Clinical Question
What is the risk of recurrence in a patient with prostate cancer who undergoes radical prostatectomy?
Evidence Summary
A previous Point-of-Care Guide1 identified a clinical decision rule that estimates the likelihood of an abnormal biopsy for prostate cancer. One of the next questions facing the patient and his physician is the likelihood of recurrence after surgery.
Several clinical decision rules have addressed this question. One is the University of California, San Francisco Cancer of the Prostate Risk Assessment score, which was developed based on 1,439 men who underwent prostatectomy.2 Recurrence occurred in 15 percent of patients and was defined as a prostate-specific antigen (PSA) of 0.2 ng per mL or greater on two consecutive occasions or the need for a second cancer treatment at least six months after surgery. The score used the preoperative PSA, Gleason grade, tumor stage, percentage of positive biopsies, and patient age. Each variable was assigned points, and the point total corresponded to a five-year recurrence rate (e.g., 5.6 percent for a score of 0, 17 percent for a score of 4, and 78 percent for a score of 7 or higher). Although promising, this score has not been prospectively validated.
The most widely validated score was developed by Kattan and colleagues.3 It was developed based on a group of 983 men with clinically localized prostate cancer (stages T1a through T3a) who underwent radical prostatectomy between 1983 and 1996. The score uses the PSA, clinical stage, and Gleason grade to predict recurrence, which is defined as a postoperative serum PSA of 0.4 ng per mL or higher, clinical evidence of recurrence, or the need for subsequent treatment. The rule was initially validated in a group of 168 patients treated by the same group of surgeons. It also has been validated in a group of 1,033 patients in Europe4 and in a large community-based cohort of 1,701 patients in the United States.5 In the latter group, 24 percent experienced recurrence. The accuracy of the Kattan score was measured using a concordance index (a statistical measure of agreement from 0 to 1 in which 1 is perfect agreement and 0.5 is no better than chance). The concordance index for the Kattan nomogram was 0.68 in the large community validation study. The Kattan nomogram also was prospectively validated in a group of 331 black men, with a concordance index of 0.78.6
A score developed by D'Amico and colleagues included not only men who had prostatectomy but also those who underwent external beam radiation.7 However, the rule only predicted two-year recurrence and was less accurate than the Kattan rule used in the European validation study.4
The Kattan nomogram is shown in Figure 1. It is also available as a free handheld computer application at http://www.nomograms.org. The electronic version includes the estimated recurrence risk following radiotherapy.8 Although this nomogram provides useful guidance for patients and their physicians, it is important that they understand that the nomogram is not perfectly accurate and should not be the only tool used in decision making about cancer treatment and follow-up. Other factors such as the patient's age, how they feel about the risk of impotence and incontinence, and comorbidities also must be considered. Also, this nomogram is only applicable for a man who has already selected radical prostatectomy as a treatment for his prostate cancer, because that is the group in which it was validated.
Figure 1. Preoperative Nomogram for Prostate Cancer Recurrence
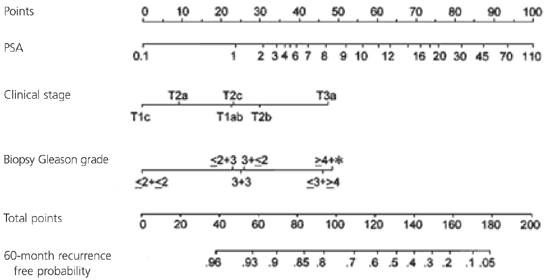
Preoperative nomogram for predicting prostate cancer recurrence after radical prostatectomy. (PSA = prostate-specific antigen.)
To use the nomogram, find the patient's PSA, clinical stage, and Gleason grade. Draw a vertical line up to the “Points” scale at the top of the nomogram for each variable, and sum the total points. Find the “Total points” scale at the bottom of the nomogram, and draw a vertical line downward to the estimate probability of remaining recurrence free during the next five years, given as a probability from 0 to 1.0.
Adapted with permission from Kattan MW, Eastham JA, Stapleton AM, Wheeler TM, Scardino PT. A preoperative nomogram for disease recurrence following radical prostatectomy for prostate cancer. J Natl Cancer Inst 1998;90:768.
Applying the Evidence
A patient with an abnormal digital rectal examination undergoes a prostate biopsy. His PSA score is 2.9, his Gleason grade is 3 + 3 = 6, and his tumor stage is 2a. What is the likelihood that he will remain free of cancer recurrence five years after surgery?
Answer: The physician consults the Kattan nomogram (Figure 1) and assigns 33 points for the PSA of 2.9, 9 points for the 2a tumor stage, and 25 for the Gleason grade of 6. The total score is 67 points, which predicts a 91 percent chance that he will remain recurrence free for five years.

