIV infection |
| Joint inflammation (monoarticular) with negative bacterial cultures |
| Persistent sterile pyuria |
| Tuberculosis-endemic country of origin |
| Unexplained pericardial effusion, constrictive pericarditis, or pericardial calcification |
| Vertebral osteomyelitis involving the thoracic spine |
CSF = cerebrospinal fluid; HIV = human immunodeficiency virus.
A six- to nine-month regimen (two months of isoniazid [INH], rifampin [Rifadin], pyrazinamide, and ethambutol [Myambutol], followed by four to seven months of isoniazid and rifampin) is recommended as initial therapy for all forms of extrapulmonary tuberculosis unless the organisms are known or strongly suspected to be resistant to the first-line drugs.8 For patients with central nervous system tuberculosis, including meningitis, at least nine to 12 months of therapy is recommended. Extended therapy also may be required for patients with bone and joint tuberculosis, delayed treatment response, or drug resistance. Adjunctive corticosteroids may be useful in patients who have tuberculous meningitis, tuberculous pericarditis, or miliary tuberculosis with refractory hypoxemia.7–11 Physicians should consider noncompliance, malabsorption, and drug resistance as possible reasons for delayed or suboptimal response to appropriate therapy. Directly observed therapy is strongly recommended to encourage medication compliance.12 A detailed discussion of baseline evaluation, antituberculous therapy, and follow-up is beyond the scope of this article but can be found elsewhere.8
Tuberculous Lymphadenitis
Lymphadenitis is the most commonly occurring form of extrapulmonary tuberculosis. Cervical adenopathy is most common, but inguinal, axillary, mesenteric, mediastinal, and intramammary involvement all have been described.13–17 Although previously considered a disease of childhood, lymphadenitis has a peak age of onset of 20 to 40 years, and in the United States it is most common in women and immigrants. Patients without HIV infection typically present with chronic, nontender lymphadenopathy.15,17 Patients with HIV infection usually present with fever, night sweats, and weight loss.5,18 The nodes are discrete, firm, and nontender; with time, a firm mass of matted nodes becomes visible (Figure 1). If untreated, the nodes become fluctuant and drain spontaneously with sinus tract formation.
Figure 1.
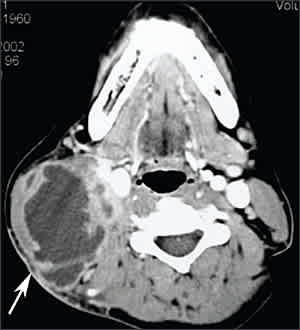
Tuberculous lymphadenitis. Computed tomographic scan of the neck reveals a heterogeneous mass in the right posterior cervical space (arrow) with central necrosis.
Most patients have a positive tuberculin skin test result and a normal result on chest radiography.13,15 Excisional biopsy of the lymph nodes with histology, AFB stain, and mycobacterial culture is the diagnostic procedure of choice.16,17 The utility of fine-needle aspiration in patients without HIV infection is highly variable.17 Fine-needle aspiration is more reliable in patients with HIV infection because of the higher mycobacterial burden, and in these patients should be the initial diagnostic procedure.17 Polymerase chain reaction (PCR) for Mycobacterium tuberculosis on the fine-needle aspiration specimen enhances test sensitivity.
During antituberculous therapy (see Principles of Management), affected nodes may enlarge or new nodes may appear, representing an immune response to killed mycobacteria. A similar phenomenon in patients with HIV infection who begin concurrent antiretroviral therapy is a result of immune reconstitution. Lymph node excision usually is not indicated. When lymph nodes are fluctuant and ready to drain, aspiration or incision and drainage appear to be beneficial.
Pleural Tuberculosis
In the United States, pleural tuberculosis accounts for about 5 percent of all tuberculosis cases.19 Tuberculous effusions can follow early postprimary, chronic pulmonary, or miliary tuberculosis. Pleural tuberculosis often is an acute illness with cough, pleuritic chest pain, fever, or dyspnea.
Chest radiography typically reveals a small to moderate, unilateral pleural effusion; about 20 percent of patients have associated pulmonary lesions.20 Computed tomography (CT) of the chest may show lymphadenopathy, pulmonary infiltrates, or cavitation not obvious on chest radiography (Figure 2). Pleural thickening of more than 1 cm is seen in most instances.21
Figure 2.
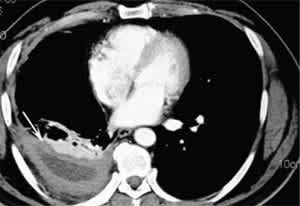
Tuberculous empyema. Computed tomographic scan showing loculated pleural fluid and pleural thickening (arrow) in the right chest with associated right lower lobe atelectasis.
Pleural fluid is exudative with a lymphocyte predominance (i.e., more than 50 percent of white blood cells in more than 90 percent of effusions)16,20; in patients with less than two weeks of symptoms, an initial predominance of neutrophils may be seen. Pleural fluid glucose and pH can be low or normal. AFB smears of pleural fluid are seldom positive (5 percent of cases) unless the patient has tuberculous empyema. Pleural fluid cultures for M. tuberculosis are positive in less than 40 percent of cases.20 The combined sensitivity of the analyses of pleural biopsy specimens (i.e., observation for caseating granulomas, AFB smear, and culture) is more than 90 percent.20 Tuberculin skin test results are positive in two thirds of patients. Biochemical markers such as adenosine deaminase, interferon gamma, and lysozyme in the pleural fluid can be useful. In one study,20 a high level of adenosine deaminase (greater than 47 U per L [783 nkat per L]) was seen in 99 percent of tuberculous effusions. In countries with a low prevalence of tuberculosis, such as the United States, a normal or low level of pleural fluid adenosine deaminase has a high negative predictive value and can be used to exclude tuberculous pleurisy.22,23 Pleural fluid PCR for M. tuberculosis has a sensitivity of 80 percent and a specificity of 100 percent.24
Tuberculous pleurisy responds well to medical therapy, with resorption of pleural fluid in six to 12 weeks. The effusion may resolve without therapy, but tuberculosis later recurs. Rare complications include bronchopleural fistula, empyema, and fibrothorax.
Skeletal Tuberculosis
Bone and joint tuberculosis may account for up to 35 percent of cases of extrapulmonary tuberculosis. Skeletal tuberculosis most often involves the spine, followed by tuberculous arthritis in weight-bearing joints and extraspinal tuberculous osteomyelitis.25–27
Spinal tuberculosis (Pott’s disease) most commonly involves the thoracic spine. Infection begins in the anteroinferior aspect of the vertebral body with destruction of the intervertebral disc and adjacent vertebrae (Figure 3). The resulting anterior wedging and angulation of adjacent vertebral bodies with disc space obliteration are responsible for the palpable spinal prominence (gibbus) and a classic radiographic appearance. Paraspinal and psoas abscesses can develop, with extensions to the surface or adjacent tissues (Figure 4). Patients present with local pain, constitutional symptoms, or paraplegia secondary to cord compression.
Figure 3.
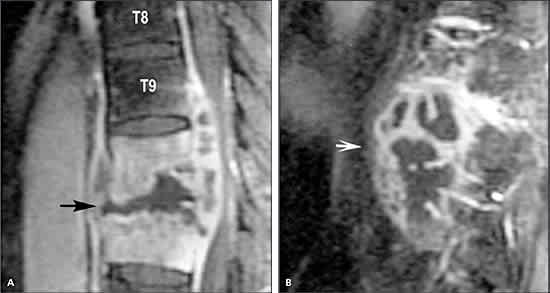
Spinal tuberculosis. Magnetic resonance imaging of the spine revealing osteomyelitis involving T10 and T11 vertebral bodies and disc space (A; arrow) and an adjacent multiloculated paravertebral abscess (B; arrow).
Figure 4.
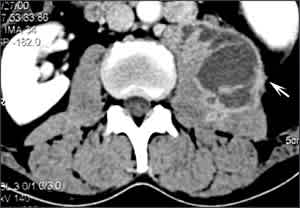
Psoas abscess. Computed tomographic scan of the abdomen showing a left iliopsoas abscess (arrow) that likely originated from tuberculous osteomyelitis involving the T12, L1, and L2 vertebrae.
Articular tuberculosis is a slowly progressive mono-arthritis of the hip or knee. Presentation is indolent with pain, joint swelling, and decreased range of motion. Draining sinuses and abscesses are seen in chronic cases. Systemic symptoms usually are absent. Radiographic changes are nonspecific and include soft tissue swelling, juxta-articular osteopenia, joint space narrowing, and subchondral erosions (Figure 5).26
Figure 5.
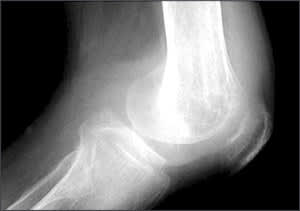
Osteoarticular tuberculosis. Radiograph of the right knee showing a large effusion, osteopenia, joint space narrowing, and lucencies in the distal femur.
Extraspinal tuberculous osteomyelitis often presents with local pain and can involve any bone. Involvement of adjacent structures may result in complications such as carpal tunnel syndrome, tenosynovitis, and facial palsy.
Chest radiography shows pulmonary disease in one half of patients with osteoarticular tuberculosis, but active pulmonary disease is uncommon.25 Magnetic resonance imaging may be helpful to assess the degree of bony destruction and to identify soft tissue extension and encroachment on adjacent structures such as the spinal cord.
To establish the diagnosis of skeletal or articular tuberculosis, a high index of suspicion is critical. Physicians should consider skeletal tuberculosis in patients with an indolent clinical course manifesting as osteomyelitis involving the thoracic spine or monoarticular septic arthritis with negative bacterial cultures. Arthrocentesis with mycobacterial cultures of synovial fluid yields positive results in up to 80 percent of patients with tuberculous arthritis. Synovial biopsy also may be diagnostic (caseating granulomas on histology or positive mycobacterial culture).26,27 Bone biopsy for culture and histology is required for diagnosis of tuberculous osteomyelitis.
Surgery may be necessary to drain abscesses, debride infected tissue, or stabilize the spine and relieve spinal cord compression.28 In the absence of neurologic impairment, unstable spine, or spinal cord compression, medical therapy alone (see Principles of Management)8,28 should result in an excellent response.
Central Nervous System Tuberculosis
Central nervous system tuberculosis includes tuberculous meningitis (the most common presentation), intracranial tuberculomas, and spinal tuberculous arachnoiditis. Meningitis results from intense inflammation following rupture of a subependymal tubercle into the subarachnoid space.29 The ensuing arachnoiditis encases both cranial nerves and penetrating vessels, leading to cranial nerve palsies and communicating hydrocephalus. Cranial vasculitis may lead to focal neurologic deficits. Hypersensitivity to tuberculoproteins may cause meningismus and typical cerebrospinal fluid (CSF) findings. Cerebral edema causes impairment of consciousness, seizures, and raised intracranial pressure, whereas tuberculomas can manifest as space-occupying lesions.30
An initial phase of malaise, headache, fever, or personality change is followed in two to three weeks by protracted headache, meningismus, vomiting, confusion, and focal neurologic findings. If untreated, mental status deteriorates into stupor or coma.31 Atypical presentations include a rapid progression simulating pyogenic meningitis, subtle cognitive decline mimicking dementia, and a predominant syndrome of encephalitis. Convulsions can occur at all stages of the illness.
A high index of suspicion is necessary for timely diagnosis and prompt initiation of therapy. CSF typically reveals moderate lymphocytic pleocytosis (100 to 500 cells per μL [0.10 to 0.50×109 per L])16; initially, a neutrophilic predominance can be seen. CSF protein levels range from 100 to 500 mg per dL (1,000 to 5,000 mg per L) and can be extremely high (2 to 6 g per dL [20 to 60 g per L]), with xanthochromia in the presence of subarachnoid block. CSF glucose concentration usually is less than 45 mg per dL (2.50 mmol per L). AFB smears on CSF are positive in 10 to 90 percent of patients; sensitivity can be improved if large volumes of CSF from multiple lumbar punctures are examined, CSF is centrifuged and AFB smears are performed on the pellicle, or an experienced reviewer examines several high-powered fields.16,30,31 CSF culture for M. tuberculosis is positive in 45 to 90 percent of cases but takes four to six weeks. CSF PCR for M. tuberculosis has a sensitivity of 56 percent and a specificity of 98 percent, and therefore should not be used to exclude tuberculous meningitis.32 An elevated CSF adenosine deaminase level supports the diagnosis in the appropriate clinical setting.33 A CT scan of the head with contrast may reveal basilar arachnoiditis, infarction, hydrocephalus, or tuberculomas.
Empiric antituberculous therapy (see Principles of Management) should be initiated as soon as clinical, laboratory, or imaging findings suggest tuberculous meningitis. Delay in initiation of therapy has been directly associated with adverse outcomes. Antituberculous therapy is recommended for at least nine to 12 months.8 Adjunctive corticosteroid therapy with dexamethasone (Decadron) for the initial six to eight weeks in patients with tuberculous meningitis has been associated with reduced mortality and fewer neurologic sequelae.8,11,34 Mortality is highest in patients younger than five years, those older than 50 years, and those in whom illness has been present for longer than two months.
Abdominal Tuberculosis
Abdominal tuberculosis may involve the gastrointestinal tract, peritoneum, mesenteric lymph nodes, or genito-urinary tract. Other organs (e.g., liver, spleen, adrenal glands) usually are affected as a consequence of miliary tuberculosis.
GASTROINTESTINAL TUBERCULOSIS
Tuberculous enteritis can result from swallowing of infected sputum, ingestion of contaminated food, hematogenous spread, and direct extension from adjacent organs.35 The intestinal lesions can be ulcerative (most common), hypertrophic, or ulcero-hypertrophic. Symptoms include abdominal pain, diarrhea, weight loss, and fever. Melena, rectal bleeding, and abdominal tenderness also can be present. A mass in the right lower quadrant is palpable in 25 to 50 percent of patients.
The ileocecal area and jejunoileum are the most common sites of involvement. Complications include obstruction, perforation, and fistula formation. Rectal lesions usually present as anal fissures, fistulas, or perirectal abscesses.16 Barium contrast studies and colonoscopy may show ulcers, strictures, a deformed cecum, incompetent ileocecal valve, or fistulas. An abdominal CT scan can define extraluminal pathology, especially lymphadenopathy.
It is essential to distinguish tuberculous enteritis from Crohn’s disease because initiation of immunosuppressive therapy in a patient with tuberculous enteritis can lead to dissemination. Definitive diagnosis is based on histopathology and culture of biopsy specimens obtained by colonoscopy or laparotomy. Differential diagnosis of tuberculous enteritis includes Crohn’s disease, amebiasis, neoplasm, Yersinia infection, and actinomycosis.35 A six-month course of antituberculous therapy is recommended. Surgery is reserved for patients with complications.
TUBERCULOUS PERITONITIS
The risk of tuberculous peritonitis is greater in patients with HIV infection or cirrhosis and in those undergoing continuous ambulatory peritoneal dialysis.35 Tuber culous peritonitis results from reactivation of latent foci in the peritoneum. Patients present with the insidious onset of ascites, abdominal pain, and fever. Peritoneal fluid is exudative, with a serumascites albumin gradient of less than 1.1 g per dL (11 g per L). Peritoneal fluid leukocyte count varies from 150 to 4,000 per mm3 (0.15 to 4.00 × 109 per L) with a lymphocytic predominance16; a neutrophilic pleocytosis may be seen with tuberculous peritonitis complicating continuous ambulatory peritoneal dialysis.36 Centrifugation of 1 L of peritoneal fluid enhances the yield of AFB smear and mycobacterial culture. Peritoneal fluid adenosine deaminase level has a high sensitivity and specificity if a cutoff value greater than 33 U per L (550 nkat per L) is used. Peritoneal biopsy guided by laparoscopy or mini-laparotomy can be diagnostic in more than 95 percent of patients and should be strongly considered.35
GENITOURINARY TUBERCULOSIS
Renal disease may be the result of direct infection of the kidney and lower urinary tract or may present as secondary amyloidosis. Patients present with dysuria, hematuria, or flank pain.37,38 More than 90 percent of asymptomatic patients have sterile pyuria with or without microscopic hematuria.37,38 Intravenous pyelography may show a “motheaten calyx” or papillary necrosis. Abdominal CT scan may reveal renal calcifications, calculi, scarring, hydronephrosis, or evidence of extrarenal disease (e.g., ureteral strictures; contracted bladder; calcifications in the vas deferens, seminal vesicles, or prostate). Mycobacterial culture of three morning urine specimens establishes the diagnosis in 90 percent of patients.37 Nephrectomy rarely is indicated for persistent flank pain or hypertension.8 Renal function usually is preserved, except in the setting of tuberculous interstitial nephritis.
Male genital tuberculosis usually is associated with renal tuberculosis. It involves the prostate, seminal vesicles, epididymis, and testes, in order of incidence. Patients usually present with a scrotal mass (Figure 6), and diagnosis is made by surgery. Oligospermia is common and may be persistent. Female genital tuberculosis begins in the endosalpinx and can spread to the peritoneum, endometrium, ovaries, cervix, and vagina.38 Patients present with pelvic pain, infertility, and vaginal bleeding. Response to chemotherapy is excellent for all forms of genital tuberculosis; surgery in women is necessary for large tubo-ovarian abscesses.8
Figure 6.
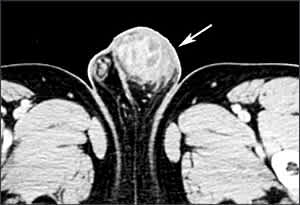
Testicular tuberculosis. Computed tomographic scan of the pelvis showing a large, irregular, mixed solid and cystic left testicular mass (arrow).
Other Forms
Three additional forms of extrapulmonary tuberculosis warrant discussion: miliary tuberculosis, tuberculous pericarditis, and tuberculosis associated with tumor necrosis factor-3 (TNF-α) inhibitors.
MILIARY TUBERCULOSIS
The term miliary tuberculosis refers to any progressive, disseminated form of tuberculosis; the disease can occur during primary dissemination or after years of untreated tuberculosis. Miliary disease is seen in 10 percent of patients who have AIDS and pulmonary tuberculosis, and in 38 percent of those who have AIDS and extra-pulmonary tuberculosis.5 Presenting symptoms include fever, chills, night sweats, weight loss, and anorexia. Clinical manifestations depend on the organs involved. Fulminant disease including septic shock, acute respiratory distress syndrome, and multiorgan failure has been described. A chest radiograph or CT scan reveals numerous 2- to 3-mm nodules scattered throughout the lung in more than 85 percent of patients (Figure 7).16,39 Common laboratory abnormalities include normochromic anemia, leukopenia or leukocytosis, elevated sedimentation rate, and hyponatremia.40 Examination of the sputum, bronchoalveolar lavage, gastric washings, CSF, blood culture, or biopsies of liver and bone marrow may be necessary for diagnosis.16 A tuberculin skin test result is positive in less than 50 percent of patients. Antituberculous therapy (and corticosteroids in select situations) is as previously outlined (see Principles of Management).
Figure 7.
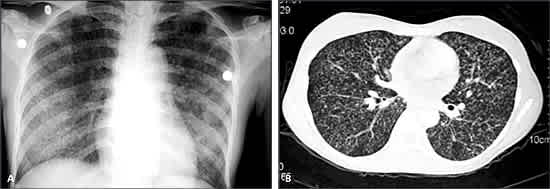
Miliary tuberculosis. Chest radiograph (A) and chest computed tomographic scan (B) showing bilateral miliary nodular pattern.
TUBERCULOUS PERICARDITIS
Tuberculous pericarditis develops secondary to contiguous spread from mediastinal nodes, lungs, spine, or sternum, or during miliary dissemination. The onset may be abrupt or insidious with symptoms such as chest pain, dyspnea, and ankle edema. Cardiomegaly, tachycardia, fever, pericardial rub, pulsus paradoxus, or distended neck veins may be found on examination.41 Pericardial biopsy yields a definitive diagnosis more often than pericardial fluid alone.41 In addition to antituberculous therapy, corticosteroids are recommended to hasten resolution of symptoms and to reduce reaccumulation of fluid.8,41 The risk of progression to constrictive pericarditis or mortality is not altered by corticosteroids.11 Open pericardial drainage is favored over repeated pericardiocentesis.41
TNF-α INHIBITOR–ASSOCIATED TUBERCULOSIS
Two recent reports describe several cases of active tuberculosis occurring after treatment with the TNF-α inhibitors infliximab (Remicade)42 and etanercept (Enbrel),43 mainly in patients with rheumatoid arthritis or Crohn’s disease. Tuberculosis was diagnosed sooner after initiation of infliximab than after initiation of etanercept (median of 12 weeks versus 12 months).42,43 Extrapulmonary tuberculosis accounted for 52 to 57 percent of cases. As a result, it is now recommended that patients be screened for latent tuberculosis infection or active disease before initiation of therapy with a TNF-α inhibitor.

