Most women can safely begin taking hormonal birth control products immediately after an office visit, at any point in the menstrual cycle. Because hormonal contraceptives do not accelerate cervical neoplasia or interfere with cervical cytology, women who have not had a recent Papanicolaou smear can begin using hormonal contraceptives before the test is performed. After childbirth, most women can begin using progestin-only contraceptives immediately. Estrogen-containing methods can safely be initiated six weeks to six months postpartum for women who are breastfeeding their infants and three weeks postpartum for women who are not breastfeeding. Women can begin any appropriate contraceptive method immediately following an early abortion. Delaying contraception may decrease adherence. Physicians can help patients improve their use of birth control by providing anticipatory guidance about the most common side effects, giving comprehensive information about available choices, and honoring women’s preferences. An evidence-based, flexible, patient-centered approach to initiating contraception may help to lower the high rate of unintended pregnancy in the United States.
While waiting to start a new birth control method, many women become pregnant unintentionally. Tradition determines that women delay starting hormonal contraceptives until the next menses, until a certain number of weeks have passed after childbirth, or until a breastfeeding infant is weaned. In addition, many physicians delay prescribing contraceptives for women who have not had a recent physical examination and Papanicolaou (Pap) smear.
Unintended pregnancy poses significant health risks to women and their families—it is associated with higher rates of domestic violence, maternal drug and alcohol use during pregnancy, delayed prenatal care, and low birth weight.1 Almost one half of pregnancies in the United States are unintended, and about 50 percent of those, or 1.3 million per year, lead to abortion.2 Although the incidence of unintended pregnancy in the United States has declined in recent years, it remains much higher here than in other developed countries, with a widening disparity between wealthy and indigent groups of American women.2 Limited access to primary health care services contributes to high rates of unintended pregnancy in women with low incomes. American teens, in particular, face multiple barriers in timely access to contraception.3
To address this problem, family physicians can make contraception safely and promptly available to their patients, with special attention to those at highest risk of unintended pregnancy. This article reviews the rationale for current practice and the evidence supporting a more timely approach.
SORT: KEY RECOMMENDATIONS FOR PRACTICE
| Clinical recommendation | Evidence rating | References | Comments |
|---|---|---|---|
| Hormonal birth control may be started at any point in the menstrual cycle. Taking the first pill immediately after requesting birth control enhances continuation rates. | C | 8 | Prospective study of 250 women who requested oral contraceptives |
| Hormonal birth control may be started without a recent Papanicolaou (Pap) smear test. Lack of a Pap smear before initiation or renewal of oral contraceptives does not increase the risk of cervical neoplasia. | C | 25 | Retrospective study with 400 participants in each group |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, see page 17 orhttps://www.aafp.org/afpsort.xml.
Office Visits Between Menses
When women request birth control at an office visit occurring between menses, many physicians delay starting hormonal contraceptives. Waiting until the next menses provides assurance that the woman is not already pregnant when she begins the new method. This practice probably began in order to avoid exposing a fetus to hormones, before studies had evaluated teratogenicity. Now there is a large body of evidence that refutes this risk; combined estrogen/progestin contraceptives do not cause birth defects.4 A more limited body of evidence indicates that hormonal contraceptives taken in early pregnancy cause no significant increase in the risks of miscarriage or fetal growth problems.5–7 Concern that hormones can mask the symptoms of early pregnancy, thus delaying diagnosis and leading to a later abortion or later onset of prenatal care, can be addressed through appropriate use of urine pregnancy tests, consideration of emergency contraception, and use of backup contraception during the first week of hormonal contraceptive use.8
The “quick start” method (Figures 19 and 29) allows most women with a negative urine pregnancy test to begin using the birth control pill, patch, or vaginal ring immediately after an office visit, at any point in the menstrual cycle.8 This strategy eliminates the delay between receiving a prescription and starting the new contraceptive method, and may improve adherence. With standard delayed contraceptive initiation, about 25 percent of women given a contraceptive prescription never fill it,10 and about 50 percent of women who start using birth control pills discontinue use within one year.11 In the quick start trial,8 women who took their first birth control pill during an office visit had significantly higher adherence three months later than women randomized to the delayed start group. Women who begin their new method after the first day of their last menstrual period should use a backup method during the first week.12
Figure 1. “Quick Start” Initiation of Hormonal Contraception: Pill, Patch, Ring, or Injection
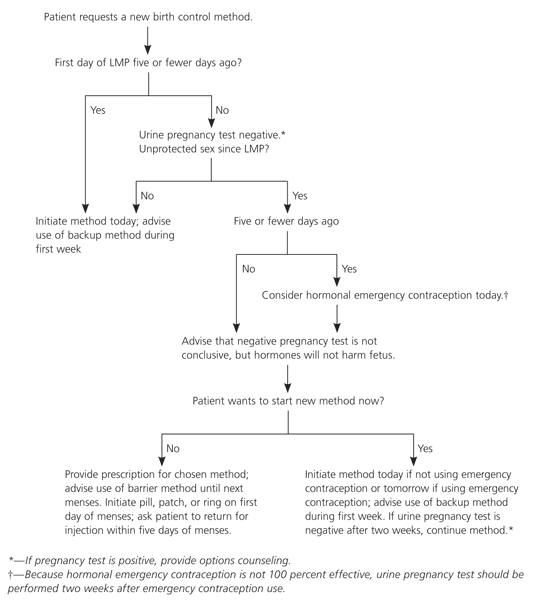
Algorithm for “quick start” initiation of hormonal contraception: pill, patch, ring, or injection. (LMP = last menstrual period.)
Adapted from Hatcher RA, Zieman M, Cwiak C, Darney PD, Creinin MD, Stosur HR. A Pocket Guide to Managing Contraception. Tiger, Ga.: Bridging the Gap Foundation, 2005:135.
Figure 2. “Quick Start” Initiation of Hormonal Contraception: Progestin IUD or implant
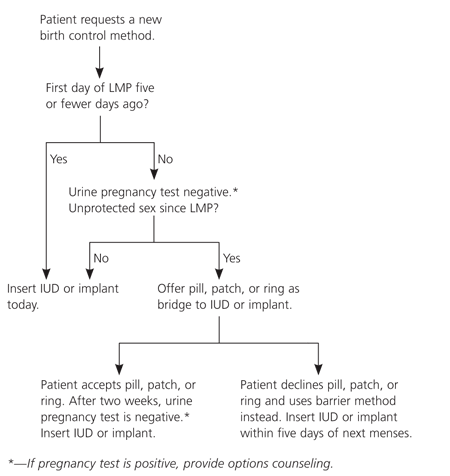
Algorithm for “quick start” initiation of hormonal contraception: progestin IUD or implant. (LMP = last menstrual period; IUD = intrauterine device.)
Adapted from Hatcher RA, Zieman M, Cwiak C, Darney PD, Creinin MD, Stosur HR. A Pocket Guide to Managing Contraception. Tiger, Ga.: Bridging the Gap Foundation, 2005:135.
Even women who have had recent unprotected intercourse can use the quick start method. Women who have had unprotected intercourse within five days of their visit can be offered hormonal emergency contraception that day, after appropriate counseling, and can begin their new contraceptive method the next day.8,12–14 The copper intrauterine device (IUD) can be used for emergency contraception as well as for long-term contraception, and is close to 100 percent effective when used within five days after unprotected intercourse15; however, the progestin IUD cannot be used for emergency contraception. Women who choose a hormonal method that is more difficult to discontinue in the event of pregnancy—such as hormonal injections, implants, or a progestin-releasing IUD—can use short-term hormones as a bridge until pregnancy is ruled out, or can wait until the next menses to begin the chosen method.
Some physicians avoid starting patients on hormones between menses even when there is no risk of undiagnosed pregnancy, because of concern about subsequent bleeding patterns. Most women experience spotting and other menstrual cycle changes during their first few months on hormonal contraceptives. A recent study16 indicates that women who initiate oral contraceptives between periods have no more disruption in menstrual patterns than those who wait until menses. Thorough counseling about side effects, including anticipatory guidance regarding spotting, may improve adherence to hormonal contraception.17
Postpartum
Because of a concern about hypercoagulability during the postpartum phase, many physicians withhold hormonal contraceptives from women after childbirth, whether or not the women are breastfeeding.18 The World Health Organization (WHO) reviewed available evidence on this issue while preparing its recent contraceptive guidelines (Table 1),19 and suggests that the risks of estrogen-containing contraceptives may outweigh the benefits during the first three weeks post-partum. After three weeks, however, when thrombosis risk returns to normal, postpartum women who are not breastfeeding can use estrogen-containing oral contraceptives without additional restrictions.19 Because low-dose progestins are not associated with thrombosis, WHO recommends initiating progestin-only contraceptives at any point postpartum.19 A progestin or copper IUD can be used immediately after childbirth. Both types of IUD have lower expulsion rates if inserted within the first 48 hours postpartum.19 For postpartum timing of IUD insertion, see Table 1.19
TABLE 1 WHO Medical Eligibility Criteria for Initiating Contraceptive Methods
| Categories | |||||||
|---|---|---|---|---|---|---|---|
| 1 Method can be used without restriction. | |||||||
| 2 Advantages of method generally outweigh risks. | |||||||
| 3 Method not usually recommended unless other, more appropriate methods are not available or not acceptable. | |||||||
| 4 Method not to be used. | |||||||
| Condition | Qualifier for condition | Combined methods* | Progestin-only | ||||
| Pill | Injection | Implant† | IUD | Copper IUD | |||
| Anemia | Thalassemia | 1 | 1 | 1 | 1 | 1 | 2 |
| Sickle cell disease | 2 | 1 | 1 | 1 | 1 | 2 | |
| Iron-deficiency anemia | 1 | 1 | 1 | 1 | 1 | 2 | |
| Breast disease | Family history of cancer | 1 | 1 | 1 | 1 | 1 | 1 |
| Current breast cancer | 4 | 4 | 4 | 4 | 4 | 1 | |
| Breast cancer in past, no evidence of disease for > 5 years | 3 | 3 | 3 | 3 | 3 | 1 | |
| Undiagnosed breast mass | 2 | 2 | 2 | 2 | 2 | 1 | |
| Benign breast disease | 1 | 1 | 1 | 1 | 1 | 1 | |
| Cervical cancer | Cervical intraepithelial neoplasia | 2 | 1 | 2 | 2 | 2 | 1 |
| Awaiting treatment | 2 | 1 | 2 | 2 | 4 | 4 | |
| Cervical ectropion | 1 | 1 | 1 | 1 | 1 | 1 | |
| Depression | 1 | 1 | 1 | 1 | 1 | 1 | |
| Diabetes mellitus | History of gestational disease | 1 | 1 | 1 | 1 | 1 | 1 |
| Without vascular disease | 2 | 2 | 2 | 2 | 2 | 1 | |
| With end-organ damage or > 20 years’ duration | 3 | 2 | 3 | 2 | 2 | 1 | |
| Drug interactions | Antiretrovirals | 2 | 2 | 2 | 2 | 2 | 2 |
| Certain anticonvulsants | 3 | 3 | 2 | 3 | 1 | 1 | |
| Griseofulvin (Grisactin) | 2 | 2 | 1 | 2 | 1 | 1 | |
| Rifampin (Rifadin) | 3 | 3 | 2 | 3 | 1 | 1 | |
| All other antibiotics | 1 | 1 | 1 | 1 | 1 | 1 | |
| Endometrial cancer | 1 | 1 | 1 | 1 | 4 | 4 | |
| Endometriosis | 1 | 1 | 1 | 1 | 1 | 2 | |
| Gallbladder disease | Asymptomatic gallstones | 2 | 2 | 2 | 2 | 2 | 1 |
| Symptomatic gallstones, without cholecystectomy | 3 | 2 | 2 | 2 | 2 | 1 | |
| Gallstones treated with cholecystectomy | 2 | 2 | 2 | 2 | 2 | 1 | |
| Pregnancy-related cholestasis in past | 2 | 1 | 1 | 1 | 1 | 1 | |
| Hormone-related cholestasis in past | 3 | 2 | 2 | 2 | 2 | 1 | |
| Headaches | Nonmigrainous | 1 | 1 | 1 | 1 | 1 | 1 |
| Migrainous | |||||||
| Without aura, age < 35 years | 2 | 1 | 2 | 2 | 2 | 1 | |
| Without aura, age ≥ 35 years | 3 | 1 | 2 | 2 | 2 | 1 | |
| With aura, any age | 4 | 2 | 2 | 2 | 2 | 1 | |
| HIV/AIDS | High risk | 1 | 1 | 1 | 1 | 2 | 2 |
| HIV infected | 1 | 1 | 1 | 1 | 2 | 2 | |
| AIDS (without drug interactions) | 1 | 1 | 1 | 1 | 3 | 3 | |
| Hypertension | During prior pregnancy, now resolved | 2 | 1 | 1 | 1 | 1 | 1 |
| Well controlled | 3 | 1 | 2 | 1 | 1 | 1 | |
| Systolic BP 140–159 mm Hg or diastolic BP 90–99 mm Hg | 3 | 1 | 2 | 1 | 1 | 1 | |
| Systolic BP ≥160 mm Hg or diastolic BP ≥ 100 mm Hg | 4 | 2 | 3 | 2 | 2 | 1 | |
| With vascular disease | 4 | 2 | 3 | 2 | 2 | 1 | |
| Ischemic heart disease | Past or current | 4 | 2 | 3 | 2 | 2 | 1 |
| Liver disease | Cirrhosis, mild | 3 | 2 | 2 | 2 | 2 | 1 |
| Cirrhosis, severe | 4 | 3 | 3 | 3 | 3 | 1 | |
| Tumors, benign | 4 | 3 | 3 | 3 | 3 | 1 | |
| Tumors, malignant | 4 | 3 | 3 | 3 | 3 | 1 | |
| Viral hepatitis, carrier | 1 | 1 | 1 | 1 | 1 | 1 | |
| Viral hepatitis, active | 4 | 3 | 3 | 3 | 3 | 1 | |
| Obesity | BMI ≥ 30 kg per m2 | 2 | 1 | 1 | 1 | 1 | 1 |
| Ovarian cancer | 1 | 1 | 1 | 1 | 3 | 3 | |
| Ovarian cysts and benign tumors | 1 | 1 | 1 | 1 | 1 | 1 | |
| Pelvic inflammatory disease | Past, with subsequent pregnancy | 1 | 1 | 1 | 1 | 1 | 1 |
| Past, without subsequent pregnancy | 1 | 1 | 1 | 1 | 2 | 2 | |
| Current | 1 | 1 | 1 | 1 | 4 | 4 | |
| Postabortion | First trimester | 1 | 1 | 1 | 1 | 1 | 1 |
| Second trimester | 1 | 1 | 1 | 1 | 2 | 2 | |
| Immediately after septic abortion | 1 | 1 | 1 | 1 | 4 | 4 | |
| Postpartum, not breastfeeding | < 2 days postpartum | 3 | 1 | 1 | 1 | 3 | 2 |
| 2 to 21 days | 3 | 1 | 1 | 1 | 3 | 3 | |
| 22 to 28 days | 1 | 1 | 1 | 1 | 3 | 3 | |
| > 28 days | 1 | 1 | 1 | 1 | 1 | 1 | |
| Postpartum, breastfeeding | < 2 days postpartum | 4 | 3 | 3 | 3 | 3 | 2 |
| 2 to 27 days | 4 | 3 | 3 | 3 | 3 | 3 | |
| 28 to 41 days | 4 | 3 | 3 | 3 | 1 | 1 | |
| 6 weeks to 6 months | 3 | 1 | 1 | 1 | 1 | 1 | |
| > 6 months | 2 | 1 | 1 | 1 | 1 | 1 | |
| Seizure disorder | Without drug interactions | 1 | 1 | 1 | 1 | 1 | 1 |
| Sexually transmitted disease | Vaginitis | 1 | 1 | 1 | 1 | 2 | 2 |
| High risk | 1 | 1 | 1 | 1 | 3 | 3 | |
| Current purulent cervicitis, chlamydial infection, or gonorrhea | 1 | 1 | 1 | 1 | 4 | 4 | |
| Smoking | Age < 35 years | 2 | 1 | 1 | 1 | 1 | 1 |
| Age ≥ 35 years, < 15 cigarettes per day | 3 | 1 | 1 | 1 | 1 | 1 | |
| Age ≥ 35 years, ≥ 15 cigarettes per day | 4 | 1 | 1 | 1 | 1 | 1 | |
| Stroke | 4 | 2 | 3 | 2 | 2 | 1 | |
| Surgery | Minor, without prolonged immobilization | 1 | 1 | 1 | 1 | 1 | 1 |
| Major, without prolonged immobilization | 2 | 1 | 1 | 1 | 1 | 1 | |
| Major, with prolonged immobilization | 4 | 2 | 2 | 2 | 2 | 1 | |
| Thyroid disorders | Simple goiter, hyperthyroidism, hypothyroidism | 1 | 1 | 1 | 1 | 1 | 1 |
| Uterine fibroids | Without distortion of uterine cavity | 1 | 1 | 1 | 1 | 1 | 1 |
| With distortion of uterine cavity | 1 | 1 | 1 | 1 | 4 | 4 | |
| Valvular heart disease | Uncomplicated | 2 | 1 | 1 | 1 | 1 | 1 |
| Complicated | 4 | 1 | 1 | 1 | 2 | 2 | |
| Varicose veins | 1 | 1 | 1 | 1 | 1 | 1 | |
| Venous thrombosis | Family history (first-degree relatives) | 2 | 1 | 1 | 1 | 1 | 1 |
| Superficial thrombophlebitis | 2 | 1 | 1 | 1 | 1 | 1 | |
| Past DVT/PE | 4 | 2 | 2 | 2 | 2 | 1 | |
| Current DVT/PE | 4 | 3 | 3 | 3 | 3 | 1 | |
*—Estrogen/progestin pill, patch, or ring.
†—Not available in the United States.
WHO = World Health Organization; IUD = intrauterine device; HIV = human immunodeficiency virus; AIDS = acquired immunodeficiency syndrome; BP = blood pressure; BMI = body mass index; DVT = deep venous thrombosis; PE = pulmonary embolism.
Adapted with permission from Medical Eligibility Criteria for Contraceptive Use. 3rd ed. Geneva: World Health Organization, 2004:13, 173–80. Accessed December 22, 2005, at:http://www.who.int/reproductive-health/publications/mec/mec.pdf.
Lactation
As in the postpartum phase, contraceptive choice during lactation depends on the length of time since childbirth and on the type of hormone selected. WHO advises against hormonal contraception use during the first six weeks postpartum in women who are breastfeeding because of concerns about the potential effects of steroids on liver and brain development in neonates. From six weeks to six months postpartum, the risk of diminished quantity and quality of breast milk may outweigh the benefits of estrogen-containing contraceptives.19 This risk may be more important to women who breastfeed exclusively. After six months postpartum, when infants begin to eat solid food, the benefits of estrogen-containing contraceptives may outweigh their risks.19
Progestin-only contraceptives have been studied more thoroughly in the postpartum setting. Even in the first six weeks postpartum, these contraceptives do not adversely affect milk production or infant growth.20 The Planned Parenthood Federation of America (PPFA) recommends progestin-only methods at any point postpartum.9 However, WHO suggests that the risks of progestin-only methods (i.e., neonatal steroid exposure) may outweigh the benefits during the first six weeks after childbirth. The PPFA and WHO are in agreement that women who are breastfeeding can safely use progestin-only contraceptives after six weeks postpartum.19
Lactation itself prevents pregnancy in the first six months postpartum in women who remain amenorrheic and whose babies get 90 percent or more of their calories from breast milk. However, a recent review of lactational amenorrhea as a contraceptive method found pregnancy rates ranging from 0 to 7.5 percent,21 pointing to the need to explore contraceptive options even with women who are breastfeeding exclusively.
Postabortion
Evidence supports the safety of beginning hormonal contraception immediately after medication and aspiration abortion, no matter what type of procedure was performed and whether or not there were complications.19 This strategy eliminates the need for women to use backup methods during the first week after starting the new method. Hormonal contraceptives do not adversely affect bleeding patterns after medication abortion.22 Copper and progestin IUDs can be safely inserted immediately after aspiration abortion, with only a slightly increased risk of expulsion.23 Implanted contraceptives can be started immediately after an aspiration abortion or at the routine follow-up visit after a medication abortion.
Clinical Evaluation Before Initiating Contraception
Many physicians require women to have a complete physical examination and Pap smear before starting hormonal contraceptives. To rule out contraindications to hormones, physicians should obtain a thorough medical history, including cardiovascular risk factors, concurrent medications, allergies, and health problems (past and current). For details on contraceptive selection for women with medical problems, see Table 1.19 Evaluation of height, weight, and blood pressure influences the appropriate contraceptive choice. However, the rest of the physical examination contributes little to this assessment.24 The Pap smear, important as it is in screening for cervical cancer, has minimal bearing on initiating contraception.25
Route of Administration
Oral contraceptives have been used for decades and studied extensively. Hormonal contraceptives also can be taken by injection, transdermally, vaginally, subdermally, and through an IUD. Several implantable progestins are approved by the U.S. Food and Drug Administration, but none is currently available in the United States. A single-rod system is anticipated in the near future. These new products’ individual characteristics may enhance or diminish their safety in various clinical situations (see Table 1).19
Estrogen-containing contraceptives have similar contraindications regardless of their route of administration. Although the estrogen/progestin patch and vaginal ring avoid the first-pass effect on the liver, studies on the potential benefits have not been performed. Thus, the cautions in relation to liver disease and liver-mediated drug interactions apply to these newer products as well as to the older oral versions.19
In general, progestin-only contraceptives carry fewer contraindications than estrogen-containing products, although the route of administration affects clinical use. Because depot-progestin injections produce measurable progestin blood levels for many months after discontinuation, injected progestin is less appropriate than progestin-only pills for women with unstable clinical conditions (e.g., uncontrolled hypertension). Progestin implants stop releasing hormone after removal, but removal can be difficult. On the other hand, IUDs can be removed easily.18
Physicians can help patients improve their use of birth control by providing anticipatory guidance about the most common side effects,26 giving comprehensive information about available choices,27 and honoring women’s preferences.28 Routinely asking about contraceptive needs demonstrates physicians’ willingness to explore this important topic with patients at any type of office visit—during routine well-baby visits, for example. Experience demonstrates that improved access to contraception leads to a decline in unintended pregnancy.29 An evidence-based, flexible, patient-centered approach to initiating contraception may help to lower the high rate of unintended pregnancy in the United States.

