
Am Fam Physician. 2009;80(12):1413-1418
Patient information: See related handout on latex allergy, written by the authors of this article.
Author disclosure: Nothing to disclose.
The prevalence of latex allergy in the general population is low; however, the risk of developing latex allergy is higher in persons with increased latex exposure, such as health care workers or persons who work in the rubber industry. Children with spina bifida and others who undergo multiple surgeries or procedures, particularly within the first year of life, are also at greater risk of latex allergy. Reactions to latex allergy can range from type IV delayed hypersensitivity (e.g., contact dermatitis) to type I immediate hypersensitivity (e.g., urticaria, bronchospasm, anaphylaxis). Latex allergy can be diagnosed with clinical history, skin prick testing, latex-specific serum immunoglobulin E testing, and glove provocation testing. The main goals of latex allergy management are avoidance of exposure to latex allergens and appropriate treatment of allergic reactions. The use of nonlatex products from birth may prevent potentially serious allergic reactions. Widespread adoption of nonlatex or low-latex gloves has decreased the incidence of latex sensitization in health care workers.
Although delayed hypersensitivity to latex is long established, immediate hypersensitivity to latex is relatively recent, first appearing in medical literature in 1979.1 Since then, the number of reported cases of immediate hypersensitivity reactions to latex has grown, with peak incidence in the 1980s.2 This is attributed to the increased use of latex gloves as a universal infection control precaution, as well as to greater awareness and recognition of allergy to latex.2,3 Natural rubber latex comes from the rubber tree Hevea brasiliensis, and is found in many common products3 (Table 14).
| Clinical recommendation | Evidence rating | References |
|---|---|---|
| Switching to low-protein, powder-free latex gloves or to latex-free gloves provides a primary prevention of latex allergy. | C | 10, 13–15, 28 |
| Serum IgE measurement is considered the most useful test for confirming suspected severe latex allergy and carries no risk of anaphylaxis. Glove provocation testing is useful when the patient's clinical history is incongruent with IgE results, although it is not considered first-line testing. Skin patch testing is sensitive for diagnosing type IV delayed allergy symptoms. | C | 2, 3, 12 |
| Children with spina bifida or urogenital anomalies, or those who may have multiple surgical procedures, should avoid latex exposure from birth to prevent the development of latex allergy. | C | 7, 8, 26, 27 |
| Sublingual immunotherapy with latex may be effective in patients with severe latex allergy, especially in patients who cannot avoid all exposure to latex. | B | 30–32 |
| Balloons |
| Bandages (adhesives) |
| Blood pressure cuffs |
| Condoms |
| Dental dams |
| Diaphragms |
| Elastic |
| Gloves |
| Gutta-percha and gutta-balata (materials used to seal root canals) |
| Pacifiers (baby bottle nipples) |
| Spandex |
| Stethoscope tubing |
| Tourniquets |
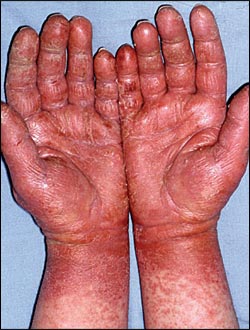
It is clinically important to distinguish between immune and nonimmune reactions to latex (Table 2).5 Immediate type I hypersensitivity reactions are immunoglobulin E (IgE)-mediated responses to latex proteins, and can range from urticaria to anaphylaxis.3,5 Type I reactions should be considered in patients who have immediate skin symptoms on contact with latex gloves.2 Delayed type IV hypersensitivity reactions are usually caused by chemicals, accelerants, and antioxidants in the gloves and not by the latex itself; this leads to a later onset of contact dermatitis symptoms that are typical of type IV reactions3 (Figure 1). Case reports of delayed type IV reaction to latex are rare.3,6
| Type of reaction | Symptoms | Cause | Time of onset |
|---|---|---|---|
| Immediate hypersensitivity (type I) | Urticaria (local or generalized), nausea, vomiting, faintness, rhinitis, conjunctivitis, bronchospasm, anaphylactic shock | Latex | Immediate (within minutes) |
| Delayed hypersensitivity or contact dermatitis (type IV) | Papular, pruritic rash; vesicles; blisters | Chemicals in latex | Delayed (several hours to 48 hours after contact) |
| Irritant contact dermatitis (nonimmune) | Dry, cracked, irritated skin | Chemicals in latex or hand washing | Gradual (over several days) |
Incidence and Prevalence
The incidence of latex allergy in the general population is 1 to 2 percent.3 Patients with spina bifida (myelomeningocele) are at the highest risk of latex allergy because of repeated exposure of mucous membranes to latex during surgeries and procedures.7,8 The prevalence of latex allergy in these patients ranges from 20 to 67 percent,2,3 and their risk of anaphylaxis in the operating room is 500 times higher than that of control groups.3 As few as five operations may increase the risk of clinically significant latex allergy.7
Health care workers have the second highest risk of developing latex allergy, particularly those who work in operating rooms, laboratories, or hemodialysis centers.2,3,9 There is a positive correlation between the risk of latex allergy and the length of employment in the health care industry.9 The sensitization rates in health care workers are three times higher than in the general population.3,10,11 Workers who are exposed to latex on a regular basis are at higher risk than those who are not directly exposed to latex products.12 Exposure to powdered gloves appears to be associated with development of asthmatic symptoms and other allergic symptoms, such as allergic rhinitis, conjunctivitis, and angioedema.3,10,12
Beginning in the late 1990s, German regulations banned the use of powdered latex gloves.13,14 Subsequently, the incidence of health care worker latex allergy from occupational exposure decreased almost 80 percent.14 Similar measures in Finland also led to decreased sensitization rates in health care workers.15 These and other studies demonstrate the impact of primary prevention of latex sensitization by reducing latex glove use in the workplace.10
Etiology
Risk factors for latex allergy include neural tube defects and occupational exposure, as well as a history of atopy, multiple surgeries, previous hand dermatitis of any kind,2,3,16 and allergies to foods known to have allergens that cross-react with latex3,16 (Table 34). A history of atopy increases a person's risk of latex sensitization fourfold.3 Previous hand dermatitis and eczema may allow greater penetration of latex from gloves into dry, cracked skin.3,16
| High association |
| Avocado |
| Banana |
| Chestnut |
| Kiwi |
| Moderate association |
| Apple |
| Carrot |
| Celery |
| Melons |
| Papaya |
| Potato |
| Tomato |
| Low or undetermined association |
| Apricot |
| Buckwheat |
| Castor bean |
| Cayenne pepper |
| Cherry |
| Chickpea |
| Citrus fruits |
| Coconut |
| Dill |
| Fig |
| Grape |
| Hazelnut |
| Lychee |
| Mango |
| Nectarine |
| Oregano |
| Passion fruit |
| Peach |
| Peanut |
| Pear |
| Persimmon |
| Pineapple |
| Plum |
| Rye |
| Sage |
| Shellfish |
| Soybean |
| Strawberry |
| Sunflower seeds |
| Sweet pepper |
| Walnut |
| Wheat |
| Zucchini |
The risk of latex allergy increases with each surgery. In children without neural tube defects, each subsequent surgery can increase the risk of latex allergy 13-fold.3 Approximately one in 7,700 pediatric surgeries is complicated by anaphylaxis; of these complications, 76 percent are from latex allergy.17,18 As in patients with spina bifida, children undergoing multiple surgeries (e.g., for malformations requiring multiple procedures) are at increased risk of a latex allergy.3 Children who are likely to have multiple surgeries early in life should be treated only with latex-free products.7,19 Likewise, adults who have had more than 10 surgeries have a significantly greater risk of developing a latex allergy.3 Of anaphylactic reactions in adult surgery, 12 to 40 percent are from latex allergy.3,18
Diagnosis
Many methods for diagnosing latex allergy have been studied, including questionnaires, preoperative screening, skin prick tests, measurement of latex-specific serum IgE levels, glove provocation tests, and skin patch testing.
HISTORY
A clinical history is essential for diagnosing latex allergy. In patients with a history suggestive of latex sensitization, physicians should ask about skin and respiratory symptoms, as well as food allergies, particularly in patients with a history of atopy.5 There are no well-defined screening recommendations, but one suggested approach is to follow guidelines from the National Institute for Occupational Safety and Health.20 This includes increasing awareness of the harmful effects of latex sensitization, especially for employers with high-risk workers. Patients with latex allergy should notify their physician about the allergy to ensure the use of latex-free products.
Given the increased risk of latex exposure and reactions during surgery, physicians should consider asking all patients to complete a routine preoperative questionnaire about latex-related symptoms. This can help identify patients who may already be sensitized to latex.17 A study of health care workers in Brazil used a patient questionnaire to identify latex sensitization in health care workers. The authors found that self-reported allergic symptoms on the hands and allergic reactions to certain foods were associated with confirmed latex sensitization using skin prick testing.21
Although obtaining a complete history is important and often very helpful, several studies report that a history alone may not be sufficient for diagnosing a type I latex allergy.5,12,22 Self-reported symptoms, although sensitive, do not reliably differentiate allergic reactions from nonallergic irritation.11,16 In an Italian study, only 9 percent of health care workers who reported symptomatic reactions to latex actually had a latex allergy; the remainder had nonallergic irritation.16 In a study of dental students, 10 percent reported reactions to latex, but only 1 percent had a confirmed diagnosis of latex sensitization.11 This suggests that the actual prevalence of latex allergy may be less than what has been reported previously, although more studies are needed to confirm this.
LABORATORY TESTING
Screening for latex allergy in the general population with preoperative laboratory or clinical testing has not been found to be useful and is not indicated.17,18 Preoperative testing may be helpful in high-risk patients (e.g., patients with a high number of previous surgeries, a history of atopy, a known history of latex reaction).17,18
There is no standardized testing protocol for diagnosing latex allergy.12,23 Skin prick testing is the most sensitive test2,3,22–24 and would be considered the preferred test for diagnosing type I immediate hypersensitivity.16,22 However, there are no extracts approved by the U.S. Food and Drug Administration. Skin prick testing is primarily performed in Europe where approved extracts are available.2,3
Because skin prick testing is not available in the United States, measurement of latex-specific serum IgE levels is the best option. It is considered the most useful test for confirming suspected severe allergy because there is no risk of anaphylaxis.3 The sensitivity and specificity of IgE testing is variable (50 to 90 percent and 80 to 87 percent, respectively).2,3 Other barriers include higher cost and longer wait time for results.
Glove provocation testing, or “glove challenge test,” is useful when the patient's clinical history is incongruent with IgE results, although it is not considered a first-line test.2,3 During the test, the patient wears one finger of a latex glove while the physician watches for a reaction. If there is no urticarial reaction after 15 minutes, the exposed surface area is increased. The test concludes when an urticarial response is identified (i.e., a positive provocation test), or when the patient is able to wear the full glove for 15 minutes with no reaction (i.e., a negative provocation test).3,12 Because of the variation of latex content in gloves, this test has a varied sensitivity and could be unsafe in highly sensitized persons.2,12
Skin patch testing is a sensitive test for diagnosing type IV delayed reactions to rubber additives (e.g., chemical accelerators, antioxidants).12 It is performed by applying allergen samples to intact skin and covering them with a dressing. After the patch is removed, the patient is checked for skin reaction at 30 minutes, 24 hours, and 48 hours.3,12
Management
The goals of latex allergy management are prevention of exposure and treatment of reactions.5 Patients with latex allergy can reduce their risk of exposure by avoiding direct contact with latex. They should be aware of common natural rubber products, as well as foods with cross-reactive proteins3 (Tables 1 and 34). Symptoms of latex allergy resolve quickly with avoidance. However, elevated IgE levels can remain detectable more than five years after exposure, suggesting that long-term avoidance of latex should be recommended for patients with known latex allergy.25 Children with spina bifida or urogenital anomalies, or those who are expected to have multiple surgical procedures, should avoid exposure to latex products from birth to prevent development of latex allergy.7,8,26,27
Patients with a history of severe type I allergy may benefit from wearing a medical alert identification, such as a bracelet, necklace, or keychain, and carrying nonlatex gloves. These patients may wish to carry a prescribed epinephrine self-injection pen in the event of a serious reaction, such as anaphylaxis.4 A systematic review showed that substituting powdered latex gloves with low-protein, powder-free, latex gloves or with latex-free gloves can provide primary prevention of latex allergy, as well as cost-savings for employers.28 Another study showed that switching gloves reduced expenses by decreasing the number of missed days of work and workers' compensation claims.29 Patients with type I allergy should also be aware of the potential for hidden latex on food prepared with latex gloves.
Symptoms of contact dermatitis can be treated with topical steroid creams if the rash is localized. If a large area of skin is involved, oral steroids are indicated. Antihistamines can be used for urticaria and to help relieve itching. Bronchodilators for bronchospasm may be needed for respiratory symptoms.5 For anaphylactic reactions, the patient's airway, breathing, and circulation must be assessed and maintained, and epinephrine should be administered. During resuscitation, it is essential to avoid all latex products.5
Randomized controlled trials have suggested that sublingual immunotherapy with latex may be effective in patients with severe latex allergy, especially in those who cannot avoid all exposure to latex.30–32 However, more information on the utility and safety of sublingual immunotherapy is needed, and it is not considered recommended practice at this time.
