Family physicians commonly encounter patients with dental infections, such as dental caries and periodontal disease. Dental caries is caused by bacteria that destroy the enamel and dentin; it can be detected by an oral examination that shows stained pits or fissures on the tooth surface. Use of fluoride is the most effective prevention measure for dental caries. Untreated caries may progress to pulpitis and, eventually, to necrosis of the pulp. In irreversible pulpitis, the tooth dies and the patient may have a localized abscess that can spread to surrounding tissue. Periodontal infections are caused by bacteria in the subgingival dental plaque. In gingivitis, the inflamed gums bleed easily with brushing or flossing; the condition can be controlled with good oral hygiene. Periodontitis is characterized by a loss of supportive bone structure caused by chronic gingivitis; it is also associated with some systemic diseases. Localized periodontitis is treated with mechanical debridement and good oral hygiene, whereas generalized periodontitis requires adjunct antibiotic therapy. Pericoronitis results when food particles become trapped under the gum of an impacted tooth. This condition can be controlled by removal of food debris and good oral hygiene. For patients in whom dental infections are disseminated and have invaded the deeper oral spaces, antibiotic treatment should be initiated at the time of referral.
Dental infections, including dental caries and periodontal disease, are commonly encountered in the primary care setting in the United States. Nationally, dental caries is the most common disease in children and adults. Fifty percent of children between six and eight years of age have dental caries,1 and 85 percent of adults have at least one tooth with decay or a filling on the crown.2 According to data from the Third National Health and Nutrition Examination Survey, in the United States 48 percent of adults between 35 and 44 years of age have experienced gingivitis, and 15 percent of adults older than 30 years have destructive periodontal disease (i.e., periodontitis).3 In 2005, dental services in the United States cost $86.6 billion, 4.4 percent of the total spent on health care that year.4
Clearly, the direct costs (i.e., the money spent on dental services) and indirect costs (i.e., the number of lost days of productivity at work and school) of oral diseases place a high burden on society. Family physicians are in a good position to address this morbidity. They can recognize common dental infections and, in some circumstances, may initiate treatment of serious infections before referral. Family physicians can also advise patients on how to prevent dental infections.
SORT: KEY RECOMMENDATIONS FOR PRACTICE
| Clinical recommendation | Evidence rating | References | Comments |
|---|---|---|---|
| Most dentists recommend a six-month interval for dental checkups for all patients. | C | 7 | There is insufficient evidence to support or refute biannual dental visits |
| Tooth brushing with a small amount of low-fluoride toothpaste should be introduced to children at two years of age. After six years of age, children can safely use regular fluoridated toothpaste. | B | 8, 11–13 | — |
| Tooth brushing with fluoridated toothpaste twice a day after meals is recommended as an effective way to prevent tooth decay. | A | 8, 15 | The benefits of topical fluorides have been firmly established |
| Use of powered toothbrushes that have a circular oscillating motion can reduce plaque and gingivitis more effectively than manual brushing. | B | 16 | Long-term benefits of using powered toothbrushes for dental health are unclear |
| Decreasing the amount and frequency of consumption of foods with a high sugar content can improve oral health. | B | 5, 8 | — |
A = consistent, good quality patient-oriented evidence; B = inconsistent or limited quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, see page 739 or https://www.aafp.org/afpsort.xml.
Basic Dental Anatomy
Human beings have two sets of teeth: primary (milk or baby) and permanent. There are 20 primary teeth, including two incisors, one canine, and two molars in each half jaw. These teeth erupt at approximately six months, and the set is complete by two years of age. The primary teeth are shed between six and 12 years of age. These are eventually replaced by permanent teeth starting at approximately age six and usually ending by age 18. The permanent teeth include two incisors, one canine, two premolars, and three molars in each half jaw.
Structurally, a tooth has a visible crown that projects above the gingiva. Enamel is the outermost layer of the crown and is the hardest structure of the body. In the middle is the dentin, a bone-like material, and the innermost layer is the pulp chamber (Figure 1). Similarly, the roots of the tooth have cementum as the outermost layer, the dentin is in the middle, and the pulp canal is in the center. The blood vessels and nerves enter the pulp chamber through the apical foramen and the pulp canal. The tooth is held in its socket by periodontal ligaments (periodontium) connecting the tooth to the alveolar bone structure.
Figure 1.
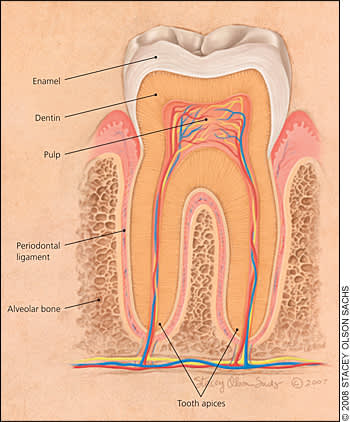
Anatomy of a tooth.
Dental Caries
Dental caries is caused mainly by micro-organisms that generate acids on the tooth surface from disaccharides and monosaccharides present in sugar-rich foods; this leads to demineralization of the protective enamel coating and subsequent tooth decay. Certain foods such as sweetened pastries may be more potent than others at inducing dental caries.5 Even diet sodas, which contain no sugars, have been implicated in decalcification caused by low pH from phosphoric acid. It is the frequency of exposure rather than the amount of carbohydrate substrate that makes the difference in caries rates.
Initially, dental caries is asymptomatic. As the infection progresses, depending on the severity of the caries, patients may experience sensitivity to thermal changes, mild pain, or severe pain (when dental caries encroaches on the pulp). Dental caries initially presents as a pit or fissure in the tooth surface that can be detected by a dental probe. These pits or fissures will eventually become stained because of demineralization of the enamel and dentin (Figure 2). Recession of the gums in aging patients may expose the roots to dental caries. Dental caries is also diagnosed by radiographic examination; low-level lasers can detect early tooth decay.
Figure 2.
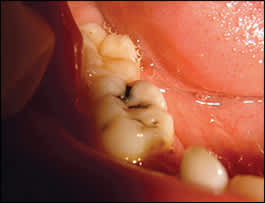
Dental caries of a right lower molar.
DENTAL CARIES PREVENTION
Examinations by a dentist should begin when patients are one year of age.6 Although most dentists have recommended a six-month interval for dental checkups, there is insufficient evidence to support or refute the biannual versus annual visits.7 The most cost-effective intervention for reducing dental caries is the public health policy of adding fluoride at a concentration of 0.7 to 1.0 parts per million (ppm) to the municipal water supply.8 Fluoride forms a complex with the apatite crystals in the enamel and dentin; where there are lesions, fluoride enhances remineralization at the site of the caries, thereby strengthening the tooth structure. Fluoride also has a bacteriostatic effect. There is a recent trend toward using bottled water instead of water from the municipal supply. Most bottled waters contain less than 0.3 ppm of fluoride; therefore, persons who rely on bottled water alone may need to add oral or topical fluoride to their oral hygiene regimen.9
Before prescribing fluoride tablets for young children in areas where water is fluoride deficient, physicians should consider other sources of fluoride use by the patient and weigh the risk of enamel fluorosis versus benefits of dental caries prevention.8,10 The benefits of topical fluorides have been firmly established, regardless of whether the water supply is fluoridated.11 When compared with mouth rinses or gels, fluoridated toothpastes have a similar degree of effectiveness for the prevention of dental caries in children.12 Parents should introduce tooth brushing with a pea-size amount of low-fluoride toothpaste to children at two years of age. In children younger than two years, parental brushing without toothpaste is recommended. After the age of six, children can safely use regular fluoridated toothpaste.8 Topical fluorides (i.e., mouth rinses, gels, or varnishes) used in addition to toothpaste for dental caries allow high-risk patients to achieve a modest additional reduction in caries compared with toothpaste alone.13 The use of mouth rinses and gels at home is not recommended for children younger than six years.14
Removing dental plaque helps the patient maintain good oral health. Tooth brushing with fluoridated tooth-paste twice a day after meals is recommended as an effective way to prevent tooth decay.8,15 Powered toothbrushes that have a circular oscillating motion can reduce plaque and gingivitis more effectively than manual tooth brushing.16 Finally, the physician could recommend dietary changes, such as decreasing the amount and frequency of consumption of foods with a high sugar content.5,8
Pulpitis
Pulpitis, an inflammation of the dental pulp, can occur when caries or its bacterial byproducts encroach on the dental pulp, leading to infection, or when pathogens enter the pulp through the apical foramen or through a tooth fracture. Reversible pulpitis occurs when dental caries encroaches on the pulp; it is associated with mild inflammation of the pulp and mild intermittent pain that can be elicited by thermal changes, especially cold drinks. The patient should be referred to a dentist; treatment involves removal of the carious tissue and reconstruction. However, the pulp may be irreversibly damaged by an ongoing inflammation within the rigid pulp chamber. In this situation, a rapid buildup of pressure, occlusion of blood vessels at the apical foramen, ischemia, and eventual necrosis of the pulp tissue occur. In irreversible pulpitis, the pain is often poorly localized, persistent, and dull. There is insufficient evidence in the literature to determine whether antibiotics are effective in relieving pain in patients with pulpitis.17
A complication of pulpitis is periapical abscess in the periodontal tissue around the apical foramen (Figure 3). Symptoms include tenderness on tapping the affected tooth and local lymphadenopathy. The periapical abscess is usually a localized infection that does not require antibiotic therapy before referral. The dentist will remove the necrotic pulp tissue, drain the pus, and reconstruct or extract the tooth. If the abscess has spread to the adjacent teeth or to deeper tissue, causing cellulitis, antibiotic treatment should be initiated before referral. A broad-spectrum, well-tolerated antimicrobial such as amoxicillin/clavulanate (Augmentin) or clindamycin (Cleocin) is recommended.18,19 If facial swelling, fever, and trismus (inability to open the jaw) are present, indicating severe cellulitis that involves deeper orofacial spaces, the patient should be hospitalized to receive intravenous antibiotics.
Figure 3.
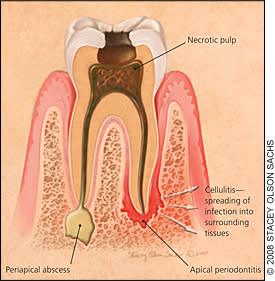
Pulpitis and its complications: periapical abscess and cellulitis.
Periodontal Diseases
Periodontal disease is caused mainly by microorganisms within the subgingival dental plaque. The pathogens can penetrate the gingival epithelium, elicit an inflammatory response, and ultimately cause the destruction of the periodontium. Periodontal disease can be classified into gingivitis and periodontitis. Periodontal infections may be transmissible by saliva20,21 and occur more readily in susceptible hosts. These infections could be a cause of fever of unknown origin.22,23 Furthermore, dental procedures that treat periodontal diseases may lead to bacteremia that can pose serious risks for older or immunocompromised patients.24–26 Patients who have structural heart disorders are particularly vulnerable to endocarditis after certain periodontal procedures, and new recommendations on antibiotic prophylaxis are available.27,28 Research supports an association among periodontal infections, atherosclerosis, and vascular disease.29–31 Recent studies have shown that successful treatment of periodontitis improves endothelial function.32 However, no large-scale studies have tested whether treating or preventing periodontal infections leads to fewer clinical cardiovascular events.33
GINGIVITIS
Gingivitis is characterized by localized inflammation of the gums without a loss of the bone that supports the teeth (Figure 4). Symptoms include erythema and swollen, bleeding gums with brushing or flossing. Pain is usually not associated with gingivitis, but there may be halitosis. Good oral hygiene, including frequent tooth brushing and use of chlorhexidine gluconate (Peridex) 0.12% or hexetidine (Oraldene) 0.1% rinse, usually reverses gingivitis.34 A mouth rinse containing essential oils (e.g., Listerine Antiseptic) is as effective as chlorhexidine in the treatment of gingivitis and may result in less calculus accumulation and tooth staining.35
Figure 4.
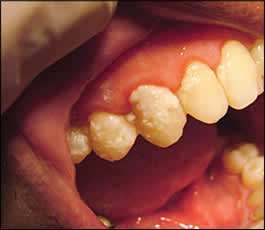
Gingivitis of the upper gum.
PERIODONTITIS
Periodontitis is characterized by the loss of supportive bone structure caused by chronic gingivitis; this results in detachment of the periodontal ligament from the tooth (Figure 5). Periodontitis can be classified as juvenile or adult according to the patient's age and to a slight difference in the microbiology and pathogenesis. In localized juvenile periodontitis (12 to 17 years of age),36 there is very little dental plaque involvement, but there is a loss of vertical alveolar bone. Adult periodontitis normally occurs in patients older than 30 years, and the periodontitis is usually asymptomatic. Clinical examination may show deep gum pockets that bleed easily when probed, subgingival dental plaque, and receding gums that expose the root of the tooth. Periodontal abscess is a severe consequence of periodontitis and may present as a red, fluctuant swelling of the gingiva that is extremely tender to palpation (Figure 6). The abscess may be focal or, if diffuse, may spread to deeper oral spaces, causing swelling of the face and jaw and lymphadenopathy.
Figure 5.
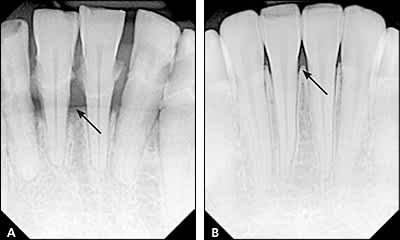
Radiographs of (A) alveolar bone loss in periodontitis (arrow) compared with (B) normal alveolar bone structure (arrow).
Figure 6.
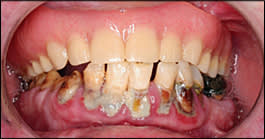
Advanced periodontitis of the lower teeth.
Antibiotics are not normally indicated if mechanical debridement is successful in the case of localized periodontitis. Adding chlorhexidine rinse after surgical debridement contributes to faster recovery of periodontal tissue.37 In generalized periodontitis involving multiple teeth, patients should be treated with antibiotics as an adjunct therapy. In patients with juvenile periodontitis, tetracycline is safe to use after 12 years of age, but the pathogens in this disease have been increasingly resistant to this treatment regimen.38 Adult periodontitis can be treated with doxycycline (Vibramycin), metronidazole (Flagyl), or topical application of minocycline microspheres (Arestin). If cellulitis occurs, patients should be treated with antibiotics.39,40
PERICORONITIS
Pericoronitis is an acute, localized infection caused by food particles and microorganisms trapped beneath the gingival flaps of a partially erupted tooth or an impacted wisdom tooth (Figure 7). Symptoms typically include localized pain and limitation of movement on opening the jaw, discomfort on chewing and swallowing, and facial swelling. Localized lymphadenopathy may be noted, and halitosis is common. Treatment of a localized pericoronitis involves removal of the food particles and good oral hygiene, which should include hot salt water or chlorhexidine rinses before referral. In severe cases of pericoronitis, the treatment would include antibiotics and referral to a dental professional.
Figure 7.
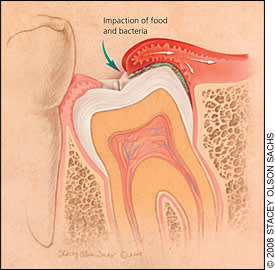
Pericoronitis of an impacted wisdom tooth.

