Tick-borne relapsing fever is characterized by recurring fevers separated by afebrile periods and is accompanied by nonspecific constitutional symptoms. It occurs after a patient has been bitten by a tick infected with a Borrelia spirochete. The diagnosis of tick-borne relapsing fever requires an accurate characterization of the fever and a thorough medical, social, and travel history of the patient. Findings on physical examination are variable; abdominal pain, vomiting, and altered sensorium are the most common symptoms. Laboratory confirmation of tick-borne relapsing fever is made by detection of spirochetes in thin or thick blood smears obtained during a febrile episode. Treatment with a tetracycline or macrolide antibiotic is effective, and antibiotic resistance is rare. Patients treated for tick-borne relapsing fever should be monitored closely for Jarisch-Herxheimer reactions. Fatalities from tick-borne relapsing fever are rare in treated patients, as are subsequent Jarisch-Herxheimer reactions. Persons in endemic regions should avoid rodent- and tick-infested areas and use insect repellents and protective clothing to prevent tick bites.
Tick-borne relapsing fever (TBRF) is transmitted by Ornithodoros ticks infected with one of several different Borrelia spirochete species, most commonly Borrelia hermsii and Borrelia turicata. The highest concentrations of TBRF in the United States are in the western states containing the Cascade, San Bernardino, Sierra Nevada, and Rocky Mountain ranges.1–3
SORT: KEY RECOMMENDATIONS FOR PRACTICE
| Clinical recommendation | Evidence rating | References |
|---|---|---|
| Diagnosis of TBRF should be made by detecting spirochetes in a thin and thick smear of peripheral blood prepared with Wright’s or Giemsa stain or by culture isolation. | C | 1, 13 |
| Doxycycline (Vibramycin, 100 mg orally, twice daily for seven to 10 days) or tetracycline (500 mg, four times daily for seven to 10 days) is recommended for treatment of TBRF. | C | 4, 17, 25 |
| Applying an insect repellent containing DEET to the skin and clothing is recommended for prevention of TBRF. | C | 9 |
TBRF = tick-borne relapsing fever; DEET =N,N- diethyl-m- toluamide.
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, see page 1949 or https://www.aafp.org/afpsort.xml.
Consideration of TBRF is critical in patients presenting with recurring fevers. Cardiac and neurologic complications may develop with TBRF, with long-term sequelae that may be permanent. Reviewing a broad differential diagnosis (Table 11, 3–6) for fever when TBRF is suspected will help rule out other possible causes of fever that, if misdiagnosed, could result in significant morbidity or mortality (e.g., Lyme disease, malaria in a returning traveler).
TABLE 1 Differential Diagnosis of Tick-Borne Relapsing Fever
| Foreign travel | General infection | Occupational and recreational exposure |
|---|---|---|
|
|
|
Illustrative Case
A 60-year-old man presented to an emergency department in Boise, Idaho, with a one-month history of intermittent fevers and headaches. The initial fever peaked at 40.6°C (105°F), was accompanied by a headache, lasted for three days, and resolved spontaneously after an episode of shaking chills. The patient had a second fever 13 days later, peaking at 39.4°C (103°F) with diffuse abdominal pain and blurred vision. This fever lasted two days and resolved spontaneously. At the time of the second fever, a family physician assessed the patient. Routine blood tests were normal except an elevated total bilirubin level (6.0 mg per dL) [102.6 mmol per L]. An abdominal computed tomography scan was normal. The patient was referred to an ophthalmologist, who diagnosed iritis.
The patient developed a third fever with headache and visual symptoms 26 days after the initial symptoms, prompting presentation to the emergency department. Past medical history and family history were unremarkable. The patient took no medications. He reported traveling to Vietnam four years earlier and Honduras 18 months earlier, and he took malaria prophylaxis for both trips. The patient also had been camping in the Bear Valley region of Idaho. Review of systems was otherwise negative.
On examination, the patient was febrile (39.7°C [103.4°F]), tachycardic (113 beats per minute), and hypertensive (195/107 mm Hg). Other vital signs were stable, and physical examination was normal.
A complete blood count was normal. Manual differential demonstrated 81 percent segmented neutrophils and a low lymphocyte count (6 percent). Total bilirubin was elevated (2.3 mg per dL [39.33 mmol per L]; direct bilirubin, 0.4 mg per dL [6.84 mmol per L]). A reticulocyte count was elevated (3.37 percent). Urinalysis was positive for protein (1+). Scant spirochetes were detected in a peripheral blood smear prepared with Giemsa stain (Figure 1).
Figure 1
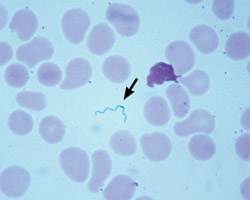
A spirochete (arrow) in a thin smear of peripheral blood with Giemsa stain.
A presumptive diagnosis of Borrelia infection was made, and the patient was admitted and started on tetracycline (500 mg orally, four times daily). The patient’s maximum temperature in the hospital was 39.3°C (102.8°F), which returned to normal after 24 hours. Visual symptoms resolved after 24 hours, and he was discharged 36 hours after admission and continued tetracycline for 10 days. Further discussion with the patient revealed that the area where he had been camping had a large squirrel population, though he did not recall being bitten by any insects.
Epidemiology
More than 450 cases of TBRF were reported between 1977 and 2000 in the United States.5 The majority of TBRF infections have occurred in Washington state and northern Idaho, with clusters of infections reported in Spokane County, Wash.; Estes Park, Colo.; New Mexico; Montana; and the north rim of the Grand Canyon.3,7–13
In North America, most cases of TBRF are associated with two species of Argasidae (soft) ticks, Ornithodoros hermsii and Ornithodoros turicata. Ornithodoros ticks are obligate blood feeders; they maintain continuous Borrelia infections in salivary gland tissue, and transmit spirochetes when feeding.
O. hermsii is found in the Pacific Northwest and the southwestern United States, and O. turicata is found sporadically from Kansas to central Mexico and California. Natural hosts for O. hermsii include squirrels and chipmunks. Hosts for O. turicata include cattle, rodents, pigs, snakes, tortoises, and possibly coyotes.
Human infection commonly occurs in the setting of a mountain cabin that has been infested with rodents and O. hermsii ticks (Figure 2). When rodents or other natural hosts vacate a cabin, humans may become the only available host. Patients usually are unaware of a tick bite or exposure.1,2,8 TBRF from B. turicata–infected ticks also commonly occurs after humans explore caves or crawl under buildings, and cases have been described in persons who have handled dead rodents.1,5 Blood feeding by soft-bodied ticks takes less than one hour and usually occurs at night.1,4
Figure 2
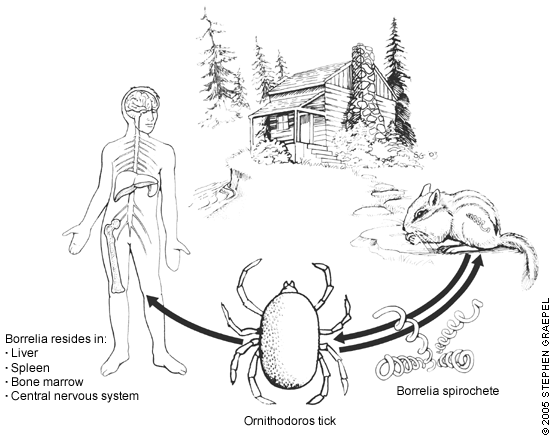
Typical setting for transmission of tick-borne relapsing fever.
Pathogenesis
Borrelia spirochetes do not infect blood cells directly, but the severity of TBRF correlates with the number of spirochetes in the bloodstream. An IgM immune response is capable of clearing one Borrelia serotype, but because of the organism’s ability to undergo antigenic variation, several serotypes may be present in the bloodstream at one time. With clearance of one serotype, less common serotypes proliferate, causing the relapsing fever pattern that characterizes TBRF.4,6,14
Borrelia spirochetes are capable of moving through or between vascular endothelial cells. During asymptomatic intervals, organisms sequester in the liver, spleen, bone marrow, and the central nervous system (CNS). The immune response may be unsuccessful in clearing Borrelia from the eye, brain, and cerebrospinal fluid (CSF), and relapsing fever spirochetes may persist in these locations for years.15,16
Clinical Presentation
TBRF should be considered in patients with recurrent fevers and a history of potential exposure to soft-bodied ticks. Fevers are sudden, high (≥ 39.2°C [≥ 102.5°F]), and are followed by an afebrile period. The first fever lasts three to six days and usually ends with a crisis of rigors, continued fever, hypertension, and tachycardia.1 Fever crises usually last less than 30 minutes and are followed by diaphoresis, hypotension, and a decreasing temperature. The interval between fevers ranges from four to 14 days.15
Headache, arthralgia, myalgia, neck stiffness, and nausea are most common with the initial fever episode (Table 21) . Neurologic symptoms including delirium, facial palsy, meningismus, and radiculopathy may occur and are more common after the second fever episode.1,17
Elevated pulse and blood pressure are common, especially during febrile episodes.5 Unilateral or bilateral Bell’s palsy or deafness from seventh or eighth cranial nerve involvement may be present. Meningitis or meningoencephalitis are serious consequences of TBRF and may result in residual hemiplegia or aphasia. Visual impairment from unilateral or bilateral iridocyclitis or panophthalmitis may be permanent. In contrast, altered mental status and nuchal rigidity not associated with other neurologic symptoms usually are the result of spirochetemia rather than direct CNS infection.17
A gallop heard on cardiac auscultation may suggest myocardial involvement; myocarditis is prominent in fatal cases of TBRF.18 Splenomegaly, often presenting as abdominal or left shoulder pain, is common. Hepatomegaly occurs in approximately 10 percent of patients with TBRF.5 Infection during pregnancy can lead to a spontaneous abortion or stillbirth. Transplacental transmission of spirochetes and infection at birth is possible. Children and pregnant women tend to have a more prolonged and serious disease course.5,19
Laboratory Diagnosis
TBRF is diagnosed by detecting spirochetes in thin and thick smears of peripheral blood prepared with Wright’s or Giemsa stain or by culture isolations between fever onset and peak.1,13 Direct and indirect immunofluorescence and experimental polymerase chain reaction testing are available in some laboratories.20–23
Mild leukocytosis, elevated erythrocyte sedimentation rate, and anemia may be present in patients with TBRF. Thrombocytopenia (i.e., ≥ 50,000 platelets per mm3), elevated serum unconjugated bilirubin levels, elevated aminotransferase levels, prolonged prothrombin and partial thromboplastin times, proteinuria, and microhematuria may be present. A prolonged corrected Q-T interval on electrocardiography may be present in persons with TBRF-induced myocarditis.3,4,16
Analysis of CSF is indicated if signs of meningitis or meningoencephalitis are present. Mononuclear pleocytosis, a mildly to moderately elevated protein level, and normal glucose levels in the CSF support the diagnosis of CNS Borrelia infection.5
Treatment
The recommended treatment for patients with TBRF is doxycycline (Vibramycin) at a dosage of 100 mg orally, twice daily for seven to 10 days, or tetracycline at a dosage of 500 mg, four times daily for seven to 10 days. When tetracycline is contraindicated (i.e., in children younger than eight years or pregnant women1), a macrolide antibiotic may be prescribed (e.g., erythromycin at a dosage of 500 mg orally, four times daily for seven to 10 days).4,19,24,25 Penicillins are effective but should be given intravenously, especially when CNS involvement is suspected. There is no evidence of antibiotic-resistant TBRF when using the above regimens.17
A Jarisch-Herxheimer reaction may occur within two hours of treatment in up to 54 percent of treated patients with TBRF.1 This reaction is caused by massive cytokine release and manifests as fever, tachycardia, chills, rigors, diaphoresis, and hypotension.5 Reaction duration usually is less than four hours, but observation should continue for 12 to 24 hours after administration of antibiotics. Jarisch-Herxheimer reactions occur more often after administration of tetracyclines.17 Cardiovascular collapse and death are rare complications. The use of acetaminophen, corticosteroids, and non-steroidal anti-inflammatory drugs does not modify outcomes significantly.1
When patients with TBRF are treated appropriately, the mortality rate is less than 1 percent.5 Indicators of poor prognosis include myocarditis, stupor or coma, poor hepatic function, and diffuse bleeding.
Prevention
Exposure to TBRF is decreased by rodent-proofing homes and avoiding animal burrows and caves. Spraying buildings with 0.5 percent malathion will reduce tick populations.4 The home environment also should be free of squirrels and chipmunks, and gloves should be worn when handling dead rodents.
Persons in tick-infected areas should wear pants and long-sleeved shirts and apply insect repellent containing N, N-diethyl-m-toluamide (DEET) to the skin and clothing, even before going to sleep. All patients with TBRF should be reported to local health departments for surveillance of outbreaks. Persons who hike and camp in endemic areas should perform daily body examinations to ensure that all ticks are promptly removed, and they should seek medical care immediately if signs or symptoms suggestive of TBRF develop.9

