Methamphetamine is a stimulant commonly abused in many parts of the United States. Most methamphetamine users are white men 18 to 25 years of age, but the highest usage rates have been found in native Hawaiians, persons of more than one race, Native Americans, and men who have sex with men. Methamphetamine use produces a rapid, pleasurable rush followed by euphoria, heightened attention, and increased energy. Possible adverse effects include myocardial infarction, stroke, seizures, rhabdomyolysis, cardiomyopathy, psychosis, and death. Chronic methamphetamine use is associated with neurologic and psychiatric symptoms and changes in physical appearance. High-risk sexual activity and transmission of human immunodeficiency virus are also associated with methamphetamine use. Use of methamphetamine in women who are pregnant can cause placental abruption, intrauterine growth retardation, and preterm birth, and there can be adverse consequences in children exposed to the drug. Treatment of methamphetamine intoxication is primarily supportive. Treatment of methamphetamine abuse is behavioral; cognitive behavior therapy, contingency management, and the Matrix Model may be effective. Pharmacologic treatments are under investigation.
The synthetic stimulant methamphetamine (most commonly known as “meth” or “crystal meth”) is a commonly abused drug in the United States. Its effects are similar to those of cocaine, and it is highly addictive, inexpensive, and easily available. It can be illicitly manufactured from over-the-counter pseudoephedrine.
SORT: KEY RECOMMENDATIONS FOR PRACTICE
| Clinical recommendation | Evidence rating | References |
|---|---|---|
| In patients who use methamphetamine, contingency management should be considered to reduce methamphetamine use and high-risk sexual behavior. | C | 43 |
| Referral to Matrix Model therapy should be considered for patients who abuse methamphetamine. | C | 44 |
| Fluoxetine (Prozac) at a dosage of 40 mg per day should be considered in patients who are addicted to methamphetamine to reduce short-term craving. | C | 41 |
| Pregnant women who use methamphetamine should be given easy access to an established drug rehabilitation program. | C | 48 |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, see page 1095 or https://www.aafp.org/afpsort.xml.
Epidemiology
Although rates of methamphetamine use have stabilized since 2002, rates of methamphetamine dependence increased from 10.6 percent of users in 2002 to 22.3 percent in 2004.1 The recently published National Longitudinal Study of Adolescent Health found that 2.8 percent of adults 18 to 26 years of age reported methamphetamine use within the past year.2
Most methamphetamine users are white men 18 to 25 years of age, although proportionally more women use methamphetamine than other illicit drugs,3 and usage rates are higher in certain ethnic groups (Figure 1).1 Methamphetamine use is more common among men who have sex with men, and it is associated with human immunodeficiency virus (HIV) infection and transmission in this population.4,5 One study in six large cities showed that 13 percent of men who have sex with men had used methamphetamine in the preceding six months.4 Methamphetamine use has also been associated with hepatitis C virus infection.6
Figure 1. Methamphetamine use in Preceding Year among Persons 12 Years and Older, by Race/Ethnicity: 2002 to 2004
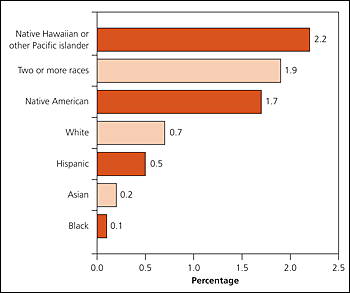
Methamphetamine use in the preceding year among persons 12 years and older, by race/ethnicity: 2002 to 2004.
Adapted from Methamphetamine use, abuse, and dependence: 2002, 2003, and 2004. NSDUH Report September 16, 2005. Accessed May 15, 2007, at: http://www.drugabusestatistics.samhsa.gov/2k5/meth/meth.htm.
There is considerable geographic variation in methamphetamine use, with the highest usage rates in Western and north central states (Figure 2).1 Methamphetamine use is a significant problem in both urban and rural areas and is associated with criminal activity.7,8
Figure 2. Methamphetamine use in Preceding Year among Persons 12 Years and Older, by State: 2002 to 2004
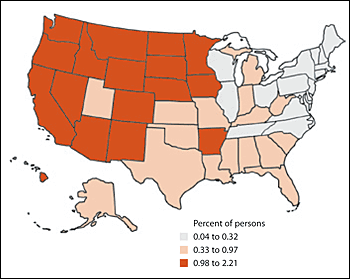
Methamphetamine use in the preceding year among persons 12 years and older, by state: 2002 to 2004.
Adapted from Methamphetamine use, abuse, and dependence: 2002, 2003, and 2004. NSDUH Report September 16, 2005. Accessed May 15, 2007, at: http://www.drugabusestatistics.samhsa.gov/2k5/meth/meth.htm.
Clinical Effects
Methamphetamine is most commonly smoked, but it can also be snorted, injected, swallowed, or inserted rectally.9
ACUTE
Methamphetamine use produces a rapid, pleasurable rush caused by release of dopamine, norepinephrine, and serotonin; it also produces euphoria, a heightened level of alertness, and increased energy.10 Increased libido and enhanced sexual pleasure also occur, and methamphetamine use is associated with high-risk sexual behavior.11,12 Prolonged sleep and mild dysphoria occur as the drug effects wear off.
Possible adverse effects are listed in Table 113–24; the incidence of these effects is unclear. One review of 145 deaths in which methamphetamine use was a contributing factor found that accidents, homicide, and suicide were responsible for the 123 deaths in which a cause could be determined.15
Table 1 Adverse Effects of Methamphetamine Use
| Acute exposure | |
| Anorexia | |
| Anxiety | |
| Death | |
| Hypertension | |
| Hyperthermia | |
| Insomnia | |
| Myocardial infarction | |
| Paranoia | |
| Psychosis | |
| Renal failure | |
| Rhabdomyolysis | |
| Seizures | |
| Sexually and parenterally transmitted infections | |
| Stroke | |
| Tachycardia | |
| Tachypnea | |
| Trauma | |
| Chronic exposure | |
| Cardiovascular complications | |
| Cardiomyopathy | |
| Myocardial infarction | |
| Stroke | |
| Dermatologic symptoms and signs | |
| Abscesses | |
| Aged appearance | |
| Skin lesions | |
| Neurologic symptoms | |
| Confusion | |
| Memory loss | |
| Motor slowing | |
| Verbal learning impairment | |
| Oral signs | |
| Dental decay (“meth mouth”) | |
| Psychiatric symptoms | |
| Anxiety | |
| Depression | |
| Paranoia | |
| Psychosis | |
| Suicidal ideation | |
Methamphetamine has a half-life of 12 hours, so its effects last longer than those of cocaine.16 It is metabolized by the liver through the cytochrome P2D6 system. Use of medications metabolized through this pathway, such as protease inhibitors and monoamine oxidase inhibitors, can raise methamphetamine levels.
CHRONIC
Chronic use of methamphetamine can cause potentially irreversible neuronal changes.17,18 These changes can result in neurologic and psychiatric symptoms, which are probably caused by dopamine depletion19,20 and are listed in Table 1.13–24 Myocardial infarction, respiratory failure, stroke, cardiomyopathy, and hepatitis have been reported.16 Tolerance and addiction can occur, and methamphetamine users often use other substances.
Distinctive changes occur in the physical appearance of long-term methamphetamine users, producing an aging effect. These changes usually result from malnutrition, severe dental decay (known as “meth mouth”), poor hygiene, and weight loss. Long-term users of methamphetamine often exhibit skin-picking behaviors, which can lead to abscesses.21 Such physical changes can alert health care professionals that their patients may be abusing methamphetamine. Examples of these changes are available at http://www.pbs.org/wgbh/pages/frontline/meth/body/faces.html.
EFFECTS IN PREGNANCY
The incidence of methamphetamine use in pregnant women is unknown, but a 2002 study found that 3 percent of pregnant women in the United States had used illicit substances in the preceding month.22 One study found that pregnant women who used methamphetamine were more likely than other pregnant women to be younger, live without a partner, have a lower income, have less education, and have received less prenatal care.23
Although methamphetamine crosses the placenta, data regarding in utero effects are limited.24 Placental insufficiency and abruption can occur, and maternal deaths have been reported.25,26 Fetal effects reported include intrauterine growth retardation, prematurity, clefting, cardiac anomalies, and death.24,26
EFFECTS ON CHILDREN
There are few data regarding methamphetamine exposure in children. Withdrawal from stimulant exposure is usually milder than opiate withdrawal in neonates, but abnormal sleep patterns, poor feeding, tremors, and hypertonia have been reported.27 One study found that 49 percent of neonates exposed to methamphetamine exhibited such withdrawal signs, although only 4 percent required medication.28 Phenobarbital and tincture of opium are sometimes used to treat stimulant withdrawal, but little supporting evidence exists.
Long-term effects of prenatal methamphetamine exposure are unclear. A 14-year follow-up study of children born to women who abused amphetamines in pregnancy showed academic and mild physical delays, but there were many potential confounders.29 Because methamphetamine is secreted in breast milk, breastfeeding is not recommended for mothers who use the drug after delivery.
Children are present in 20 percent of homes with methamphetamine laboratories, and environmental exposure may occur.30 Exposure can cause headaches, nausea, dizziness, dyspnea, chest pain, eye irritation, and burns. Exposed children are also at risk for inadvertent poisoning, trauma, neglect, abuse, and adverse psychological effects.31–33
Other Hazards
The U.S. Drug Enforcement Administration estimates that each 1 lb (0.45 kg) of methamphetamine produced in home laboratories generates 6 lb (2.7 kg) of toxic waste, resulting in complex and costly cleanup.34 Laboratory explosions may cause burns to those nearby, including producers, users, neighbors, and emergency personnel.35 Heavy metal contamination of methamphetamine products is also possible.
Screening
No specific guidelines regarding screening for methamphetamine use are available. The U.S. Preventive Services Task Force found insufficient evidence to recommend for or against routine screening for drug use by history or diagnostic testing.36
Screening for methamphetamine use by history should be considered for pregnant women, teenagers and young adults, persons with criminal histories, men who have sex with men, and persons in high-risk ethnic groups. A modified CAGE questionnaire or similar instrument can be used for screening by history, although the effectiveness of such tools in detecting methamphetamine abuse is unknown.
Diagnostic testing with informed consent can be useful in patients with stimulant-associated symptoms and signs. Methamphetamine is detectable in urine for approximately 48 hours after use.37 It is also detectable by hair analysis. Meconium testing is the most accurate method in newborns; a positive result indicates maternal use in the second half of pregnancy.38 Pseudoephedrine can cause a positive test result for amphetamines, but a confirmatory test can be performed to differentiate the use of this drug from methamphetamine.
Treatment of Acute Intoxication
The treatment of acute methamphetamine intoxication is largely supportive. Activated charcoal can be given if methamphetamine was ingested orally. Benzodiazepines may be indicated for seizures or agitation, and antipsychotics may be necessary in patients with paranoia or frank psychosis. Cooling measures may be required. Markedly elevated blood pressure should be lowered, although there are no data regarding blood pressure goals or which medications to use. The benefit of urinary acidification is unknown.
Abuse of multiple substances should be considered in patients who use methamphetamine. The Drug Abuse Warning Network Report found that more than 60 percent of methamphetamine-related emergency department visits in 2002 also involved other illicit drugs.39 Marijuana, alcohol, and cocaine were the most common substances found.
Withdrawal
Stimulant withdrawal is less dangerous than withdrawal from alcohol, opioids, or sedatives, but seizures are possible.40 Stimulant withdrawal symptoms include depression, somnolence, anxiety, irritability, inability to concentrate, psychomotor slowing, increased appetite, and paranoia.41 There are no known effective treatments.42 Methamphetamine withdrawal is associated with more severe and prolonged depression than is cocaine withdrawal, so patients with withdrawal should be monitored closely for suicidal ideation. Withdrawal occurs in 87 percent of long-term users who cease drug use.
Treatment of Abuse
Outpatient behavioral therapies are the standard treatment for methamphetamine abuse and dependence, although inpatient treatment is sometimes used.1 Cognitive behavior therapy and contingency management programs have been successfully used in treating cocaine addiction and may have some benefit in treating methamphetamine addiction.42
Contingency management rewards patients who are addicted to methamphetamine when they provide drug-free urine samples. A randomized controlled trial found that contingency management reduced methamphetamine use and high-risk sexual behavior.43 The Matrix Model is an individualized outpatient regimen that has been used successfully to treat patients who abuse stimulants.44 It is based on cognitive principles, incorporating individual, group, and family therapies, as well as drug testing and a 12-step program.44 Comprehensive case management has been used as an adjunct to behavioral treatments.45 These specialized services are offered by substance abuse treatment centers. Patients who abuse methamphetamine also may benefit from support groups or 12-step drug treatment programs.
MEDICATIONS
There are no medications approved by the U.S. Food and Drug Administration to treat methamphetamine dependence. A Cochrane review concluded that fluoxetine (Prozac) at a dosage of 40 mg per day may have modest benefit in reducing short-term methamphetamine craving but does not reduce methamphetamine use, and that imipramine (Tofranil) may improve adherence to therapy in methamphetamine users.41 One small randomized controlled trial found that bupropion (Wellbutrin) decreased subjective methamphetamine-induced effects and craving in a laboratory setting.46
PREGNANCY AND CHILDREN
Pregnant women and women with young children may require intensive and highly structured treatment plans for methamphetamine abuse, commonly in a residential setting.47 They often do not seek treatment or withdraw from treatment for fear of punishment and/or losing custody of their children. These women also may be stigmatized.47
The American Academy of Family Physicians (AAFP) opposes the imprisonment of pregnant women solely for substance abuse during pregnancy, as well as legislation that would interfere with a pregnant woman seeking prenatal care. However, the AAFP encourages easy access to an established drug rehabilitation program.48
Prevention
There is little evidence regarding the prevention of methamphetamine abuse. Two randomized controlled trials of family-based educational programs at public schools showed a decrease in adolescent methamphetamine use rates over more than five years.49 Further development of community and school-based prevention programs and prevention efforts directed at teenagers, women of child-bearing age, men who have sex with men, and high-risk ethnic groups are necessary. Limiting the availability of pseudoephedrine has resulted in short-term reductions of methamphetamine-related arrests and hospitalizations, but these reductions have not been sustained.50
Resources
Patients and physicians can find further information about treatment from the Center for Substance Abuse Treatment (800-662-HELP or http://findtreatment.samhsa.gov). Patients and their families can find information about methamphetamine from the National Clearinghouse for Alcohol and Drug Information (http://www.samhsa.gov) and from the National Institute on Drug Abuse (http://www.drugabuse.gov/drugpages/methamphetamine.html).

