Dystocia is common in nulliparous women and is responsible for more than 50 percent of primary cesarean deliveries. Because cesarean delivery rates continue to rise, physicians providing maternity care should be skilled in the diagnosis, management, and prevention of dystocia. If labor is not progressing, inadequate uterine contractions, fetal malposition, or cephalopelvic disproportion may be the cause. Before resorting to operative delivery for arrested labor, physicians should ensure that the patient has had adequate uterine contractions for four hours, using oxytocin infusion for augmentation as needed. For nulliparous women, high-dose oxytocin-infusion protocols for labor augmentation decrease the time to delivery compared with low-dose protocols without causing adverse outcomes. The second stage of labor can be permitted to continue for longer than traditional time limits if fetal monitoring is reassuring and there is progress in descent. Prevention of dystocia includes encouraging the use of trained labor support companions, deferring hospital admission until the active phase of labor when possible, avoiding elective labor induction before 41 weeks' gestation, and using epidural analgesia judiciously.
Caring for women with dystocia is a major challenge in maternity care. Dystocia refers to prolonged or slowly progressing labor. It is common in nulliparous women, as indicated by the number requiring augmentation, operative vaginal delivery, or cesarean section. In 2003, 17 percent of women in the United States received oxytocin augmentation,1 and in 2004, the primary cesarean delivery rate (i.e., cesarean delivery in women without previous cesarean) rose to 20.6 percent.2 Dystocia is responsible for more than 50 percent of primary cesarean deliveries.3 With the overall cesarean delivery rate at an all-time high of 30.2 percent4 (Figure 12,4), optimal management of dystocia can significantly impact labor outcomes.
SORT: KEY RECOMMENDATIONS FOR PRACTICE
| Clinical recommendation | Evidence rating | References | Comments |
|---|---|---|---|
| Amniotomy in the first stage of labor results in shorter labor, but it also may be associated with variable fetal heart rate decelerations; therefore, it should be reserved for slowly progressing labors. | A | 13 | Systematic review |
| High-dose oxytocin regimens result in shorter labors than low-dose regimens without adverse effects for the fetus. | A | 18, 19 | — |
| Women who receive continuous labor support from a labor support companion use less analgesia, have lower rates of operative vaginal and cesarean delivery, and are less likely to report dissatisfaction with their childbirth experiences. | A | 35 | Systematic review; results for each outcome were derived from atleast four trials including at least1,000 women |
| Epidural analgesia is associated with a prolongation of the second stage of labor and an increase in oxytocin use and operative vaginal delivery. | A | 46–49 | Systematic reviews and a meta-analysis |
| It is important to follow systematic protocols for diagnosing labor, assessing its progress, and using oxytocin. Audit and feedback regarding operative deliveries has been associated with lower institutional cesarean delivery rates. | C | 17, 57, 58 | — |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, see page 1605 or https://www.aafp.org/afpsort.xml.
Diagnosis
Normal progress in labor was initially defined by Friedman in the 1950s based on data from labors of several hundred women.5 Labor abnormalities are characterized as protraction or arrest disorders (Table 15,6). To aid in diagnosis, labor progression may be followed using a graph called a partogram, which plots cervical dilation and station across time.7,8
Table 1 Traditional Definitions of Abnormal Labor
| Stage of labor | Labor abnormality | |||
|---|---|---|---|---|
| Protracted | Arrested | |||
| Latent | ||||
| Nulliparous | > 20 hours | NA | ||
| Multiparous | > 14 hours | NA | ||
| First stage | ||||
| Nulliparous | < 1 cm per hour dilation | ≥ 2 hours of active labor without cervical change | ||
| Multiparous | < 1.2 to 1.5 cm per hour dilation | ≥ 2 hours of active labor without cervical change | ||
| Second stage | ||||
| Nulliparous or multiparous | With no regional anesthesia: | No descent after 1 hour of pushing | ||
| > 2 hours duration | ||||
| or | ||||
| < 1 cm per hour descent | ||||
| With regional anesthesia: | ||||
| > 3 hours duration | ||||
NA = not applicable.
The range of normal labor now appears to be broader than Friedman's definitions. A more recent study of labor progress among 1,329 nulliparous women delivering vaginally found it took an average of 5.5 hours to dilate from 4 to 10 cm (a mean rate of approximately 1.1 cm per hour).9 These findings contrast with Friedman's data, which had 1.2-cm dilation per hour defined as the 95th percentile (i.e., the outer limit of normal progress).5 The more recent analysis found that women who had not yet reached 7 cm dilation often had no cervical change for more than two hours. Fetal descent in the second stage of labor also appeared to take longer.9 Thus, the need for routine intervention for labor that is progressive yet protracted is questionable.10
Treatment
Physicians need to consider four issues when caring for women with dystocia: (1) if the contractions are adequate; (2) if there is fetal malposition; (3) if there is cephalopelvic disproportion caused by suspected macrosomia or a contracted pelvis; and (4) if there are other coexisting clinical issues (e.g., chorioamnionitis, nonreassuring fetal monitoring) that will impact the treatment options.
FIRST STAGE OF LABOR
Options for managing the latent phase of labor include observation, sedation with antihistamines or mild narcotics, and labor augmentation.11 Women being induced may remain in latent labor for many hours; cesarean delivery for dystocia should not be performed in women who remain in latent labor.6,12
Once a woman is in active labor, amni-otomy before oxytocin use may be sufficient to augment slowly progressing labor.13 Amniotomy with early oxytocin augmentation shortens labor by as much as two hours compared with expectant care but has not been shown to change cesarean delivery rates.13 Although amniotomy is a simple procedure, it still carries a risk of causing increased variable heart rate decelerations because of cord compression.13 Routine amniotomy in early labor is not recommended,14 and the role of amniotomy to treat protracted labor is under review.15
Abdominal palpation or an intrauterine pressure catheter, which calculates Monte-video units (MVU), can be used to evaluate the strength and frequency of uterine contractions in women with protracted- or arrested-phase labor (Figure 2). MVU of 200 or more in 10 minutes are considered evidence of adequate contractions.6 Using an intrauterine pressure catheter may be important if the contractions seem to be of sufficient frequency and duration but are not causing cervical change. A small randomized trial found no difference in labor duration or cesarean delivery rates when an intrauterine pressure catheter was used.16
Figure 2.
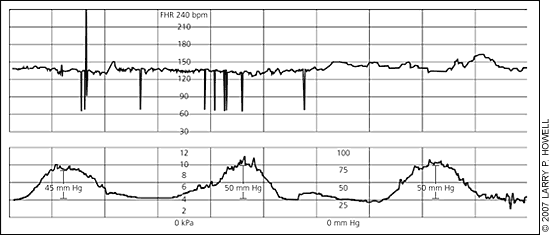
Inadequate uterine contractions as measured by an intrauterine pressure catheter, with continuous tracing of the FHR (top) and contractions as indicated by uterine pressure (bottom). This representative 10-minute monitor strip shows three contractions totaling 145 MVU. If MVU are less than 200 in 10 minutes, oxytocin augmentation should be considered. (FHR = fetal heart rate; bpm = beats per minute; kPa = kilopascal; MVU = Montevideo units.)
If contractions are inadequate, intravenous oxytocin can be administered to increase frequency, duration, and strength.6 There are numerous approaches to dosage, dosing interval, and duration of oxytocin treatment. Low-dose regimens start at 0.5 to 2.0 mU per minute and increase by 1 to 2 mU per minute every 15 to 40 minutes up to a maximal dose of 20 to 40 mU per minute.6 High-dose regimens have a starting dose of 6 mU per minute and increase by 1 to 6 mU per minute up to a maximal dose of 40 to 42 mU per minute.6,17 In nulliparous women who need augmentation, high-dose oxytocin regimens decrease the time to delivery by an average of two hours compared with low-dose regimens without causing adverse effects for the fetus.18,19
Traditionally, arrested labor has been defined as having adequate contractions for at least two hours without cervical change; a woman should be observed for at least that long before resorting to operative intervention. Extending the time to four hours before operative treatment has been shown to decrease the cesarean delivery rate for arrested labor from 26 to 8 percent.20,21
SECOND STAGE OF LABOR
Dystocia in the second stage of labor is characterized by prolonged duration or arrested descent. This may be caused by fetal malposition, inadequate contractions, poor maternal efforts, or true cephalopelvic disproportion.
The most common fetal malposition is occipitoposterior (i.e., the fetus lying with the occiput toward the mother's spine and face toward the mother's pubic symphysis). Typically, the fetus will rotate spontaneously to the occipitoanterior position before delivery, but in 2 to 7 percent of nulliparous women, the fetus will still deliver in the persistent occipitoposterior position.22,23 This position is associated with prolonged second stage of labor and increased oxytocin augmentation.22,23 Less than 30 percent of nulliparous women with a fetus in the persistent occipitoposterior position will have a spontaneous vaginal delivery.22,23
Occipitoposterior position is diagnosed by digital vaginal examination, which can determine the orientation of fetal sutures and fontanels. If the physician cannot make this determination, transvaginal sonography can confirm fetal head position.24 If a fetus is in the persistent occipitoposterior position in the second stage of labor, manual rotation can be attempted. Although there is a lack of high-level evidence regarding the effectiveness of interventions for a fetus in this position, a retrospective cohort study of 742 women who underwent attempted manual rotation of a fetus in the occipitoposterior or occipitotransverse position to the occipi-toanterior position demonstrated a lower cesarean delivery rate with successful rotation compared with failed rotation (2 versus 34.3 percent, P <.001).25
Manual rotation is a clinical skill that requires training and practice. The physician's hand is placed palm upward into the vagina. During a contraction, the hand serves as a wedge to flex the fetal head while the fingers exert a rotating force to bring the occiput to the anterior (Figure 3).26
Figure 3.
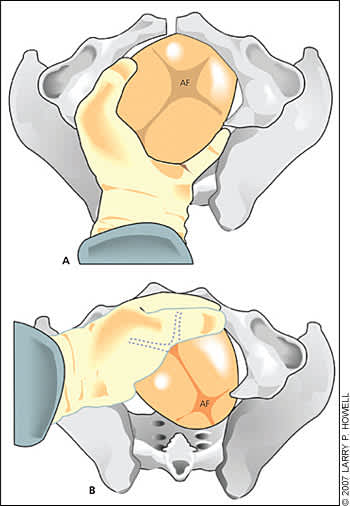
Manual rotation of a fetus in the occipitoposterior position to the occipitoanterior position. (A) The physician's hand is placed palm upward into the vagina. (B) The hand serves as a wedge to flex the fetal head while the fingers exert a rotating force to bring the occiput to anterior. (AF = anterior fontanel.)
A variety of maternal positions and movements have been proposed to resolve persistent occipitoposterior or asynclitic fetal positions. These include knee-chest, hands-and-knees, pelvic rocking, lunging, side-lying, or asymmetrical sitting or kneeling.27,28 A systematic review concluded that having a woman assume the hands-and-knees position for a specified period near the end of pregnancy had no effect on fetal position at delivery; however, no studies were conducted using women in labor.29
If contractions have decreased in strength or frequency during the second stage of labor, intravenous oxytocin can be initiated or increased.17 Studies have shown that having women without epidural analgesia push in an upright or lateral position shortened the second stage of labor and decreased the risk of operative vaginal delivery, but this position increased the risk of second-degree perineal tears and blood loss of more than 500 mL.30 For women with epidural analgesia, allowing the fetus to “labor down” to a lower station is an alternative to initiating active pushing as soon as cervical dilation is complete. In one study, delayed pushing increased the incidence of spontaneous deliveries (relative risk [RR], 1.09; 95% confidence interval [CI], 1.00 to 1.18; number needed to treat [NNT] = 21).31
Prolongation of the second stage of labor beyond an arbitrary time limit is no longer an indication for operative vaginal or cesar-ean delivery. Several studies have demonstrated the safety to the neonate of extended second stage labor based on cord blood gases and five-minute Apgar scores.32–34 A nonreassuring fetal heart tracing indicates a need for consideration of operative vaginal or cesarean delivery.
Prevention
The incidence of dysfunctional labor in nulliparous women may be decreased by four methods: (1) provision of labor support; (2) avoidance of hospital admission in latent stage of labor; (3) avoidance of elective induction with an unripe cervix; and (4) cautious use of epidural analgesia.
A meta-analysis on the use of a trained labor support companion (i.e., a doula) showed that labor support decreases the incidence of dystocia, operative vaginal deliveries, and cesarean deliveries, particularly in nulliparous women.35 The greatest effects on labor outcomes occur when a doula rather than a hospital employee is used, when support begins early in labor, and when epidural analgesia is not routinely used.35 Having a trained nurse rather than a doula provide continuous labor support does not provide similar benefits.36
Nulliparous women presenting to the hospital in the latent stage of labor undergo an increased number of obstetric interventions. It remains unclear if this is because of inherent labor abnormality or excessive intervention.37,38 One study showed that avoiding early hospital admission for women not in active labor reduced the risk of receiving augmentation of labor or epidural analgesia by more than one half.39 Physicians can educate nulliparous women about when to go to the hospital. As alternatives to admission in latent labor, physicians can encourage adequate hydration, rest, and emotional and physical support.11
The number of births involving induction of labor has more than doubled in the past decade, from 9 percent in 1989 to nearly 21 percent in 2003.1 Elective induction may be partially responsible for the increasing rate of cesarean delivery in women with dystocia. Retrospective or cohort data show that elective induction results in a two- to threefold increased risk of cesarean delivery in nulliparous women with an unripe cervix despite the use of cervical ripening agents.40,41 Cochrane reviews of misoprostol (Cytotec) and mechanical methods for cervical ripening found that they decrease the length of labor but do not change the overall cesarean delivery rate.42,43 In contrast, a retrospective study demonstrated a decreased cesarean delivery rate through selective induction of women at full term with specific risk factors for developing cephalopelvic disproportion or uteroplacental insufficiency.44 Standard labor curves may not apply to women undergoing induction of labor who may have an active phase longer than expected for spontaneous labor,45 suggesting a need to permit adequate time to pass before intervening for dystocia.
Although meta-analyses consistently find no difference in cesarean delivery rates among women receiving low-dose epidurals compared with parenteral opioids,46–49 informed and judicious use of epidural analgesia is important because of the impact on labor progress and other outcomes. Women receiving epidurals are more likely to require oxytocin augmentation in the first stage of labor, have longer second stages, have persistent occipitoposterior fetal malposition, and undergo operative vaginal delivery.46–50
Whether administering epidural analgesia early in labor (before 4 to 5 cm dilation) increases the risk of cesarean delivery is controversial.51 Epidural analgesia is not a single entity, and randomized controlled trials that have specifically investigated early versus standard (4 to 5 cm cervical dilation) placement are small or do not use contemporary low-dose techniques.51 The study that is most commonly cited to support early epidural use actually compared a combined spinal epidural analgesia technique (i.e., intrathecal opioid given at 2 cm cervical dilation) with an epidural given at 4 cm or later. This study found no significant differences in labor duration or cesarean delivery rates.52
Maternal request is a sufficient indication for pain relief during labor,53,54 and epidurals are associated with significantly lower pain scores compared with systemic opioids.46–49 If and when to administer epidural analgesia should be individualized. Women with significant pain early in labor should not be required to reach 4 to 5 cm cervical dilation before epidural placement.54 Conversely, a woman who is informed and prepared to handle labor pain with lesser interventions should not be subjected to the expectation of a routine epidural.
Women who walk or remain upright during the first stage of labor report greater comfort and ability to tolerate labor compared with women who remain recumbent.55 A randomized trial comparing women assigned to walk during early labor with those receiving usual care showed no differences in the duration of the first stage of labor, need for oxytocin augmentation, use of analgesia, or rates of operative vaginal or cesarean delivery. Ambulation did not decrease dystocia in this study, but it can be suggested safely because there were no harmful effects for mothers or infants.56
Finally, certain aspects of physician style and health care systems may prevent dystocia and resultant cesarean delivery. These include caregiver continuity during the assessment of early labor,17 encouraging a “pronatalist” cultural attitude toward natural childbirth,57 requiring consultation with a second physician before nonemergent cesarean deliveries for dystocia,57 and providing regular feedback to physicians about their cesarean delivery rates.58

