Foot infections are common in patients with diabetes and are associated with high morbidity and risk of lower extremity amputation. Diabetic foot infections are classified as mild, moderate, or severe. Gram-positive bacteria, such as Staphylococcus aureus and beta-hemolytic streptococci, are the most common pathogens in previously untreated mild and moderate infection. Severe, chronic, or previously treated infections are often polymicrobial. The diagnosis of diabetic foot infection is based on the clinical signs and symptoms of local inflammation. Infected wounds should be cultured after debridement. Tissue specimens obtained by scraping the base of the ulcer with a scalpel or by wound or bone biopsy are strongly preferred to wound swabs. Imaging studies are indicated for suspected deep soft tissue purulent collections or osteomyelitis. Optimal management requires aggressive surgical debridement and wound management, effective antibiotic therapy, and correction of metabolic abnormalities (mainly hyperglycemia and arterial insufficiency). Treatment with antibiotics is not required for noninfected ulcers. Mild soft tissue infection can be treated effectively with oral antibiotics, including dicloxacillin, cephalexin, and clindamycin. Severe soft tissue infection can be initially treated intravenously with ciprofloxacin plus clindamycin; piperacillin/tazobactam; or imipenem/cilastatin. The risk of methicillin-resistant S. aureus infection should be considered when choosing a regimen. Antibiotic treatment should last from one to four weeks for soft tissue infection and six to 12 weeks for osteomyelitis and should be followed by culture-guided definitive therapy.
In patients with diabetes, any foot infection is potentially serious. Diabetic foot infections range in severity from superficial paronychia to deep infection involving bone. Types of infection include cellulitis, myositis, abscesses, necrotizing fasciitis, septic arthritis, tendinitis, and osteomyelitis. Foot infections are among the most common and serious complications of diabetes mellitus. They are associated with increased frequency and length of hospitalization and risk of lower extremity amputation.1 Foot ulceration and infection are the leading risk factors for amputation.2 Prevention and prompt diagnosis and treatment are necessary to prevent morbidity, especially amputation.
SORT: KEY RECOMMENDATIONS FOR PRACTICE
| Clinical recommendation | Evidence rating | References |
|---|---|---|
| The existence, severity, and extent of infection, as well as vascular status, neuropathy, and glycemic control should be assessed in patients with a diabetic foot infection. | C | 3, 9 |
| Visible bone and palpable bone on probing are suggestive of underlying osteomyelitis in patients with a diabetic foot infection. | C | 13, 14 |
| Before an infected wound of a diabetic foot infection is cultured, any overlying necrotic debris should be removed to eliminate surface contamination and to provide more accurate results. | C | 3, 17 |
| Routine wound swabs and cultures of material from sinus tracts are unreliable and strongly discouraged in the management of diabetic foot infection. | B | 17–19 |
| The empiric antibiotic regimen for diabetic foot infection should always include an agent active against Staphylococcus aureus, including methicillin-resistant S. aureus if necessary, and streptococci. | A | 3, 5, 7, 8 |
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, see https://www.aafp.org/afpsort.xml.
Pathophysiology
Patients with diabetes are particularly susceptible to foot infection primarily because of neuropathy, vascular insufficiency, and diminished neutrophil function.3 Peripheral neuropathy has a central role in the development of a foot infection and it occurs in about 30 to 50 percent of patients with diabetes. Patients with diabetes lose the protective sensations for temperature and pain, impairing awareness of trauma such as abrasions, blistering, or penetrating foreign body. Motor neuropathy can result in foot deformities (e.g., claw toe) that contribute to local pressure from footwear, making skin ulceration even more likely. Once the skin is broken (typically on the plantar surface), the underlying tissues are exposed to colonization by pathogenic organisms. The resulting wound infection may begin superficially, but with delay in treatment and impaired body defense mechanisms caused by neutrophil dysfunction and vascular insufficiency, it can spread to the contiguous subcutaneous tissues and to even deeper structures.3,4
Although most diabetic foot infections begin with an ulcer, localized cellulitis and necrotizing fasciitis can develop in the absence of an ulcer or traumatic injury.
Microbiology
The most common pathogens in acute, previously untreated, superficial infected foot wounds in patients with diabetes are aerobic gram-positive bacteria, particularly Staphylococcus aureus and beta-hemolytic streptococci (group A, B, and others).5 Infection in patients who have recently received antibiotics or who have deep limb-threatening infection or chronic wounds are usually caused by a mixture of aerobic gram-positive, aerobic gram-negative (e.g., Escherichia coli, Proteus species, Klebsiella species), and anaerobic organisms (e.g., Bacteroides species, Clostridium species, Peptococcus and Peptostreptococcus species).5 Anaerobic bacteria are usually part of mixed infections in patients with foot ischemia or gangrene.6 Methicillin-resistant S. aureus (MRSA) is a more common pathogen in patients who have been previously hospitalized or who have recently received antibiotic therapy. MRSA infection can also occur in the absence of risk factors because of the increasing prevalence of MRSA in the community.7,8
Clinical Evaluation
Figure 1. Evaluation and Treatment of Diabetic Foot Infection
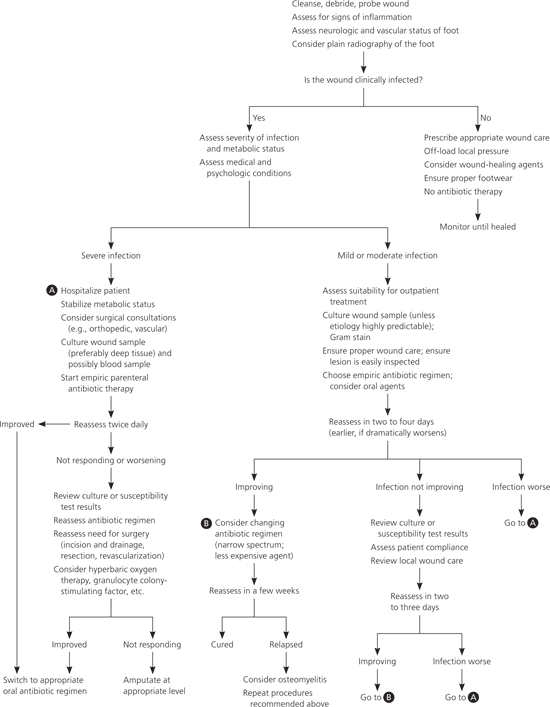
Algorithm for the evaluation and treatment of diabetic foot infection.
Adapted with permission from Lipsky BA. Medical treatment of diabetic foot infections. Clin Infect Dis. 2004;(39 suppl 2):S110.
CONFIRMING THE DIAGNOSIS
Diabetic foot infection must be diagnosed clinically rather than bacteriologically because all skin ulcers harbor micro-organisms (Figure 2). The clinical diagnosis of foot infection is based on the presence of purulent discharge from an ulcer or the classic signs of inflammation (i.e., erythema, pain, tenderness, warmth, or induration). Other suggestive features of infection include foul odor, the presence of necrosis, and failure of wound healing despite optimal management.10 Local inflammatory findings may be less prominent or absent in some diabetic foot infections. For example, pain and tenderness may be reduced or absent in patients who have neuropathy, whereas erythema may be absent in those with vascular disease.11 Acute Charcot's foot is characterized by a progressive deterioration of weight-bearing joints, usually in the foot or ankle. It can clinically mimic cellulitis and presents as erythema, edema, and elevated temperature of the foot. Most patients with diabetic foot infection do not have systemic features such as fever or chills. The presence of systemic signs or symptoms indicates a severe deep infection.12
Figure 2.
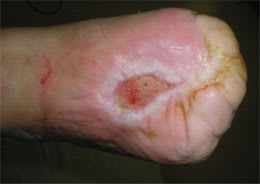
A noninfected ulcer of the dorsum of the foot in a patient with previous amputation of the toes.
ESTABLISHING EXTENT OF INFECTION
Early recognition of the area of involved tissue can facilitate appropriate management and prevent progression of the infection (Figure 3). The wound should be cleansed and debrided carefully to remove foreign bodies or necrotic material and should be probed with a sterile metal instrument to identify any sinus tracts, abscesses, or involvement of bones or joints.
Figure 3.
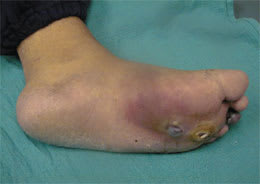
Plantar foot ulcers with a deep space infection.
Osteomyelitis is a common and serious complication of diabetic foot infection that poses a diagnostic challenge. A delay in diagnosis increases the risk of amputation.13 Risk factors associated with osteomyelitis are summarized in Table 1.3,13–16 Visible bone and palpable bone by probing are suggestive of underlying osteomyelitis in patients with a diabetic foot infection.13–14 Laboratory studies, such as white blood cell count and the erythrocyte sedimentation rate (ESR), have limited sensitivity for the diagnosis of osteomyelitis. Osteomyelitis is unlikely with normal ESR values; however, an ESR of more than 70 mm per hour supports a clinical suspicion of osteomyelitis.13 Definitive diagnosis requires percutaneous or open bone biopsy. Bone biopsy is recommended if the diagnosis of osteomyelitis remains in doubt after imaging.3
Table 1 Risk Factors for Osteomyelitis in Patients with Diabetic Foot Infection
| Appearance of a swollen, deformed red toe (also called sausage toe) |
| Bone visible or palpable on probing |
| Infected ulcer with an erythrocyte sedimentation rate of more than 70 mm per hour |
| Nonhealing ulcer after a few weeks of appropriate care and off-loading of pressure |
| Radiologically evident bone destruction beneath ulcer |
| Ulcer area greater than 2 cm2 or more than 3 mm deep |
| Ulceration presents over bony prominences for more than two weeks |
| Ulceration with unexplained leukocytosis |
ESTABLISHING SEVERITY OF INFECTION
The severity of the infection determines the appropriate antibiotic regimen and route of administration. It also is the primary consideration in determining the need for hospitalization and the indications and timing for any surgical intervention. A practical and simple approach to classifying diabetic foot infection is provided in Table 2.3
Table 2 Clinical Classification of Diabetic Foot Infection
| Clinical manifestations of infection | Infection severity |
|---|---|
| Wound lacking purulence or any manifestations of inflammation (i.e., erythema, pain, tenderness, warmth, or induration) | Not infected |
| Presence of purulence and/or two or more manifestations of inflammation, but any cellulitis or erythema extends 2 cm or less around the ulcer; infection is limited to the skin or superficial subcutaneous tissues; no other local complications or systemic illness | Mild |
| Infection (purulence and/or two or more manifestations of inflammation) in a patient who is systemically well and metabolically stable, but who has at least one of the following characteristics: cellulitis extending more than 2 cm around the ulcer; lymphangitic streaking; spread beneath the superficial fascia; deep tissue abscess; gangrene; involvement of muscle, tendon, joint, or bone | Moderate |
| Infection (purulence and/or two or more manifestations of inflammation) in a patient with systemic toxicity or metabolic instability (e.g., fever, chills, tachycardia, hypotension, confusion, vomiting, leukocytosis, acidosis, severe hyperglycemia, azotemia) | Severe |
Adapted from Lipsky BA, Berendt A, Deery G, et al., for the Infectious Diseases Society of America. Diagnosis and treatment of diabetic foot infections. Clin Infect Dis. 2004;39(7):894.
OBTAINING CULTURES
Before an infected wound is cultured, any overlying necrotic debris should be removed by scrubbing the wound with saline-moistened sterile gauze to eliminate surface contamination.3,17 For wound culture, tissue specimens should be obtained by scraping the base of the ulcer with a scalpel or curette, or by a biopsy of the wound or bone. Needle aspiration of the pus or tissue fluid performed aseptically is an acceptable alternative method. Cultures of wound swabs or material from sinus tracts are unreliable and are strongly discouraged.17–19 The specimen should be processed for a Gram-stained smear and aerobic and anaerobic cultures.
ASSESSING VASCULAR STATUS
Peripheral artery disease (PAD) can be diagnosed by absence of foot pulses and reduced ankle-brachial index (ABI). Calculation of ABI is done by measuring the resting systolic blood pressure in the ankle and arm using a Doppler probe. An ABI of 0.91 to 1.30 is borderline or normal. An ABI of 0.41 to 0.90 indicates mild to moderate PAD and an ABI of 0.40 or less indicates advanced ischemia. An ABI greater than 1.30 suggests the presence of calcified vessels and the need for additional vascular studies, such as pulse volume recording or measurement of the toe-brachial index. Patients with atypical symptoms, or whose diagnosis is in doubt, should have ABI measured after exercise on a treadmill. An ABI that decreases by 20 percent following exercise is diagnostic of PAD, whereas a normal ABI following exercise rules out PAD. If a PAD diagnosis is confirmed and revascularization is planned, magnetic resonance angiography, computed tomography angiography, or contrast angiography can be performed for anatomic evaluation.20
Venous insufficiency can be diagnosed clinically by the presence of edema and skin changes and confirmed by duplex ultrasonography.
ASSESSING NEUROPATHY
Touch, vibration, and pressure sensations should be checked routinely using cotton wool, tuning fork, and 10-g nylon monofilament, respectively.
Diagnostic Imaging
Diagnostic imaging is not necessary for every patient with diabetes who has a foot infection. Plain radiography of the foot is indicated for detection of osteomyelitis, foreign bodies, or soft tissue gas. Bony abnormalities associated with osteomyelitis may be indistinguishable from the destructive effects of Charcot's foot and are usually not evident on plain radiography until two to four weeks after initial infection.21 When plain radiography is negative but osteomyelitis is clinically suspected, radio-nuclide scan or magnetic resonance imaging should be performed (Table 321–23). Combining technetium bone scan with gallium scan or white blood cell scan may improve the diagnostic yield for osteomyelitis.21,22 Magnetic resonance imaging provides more accurate information regarding the extent of the infectious process.23 Ultrasonography and computed tomography are also helpful in evaluating abnormalities in the soft tissue (e.g., abscess, sinus tract, cortical bone involvement) and may provide guidance for diagnostic and therapeutic aspiration, drainage, or tissue biopsy.21
Table 3 Diagnostic Imaging Studies for Osteomyelitis of the Foot in Patients with Diabetes
| Imaging study | Sensitivity (%) | Specificity (%) | Comments |
|---|---|---|---|
| Plain radiography | 43 to 75 | 65 to 83 | Lateral, anteroposterior, and oblique views should be done initially in all patients with diabetes who are suspected to have a deep infection |
| Because 30 to 50 percent of the bone must be destroyed before lytic lesions appear, plain radiography should be repeated at two-week intervals if initial findings are not normal, but the infection fails to resolve | |||
| Soft tissue swelling and subperiosteal elevation are the earliest findings of osteomyelitis on plain radiography | |||
| Magnetic resonance imaging | 82 to 100 | 75 to 96 | Useful in between soft tissue and bone infection and for determining the extent of infection |
| Should be considered for patients with diabetes who have an infection with no bone exposed, who have been treated for two to three weeks with modest clinical improvement, and who have negative or inconclusive results on plain radiography | |||
| Technetium-99m methylene diphosphonate bone scan | 69 to 100 | 38 to 82 | High sensitivity for osteomyelitis and can differentiate it from cellulitis |
| Abnormal findings for osteomyelitis (which typically become evident within 24 to 48 hours after onset of symptoms) include increased flow activity, blood pool activity, and positive uptake on three-hour images | |||
| Specificity for osteomyelitis is decreased in patients with diabetes who have Charcot's foot or recent trauma or surgery; further imaging is usually required | |||
| Gallium-67 citrate scan | 25 to 80 | 67 to 85 | Sensitivity and specificity are increased when combined with technetium bone scan |
| Technetium-99m hexamethyl-propyleneamine oxime-labeled white blood cell scan | 90 | 80 to 90 | The main advantage is the marked improvement in specificity when combined with technetium bone scan |
| Computed tomography | 24 to 67 | 50 | Should not be used as part of regular osteomyelitis imaging |
| Superior to magnetic resonance imaging for detecting sequestra |
Treatment
Effective management of diabetic foot infection requires appropriate antibiotic therapy, surgical drainage, debridement and resection of dead tissue, appropriate wound care, and correction of metabolic abnormalities.
ANTIBIOTIC THERAPY
The selection of antibiotic therapy for diabetic foot infection involves decisions about choice of empiric and definitive antibiotic agent, route of administration, and duration of treatment ( Tables 43,9 and 53,24–30). Initial empiric antibiotic therapy should be based on the severity of the infection, history of recent antibiotic treatment, previous infection with resistant organisms, recent culture results, current Gram stain findings, and patient factors (e.g., drug allergy). A Gram-stained smear of an appropriate wound specimen may help guide therapy. The overall sensitivity of a Gram-stained smear for identifying organisms that grow on culture is 70 percent.9 The empiric antibiotic regimen for diabetic foot infection should always include an agent active against S. aureus, including MRSA if necessary, and streptococci.3,5,7,8
Table 4 Principles of Antibiotic Therapy for Diabetic Foot Infection
| Empiric antibiotic regimen should include an agent active against Staphylococcus aureus, including methicillin-resistant S. aureus if necessary, and streptococci |
| Coverage for aerobic gram-negative pathogens is required for severe infection, chronic infection, or infection that fails to respond to recent antibiotic therapy |
| Necrotic, gangrenous, or foul-smelling wounds usually require antianaerobic therapy |
| Initial empiric antibiotic therapy should be modified on the basis of the clinical response and culture or susceptibility testing |
| Virulent organisms, such as S. aureus and streptococci, should always be covered in polymicrobial infection |
| Coverage for less virulent organisms, such as coagulase-negative staphylococci, may not be needed |
| Parenteral antibiotics are indicated for patients who are systemically ill, have severe infection, are unable to tolerate oral agents, or have infection caused by pathogens that are not susceptible to oral agents |
| Using oral antibiotics for mild to moderate infection and switching early from parenteral to oral antibiotics with appropriate spectrum coverage and good bioavailability and tolerability are strongly encouraged |
| Although topical antibiotics can be effective for the treatment of mildly infected ulcers, they should not be routinely used |
| Discontinuation of antibiotics should be considered when all signs and symptoms of infection have resolved, even if the wound has not completely healed |
| Cost should be considered when selecting antibiotic therapy |
Table 5 Empiric Antibiotic Regimens for Treatment of Diabetic Foot Infection
| Severity or extent of infection | Recommended therapy | Comments | ||
|---|---|---|---|---|
| Soft tissue infection | ||||
| Mild (duration of treatment is one to two weeks) | Dicloxacillin 500 mg orally four times per day | Oral agent of choice for MSSA | ||
| Cephalexin (Keflex) 500 mg orally four times per day | For penicillin-allergic patients, except those with immediate hypersensitivity reactions | |||
| Amoxicillin/clavulanate (Augmentin) 875/125 mg orally twice per day | Good option for polymicrobial infection | |||
| Clindamycin (Cleocin) 300 to 450 mg orally three times per day | Potential cross-resistance and emergence of resistance in erythromycin-resistant Staphylococcus aureus; inducible resistance in MRSA | |||
| Doxycycline (Vibramycin) 100 mg orally twice per day or Sulfamethoxazole/trimethoprim (Bactrim) 160/800 mg orally twice per day | Effective for MRSA | |||
| Moderate (duration of treatment is two to four weeks, depending on response; administer orally or parenterally followed by orally) | ||||
| Risk factors for polymicrobial infection absent* | Nafcillin 1 to 2 g IV every four hours | Parenteral drug of choice for MSSA | ||
| Cefazolin 1 to 2 g IV every eight hours | For penicillin-allergic patients | |||
| Vancomycin 30 mg per kg IV twice per day | Parenteral drug of choice for MRSA | |||
| Risk factors for polymicrobial infection present* | Ampicillin/sulbactam (Unasyn) 3 g IV four times per day | — | ||
| Ceftriaxone (Rocephin) 1 to 2 g IV once per day plus clindamycin 600 to 900 mg IV or orally three times per day or metronidazole (Flagyl) 500 mg IV or orally three times per day | — | |||
| or | ||||
| Levofloxacin (Levaquin) 500 mg IV or orally once per day plus clindamycin 600 to 900 mg IV or orally three times per day | ||||
| Moxifloxacin (Avelox) 400 mg IV or orally once per day | — | |||
| Ertapenem (Invanz) 1 g IV once per day | — | |||
| Severe (duration of treatment is two to four weeks, depending on response; administer parenterally, then switch to orally) | Ciprofloxacin (Cipro) 400 mg IV twice per day plus clindamycin 600 to 900 mg IV three times per day | — | ||
| Piperacillin/tazobactam (Zosyn) 3.375 to 4.500 g IV every six to eight hours | — | |||
| Imipenem/cilastatin (Primaxin) 500 mg IV four times per day | — | |||
| Vancomycin 30 mg per kg IV twice per day plus ciprofloxacin 400 mg IV twice per day plus metronidazole 500 mg IV or orally three times per day | Vancomycin is the parenteral drug of choice for MRSA; linezolid (Zyvox) 600 mg IV or orally twice per day or daptomycin (Cubicin) 4 mg per kg IV once per day can also be used for MRSA Use vancomycin for penicillin-allergic patients | |||
| Tigecycline (Tygacil) 100 mg IV loading dose then 50 mg IV twice per day | Should be used when suspected polymicrobial infection, including MRSA | |||
| Bone or joint infection | ||||
| No residual infected tissue | Use the above parenteral or oral antibiotic regimens for two to five days | — | ||
| Residual infected tissue only | Use the above parenteral or oral antibiotic regimens for two to four weeks | — | ||
| Residual infected viable bone | Initially use the above parenteral antibiotics followed by oral antibiotics for four to six weeks | — | ||
| Residual infected dead bone | Initially use the above parenteral antibiotics followed by oral antibiotics for eight to 12 weeks | — | ||
IV = intravenously; MRSA = methicillin-resistant Staphylococcus aureus; MSSA = methicillin-susceptible S. aureus.
*— Risk factors for polymicrobial infection include chronic ulcers, recent antibiotic use, and foot ischemia or gangrene.
The patient should be reassessed 24 to 72 hours after initiating empiric antibiotic therapy to evaluate the response and to modify the antibiotic regimen, if indicated by early culture results. Several antibiotics have been shown to be effective, but no single regimen has shown superiority.3,24–30 Antibiotic therapy should not be used for foot ulcers without signs of infection because it does not enhance wound healing or prevent infection.31 Clinical failure of appropriate antibiotic therapy might be because of patient nonadherence, antibiotic resistance, superinfection, undiagnosed deep abscess or osteomyelitis, or severe tissue ischemia.
SURGICAL TREATMENT
Surgery is the cornerstone of treatment for deep diabetic foot infection. Procedures range from simple incision and drainage to extensive multiple surgical debridements and amputation. Timely and aggressive surgical debridement or limited resection or amputation may reduce the need for more extensive amputation.32 Emergent surgery is required for severe infection in an ischemic limb, necrotizing fasciitis, gas gangrene, and an infection associated with compartment syndrome. Surgical excision of affected bone has historically been the standard of care in patients with osteomyelitis. Nevertheless, successful therapy with a long course of antibiotics alone has been achieved in two thirds of patients with osteomyelitis.12 As infection is controlled and the wound starts to granulate, primary closure may be successful. The wound may also be treated surgically with a flap or graft, left to heal by secondary intention, or managed with negative pressure dressings.33
If the infected limb appears to be ischemic, the patient should be referred to a vascular surgeon. Although noncritical ischemia can usually be treated without a vascular procedure, early revascularization within a few days of the infection is required for successful treatment of an infected foot with critical ischemia.34
WOUND MANAGEMENT
The wound should be dressed to allow for careful inspection for evidence of healing and early identification of new necrotic tissue. Necrotic or unhealthy tissue should be debrided, preferably surgically or with topical debriding agents. Removing pressure from the foot wound is crucial for healing35 and can be achieved through total contact casting, removable cast walkers, and various ambulatory braces, splints, modified half-shoes, and sandals.36 Edema of the legs can delay wound healing; controlling edema with leg elevation, compression stockings, or a pneumatic pedal compression device enhances the healing process.37 Evidence of resolution of infection includes formation of granulation tissue, absence of necrotic tissue, and closing of the wound. If osteomyelitis is present, signs of healing include a drop in ESR and loss of increased uptake on nuclear scan.
METABOLIC STABILITY
Correction of fluid and electrolyte imbalances, hyperglycemia, acidosis, and azotemia is essential. Good glycemic control may help eradicate the infection and promote wound healing.38 All patients should have blood glucose and A1C levels measured at initial presentation and then at regular intervals. Frequent home blood glucose monitoring is strongly encouraged. Appropriate therapeutic adjustments (e.g., adding or changing oral antihyperglycemic agents, initiating or increasing insulin) must be made to optimize glycemic control.
OTHER THERAPIES
Maggot debridement therapy, granulocyte colony-stimulating factor, and hyperbaric oxygen therapy have been used for diabetic foot infection, but should not be used routinely because of lack of evidence of effectiveness.3
Prevention
Prevention of diabetic foot ulcers begins with identifying patients at risk. All patients with diabetes should have an annual foot examination that includes assessment for anatomic deformities, skin breaks, nail disorders, loss of protection sensation, diminished arterial supply, and inappropriate footwear. Patients at higher risk of foot ulceration should have examinations more often.39 Educating patients and caretakers about proper foot care and periodic self-foot examinations are effective interventions to prevent ulceration. Other effective clinical interventions include optimizing glycemic control, smoking cessation, debridement of calluses, and certain types of prophylactic foot surgery.40

