Clinical Question
At what age is it safe for children to take aspirin for fever or viral syndromes?
Evidence-Based Answer
Aspirin should not be used to treat acute febrile viral illness in children. (Strength of Recommendation [SOR]: C, based on case-control studies). Although no causal link has been proven, data from case-control and historic cohort studies demonstrate an association between aspirin use and Reye syndrome. The risk of Reye syndrome decreases with age, becoming extremely rare by the late teenage years. Other nonsteroidal anti-inflammatory drugs are effective antipyretics and are not associated with the constellation of symptoms seen in Reye syndrome, which includes nausea, vomiting, headache, excitability, delirium, combativeness, and coma. Aspirin use in children younger than 19 years should be limited to diseases in which aspirin has a proven benefit, such as Kawasaki disease and the juvenile arthritides. (SOR: C, based on expert opinion).
Evidence Summary
The primary concern associated with aspirin use in the treatment of fever in children is Reye syndrome. First described in 1963, Reye syndrome is an acute, noninflammatory encephalopathy and hepatotoxicity that follows an acute viral illness. Although it is associated with salicylate treatment in case-control and historic cohort studies, accurate diagnosis of Reye syndrome is difficult.1–3 From 1980 to 2000, approximately 25 percent of initial Reye syndrome diagnoses were subsequently changed.4,5
Aspirin's role in the pathogenesis of Reye syndrome is unclear, and some believe the syndrome is caused by a viral infection.2,6 However, case-control studies and other historic data associate use of salicylates during febrile viral infections to the development of Reye syndrome in children.7–9 A comprehensive review of studies linking aspirin to Reye syndrome concludes that the methodology of many early studies was flawed, but later cohort studies linking Reye syndrome to aspirin employed methodologies that addressed those criticisms.4
The absolute risk of aspirin treatment is unknown and difficult to calculate precisely. Peak incidence of Reye syndrome in the United States occurred in 1980, when 555 cases were reported. Since peaking in 1980, the incidence of Reye syndrome has dropped to less than two cases per year.2 This dramatic decline coincides with increasing reports linking aspirin to Reye syndrome. In 1986 the U.S. Food and Drug Administration (FDA) required that aspirin labels state that children and teenagers should not use the product.1 Today, Reye syndrome is rare. Recent estimates indicate 0.2 to 1.1 cases per 1 million children younger than 18 years.3
No study indicates the lowest “safe” age for salicylate use during viral infections. Epidemiologic data show that only 8 percent of Reye syndrome cases occur in children 15 to 17 years of age; the syndrome is extremely rare in patients older than 18 years. Figure 1 summarizes the age-related prevalence data.3,10
Figure 1.
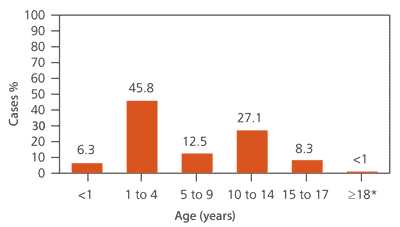
Bar graph showing the percentage of reported Reye syndrome cases by age.
Percentage of Reported Reye Syndrome Cases by Age
*—There are case reports of Reye syndrome occurring in adults up to 61 years of age.10
Information from references 3 and 10.
Recommendations from Others
The National Reye's Syndrome Foundation, the U.S. Surgeon General, the FDA, the Centers for Disease Control and Prevention, and the American Academy of Pediatrics recommend that aspirin and combination products containing aspirin not be taken by anyone younger than 19 years during fever-causing illnesses.11 The British Medicines and Health-care products Regulatory Agency recommends that aspirin labels state that aspirin is not for use in children younger than 16 years unless recommended by a physician.12

