The increasing incidence of skin and soft tissue infections requires family physicians to be familiar with the management of these conditions. Evidence of systemic infection, such as fever, tachycardia, and hypotension, is an indication for inpatient management. Urgent surgical referral is imperative for those with life-threatening or rapidly advancing infections. In patients with uncomplicated abscesses measuring less than 5 cm in diameter, surgical drainage alone is the primary therapeutic intervention. Wound irrigation using tap water has similar outcomes as irrigation using sterile water. When antimicrobials are indicated, choice of agents depends on local resistance and susceptibility patterns. In settings where suspicion of methicillin-resistant Staphylococcus aureus (MRSA) is low, beta-lactam antibiotics are the first-line treatments for uncomplicated skin and soft tissue infections without focal coalescence or trauma. When empiric coverage for MRSA is indicated and the infection is uncomplicated, oral agents, such as tetracyclines, trimethoprim/sulfamethoxazole, and clindamycin, are preferred. Vancomycin is the first-line agent for MRSA in hospitalized patients, and newer agents, such as linezolid, daptomycin, and tigecycline, should be reserved for patients who do not respond to or cannot tolerate vancomycin therapy. There are insufficient data to support eradicating the carrier state in patients with MRSA or their contacts with nasal mupirocin or antibacterial body washes. Standard infection-control precautions, including proper and frequent handwashing, are a mainstay of MRSA prevention.
Superficial soft tissue infections are increasingly common in the outpatient setting. The diagnosis of skin infections increased nearly threefold in U.S. emergency departments from 1993 to 2005.1 A large increase in community-acquired methicillin-resistant Staphylococcus aureus (MRSA) infections has prompted changes in the approach to skin and soft tissue infections (SSTIs). This article reviews updated diagnostic and treatment strategies for immunocompetent patients, particularly those with community-acquired MRSA.
SORT: KEY RECOMMENDATIONS FOR PRACTICE
| Clinical recommendation | Evidence rating | References | Comments |
|---|---|---|---|
| Wound and blood cultures are not necessary in patients with uncomplicated SSTIs because results rarely alter management decisions. | C | 17, 18 | Retrospective analyses |
| Incision and drainage alone is often curative for uncomplicated SSTIs with abscesses measuring less than 5 cm in diameter. | A | 4, 20, 21 | Retrospective chart review; randomized, double-blind trials |
| When surgical drainage of SSTIs is performed, there is no difference in clinical outcomes between wound irrigation with tap water versus sterile water. | A | 22, 23 | Prospective trials from urban pediatric emergency departments |
| When initiating empiric antimicrobial therapy for uncomplicated SSTIs, physicians should consider local prevalence of pathogens, including MRSA, and resistance patterns. | C | 12 | Expert opinion |
| Eradication of MRSA carrier state is not associated with reduced incidence of clinical MRSA infection. | A | 36, 37 | Randomized, double-blind trial; Cochrane review |
MRSA = methicillin-resistant Staphylococcus aureus ; SSTI = skin and soft tissue infection.
A = consistent, good-quality patient-oriented evidence; B = inconsistent or limited-quality patient-oriented evidence; C = consensus, disease-oriented evidence, usual practice, expert opinion, or case series. For information about the SORT evidence rating system, go to https://www.aafp.org/afpsort.xml.
Although the clinical manifestations of SSTIs are varied (Table 12,3 ), the most common include cellulitis (Figure 1), furuncles, abscesses (Figure 2), impetigo (Figure 3), and erysipelas.4 SSTIs often occur at the site of a disruption in the epidermal layer. In some cases, the portal of entry is remote from the site of infection, such as in patients with lower extremity cellulitis associated with tinea pedis. Risk factors for SSTIs include trauma and burns; intravenous drug use; chronic conditions, such as diabetes or malignancy; previous SSTI; surgeries that disrupt the lymph tracts, such as saphenous vein harvesting or axillary lymph node dissections; and animal or human bites.5,6
Table 1. Characteristics and Microbiology of SSTI Manifestations
| Manifestation | Characteristics | Microbiology | Comments |
|---|---|---|---|
| Purulent SSTIs | |||
| Abscess | Collection of pus within dermis, erythema with fluctuance on palpation, overlying pustule may be present | Polymicrobial, commonly skin flora (staphylococci and streptococci), organisms from adjacent mucous membranes | Considered complicated if perianal or perineal areas are affected |
| Folliculitis | Purulence limited to epidermis | Immunocompetent patients: Staphylococcus aureus Immunocompromised patients: gram-negative organisms (e.g., Klebsiella, Proteus, or Enterobacter species) | Common in body areas associated with friction and heavy perspiration |
| Furuncle | Purulence surrounding hair follicles; extends to subcutaneous tissue Carbuncle is the coalescence of several furuncles | Immunocompetent patients: S. aureus Immunocompromised patients: gram-negative organisms (e.g., Klebsiella, Proteus, or Enterobacter species) | Common in body areas associated with friction and heavy perspiration |
| Nonpurulent SSTIs | |||
| Cellulitis | Well-demarcated border of erythema, warmth, edema, pain | Streptococci without abscess formation, staphylococci with abscess | Often occurs with trauma; sequelae may include lymphangitis, necrotizing infections, gangrene |
| Erysipelas | Intense erythema; well-demarcated, painful plaque | Beta-hemolytic streptococci, commonly Streptococcus pyogenes | More common with extremes of age (very young or old) |
| Impetigo | Crusted exudates with pustules or vesicles, common on face and extremities | Staphylococci or streptococci | Commonly seen in preschool-aged children; associated with poor hygiene, humid or warm temperatures |
SSTI = skin and soft tissue infection.
Figure 1.
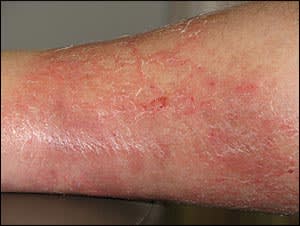
Cellulitis and edema associated with skin and soft tissue infection.
Figure 3.
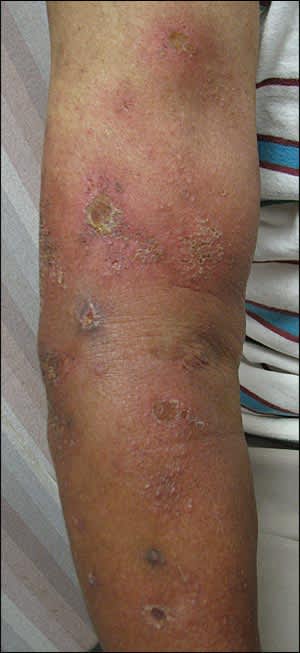
Cellulitis and impetigo associated with skin and soft tissue infection.
Complicated SSTIs
Complicated SSTIs include those with evidence of systemic toxicity (e.g., fever, tachycardia, hypotension); surgical wound infections; perianal infections; animal or human bites; necrotizing soft tissue infections; and those in patients with comorbid, immunocompromising conditions such as diabetes mellitus.3
Surgical wound infections commonly involve gram-positive cocci, such as S. aureus, as well as enterococci and Escherichia coli. In addition to common staphylococcal and streptococcal infections, human bites (intentional bites or clenched-fist injuries) often involve other organisms, such as Eikenella corrodens, Bacteroides fragilis, and Coryne-bacterium species.3 Cat and dog bite wounds may contain normal human skin flora, as well as animal oral flora (e.g., Pasteurella multocida).7
S. aureus and other aerobic gram-positive cocci are the most commonly isolated organisms in patients with acutely infected diabetic foot ulcers who have not been previously treated with antibiotics. Among persons with diabetes, the incidence of MRSA infections is increasing most rapidly in those with chronic foot ulcers who have previously received antibiotic therapy. These ulcers also are more prone to polymicrobial infection.8,9 Physicians should have a high index of suspicion for underlying osteomyelitis in patients with diabetic foot ulcers; surgical debridement and pathologic confirmation of diagnosis are required in these patients.9
Community-Acquired MRSA
The recent rapid increase in community-acquired MRSA soft tissue infections has forced physicians to reconsider their approach to routine SSTIs. Recent reports of antimicrobial resistance patterns have found that community-acquired MRSA is responsible for up to 60 percent of SSTIs in U.S. emergency departments.10,11 These rates are higher in certain populations, including ethnic minorities, children, intravenous drug users, patients who have received recent antibiotic treatment, men who have sex with men, residents of long-term care facilities and prisons, and patients receiving hemodialysis.5,12,13 In addition to purulent SSTIs, community-acquired MRSA is associated with other infectious processes, such as necrotizing pneumonia and sepsis.10 Although many patients with community-acquired MRSA infections describe the initial presentation as resembling a spider bite, there are no reliable signs and symptoms to distinguish community-acquired MRSA infections from other purulent SSTIs.13,14
Diagnosis and Assessment of SSTIs
SSTIs in the presence of comorbid infections, such as diabetes, neutropenia, or cirrhosis, are more likely to be severe and caused by uncommon organisms (e.g., Pseudomonas species, Klebsiella species, yeast, fungi).7 Complicated and potentially life-threatening SSTIs are characterized by fever, widespread or a rapidly spreading area of involvement, firm and hard feel of subcutaneous tissues, pain disproportionate to examination, skin sloughing, cutaneous bleeding with or without bullae, and skin crepitus.15 Additionally, patients with necrotizing infections may be lethargic or disorientated.7 Rapidly progressive and life-threatening infections warrant urgent surgical referral.3,7,16
Patients who present with signs or symptoms of systemic toxicity should undergo further evaluation to determine the need for inpatient care. Hospitalization should be considered in patients with the following laboratory findings: left shift in complete blood count with differential, elevated serum creatinine level, reduced serum bicarbonate level, elevated creatine kinase level, or C-reactive protein level greater than 13 mg per L (123.81 nmol per L).7
Wound and blood cultures are not routinely performed in patients with uncomplicated infections because results usually do not alter management decisions.7,17,18 However, wound cultures of purulent secretions should be performed in patients with multiple or extensive lesions, fever or other evidence of systemic illness, prior treatment failure, immunocompromise, trauma, water contact, or animal or human bites.7 If wound cultures are indicated, fluid or tissue specimens should be collected using aseptic technique via needle aspiration or tissue biopsy. Swab samples are discouraged because of a high rate of contamination, and insufficient volumes for culture because of inhibition of bacterial growth and adherence of organisms to swabs.19
Initial Management of SSTIs
In patients who have uncomplicated SSTIs with focal coalescence, incision and drainage alone is often curative, and antibiotics do not improve outcomes. In one prospective study of 69 children with uncomplicated SSTIs caused by community-acquired MRSA, incision and drainage alone was curative of abscesses measuring less than 5 cm in diameter, and the addition of antimicrobial therapy offered no therapeutic advantage.20 Two prospective studies with a total of more than 320 patients also support this approach.4,21 Two prospective trials suggest that when surgical drainage is performed, there is no difference in outcomes between wound irrigation with tap water versus sterile water.22,23
Antimicrobial Treatment
When indicated, antimicrobial therapy for uncomplicated SSTIs is usually initiated empirically. Familiarity with common pathogens and with local flora and resistance patterns is essential in choosing initial agents. It is important that physicians consider empiric regimens that are likely to be effective against the most prevalent local pathogens, including community-acquired MRSA. Beta-lactam antibiotics are appropriate first-line therapies when presentation and local patterns do not suggest MRSA infection (e.g., an infection with diffuse, noncoalescing cellulitis without a defined portal of entry).7,12 One recent study of 2,977 patients with cellulitis of unknown etiology showed comparable outcomes between oral beta-lactam and other antibiotics.24 However, in patients who have uncomplicated infections with impetigo, topical mupirocin (Bactroban) has been shown to have equal or better effectiveness than oral antibiotics.25
Oral antibiotics that have been shown to be effective against community-acquired MRSA include trimethoprim/sulfamethoxazole (Bactrim, Septra), tetracyclines, and clindamycin (Cleocin).3,7 Local patterns of sensitivity may vary, however. In one study, patients with uncomplicated cellulitis who received five days of antibiotic treatment had similar outcomes to those who were treated for 10 days.26 In patients with widespread or systemic infection, comorbidities, or an inability to tolerate oral treatment, vancomycin is the first-line parenteral anti-MRSA agent, although clindamycin, quinolones, linezolid (Zyvox), tigecycline (Tygacil), and daptomycin (Cubicin) may also be used.3 If clindamycin therapy is considered, a D-zone test should be performed to identify patients with inducible clindamycin resistance.12
The susceptibility of MRSA to different antibiotics varies by community. In one retrospective study of 492 adults with community-acquired MRSA SSTIs, susceptibility ranged from 100 percent for vancomycin to 73 percent for ciprofloxacin (Cipro).27 Another study of 508 children hospitalized with community-acquired MRSA SSTIs showed no statistically significant difference in recurrence or failure rates between trimethoprim/sulfamethoxazole and oral clindamycin.28 Table 2 presents dosing information for antimicrobial therapies for MRSA infection.5,7,29
Table 2. Antimicrobial Agents for the Treatment of MRSA Infection
| Agent | Dosing | Cost of generic (brand) | In retail discount programs* | Comments | |
|---|---|---|---|---|---|
| Outpatient treatment | |||||
| Doxycycline | Adults: 100 mg orally twice daily | $13 ($136)† | ✓ | Not for use in children younger than eight years | |
| Mupirocin (Bactroban) | Adults and children: Apply topically three times daily for five days (impetigo: 2% ointment, secondary skin infections: 2% cream) | Ointment: $35 ($73) for 22 g† Cream: NA ($62) for 15 g† | For topical treatment of impetigo and folliculitis | ||
| Retapamulin (Altabax) | Adults and children: Apply 1% ointment twice daily for five days | NA ($50) for 5 g† | For treatment of impetigo | ||
| Trimethoprim/sulfamethoxazole (Bactrim, Septra) | Adults: One or two double-strength tablets twice daily Children7: 8 to 12 mg per kg (trimethoprim) orally daily in two divided doses | Adults: $17 ($47)† Children: $18 (NA)† | ✓ | For skin eruptions, including those related to Stevens-Johnson syndrome | |
| Outpatient or inpatient treatment | |||||
| Clindamycin (Cleocin) | Adults: 300 to 450 mg orally every six hours; 600 mg IV every eight hours Children7: 10 to 20 mg per kg orally daily in three divided doses; 24 to 40 mg per kg IV daily in three divided doses | Adults: | D-zone test should be performed for inducible clindamycin resistance; associated with Clostridium difficile colitis | ||
| Oral, $65 ($330)† | |||||
| IV, $138 ($292)‡ | |||||
| Children: | |||||
| Oral, $42 ($59)† | |||||
| IV, $115 ($139)‡ | |||||
| Linezolid (Zyvox)§ | Adults: 600 mg orally or IV every 12 hours Children7: 10 mg per kg orally or IV every 12 hours | Adults: | Oral and intravenous therapy have nearly equal bioavailability; use cautiously with SSRIs and MAOIs | ||
| Oral, NA ($1,724)† | |||||
| IV, NA ($2,280)‡ | |||||
| Children: | |||||
| Oral, NA ($440)† | |||||
| IV, NA ($1,140)‡ | |||||
| Preferred inpatient treatment | |||||
| Vancomycin | Adults: 15 mg per kg IV every 12 hours Children7: 40 mg per kg IV daily in four divided doses | Adults and children: $220 ($690)‡ | Preferred drug for MRSA; adjust dosing for patients with renal impairment | ||
| Treatment of highly resistant organisms | |||||
| Daptomycin (Cubicin) | Adults: 4 mg per kg IV every 24 hours | NA ($2,380)‡ | Associated with myopathy; gastrointestinal adverse effects common | ||
| Linezolid | Same as above | ||||
| Tigecycline (Tygacil) | Adults: 100-mg loading dose IV, then 50 mg IV every 12 hours | NA ($1,524)‡ | Not approved for children | ||
IV = intravenously; MAOI = monoamine oxidase inhibitor; MRSA = methicillin-resistant Staphylococcus aureus; NA = not available; SSRI = selective serotonin reuptake inhibitor.
*—May be available at discounted prices ($10 or less for one month's treatment) at one or more national retail chains.
†—Estimated retail cost of 10-day treatment based on information obtained at http://www.drugstore.com (February 2010). Generic price listed first; brand price listed in parentheses.
‡—Average wholesale cost of 10-day treatment based on Red Book. Montvale, N.J.: Medical Economics Data; 2009. Generic price listed first; brand price listed in parentheses.
§—Linezolid should not be considered for routine outpatient treatment.
Newer Therapeutic Options for MRSA
LINEZOLID
The oxazolidinone antibiotic linezolid is bacteriostatic against staphylococci and enterococci, and is bacteriocidal against streptococci. Linezolid is effective against methicillin-sensitive S. aureus (MSSA) and MRSA, and the nearly equal bioavailability of oral and intravenous preparations makes it a convenient alternative to vancomycin. One study of 1,180 hospitalized patients with MRSA SSTIs reported a superior clinical response to linezolid over vancomycin (88 percent versus 66 percent), with similar medication-related adverse events.30 Because of cost (approximately $1,700 for a 10-day oral course) and the potential for the development of additional drug resistance, linezolid should be reserved for patients with MRSA who do not respond to other agents and for patients with complicated SSTIs in whom alternative treatment has been inferior.
As a weak monoamine oxidase inhibitor, linezolid may be associated with an increase in blood pressure. Serotonin syndrome has been reported in patients taking selective serotonin reuptake inhibitors with line-zolid. Linezolid also should be used with caution in patients with chronic kidney disease because it is renally excreted.26
TIGECYCLINE
Tigecycline, the first in a new class of broad-spectrum antibiotics, is a derivative of minocycline (Minocin) with decreased potential for inducing drug resistance. Tigecycline is indicated for the treatment of complicated SSTIs caused by MSSA and MRSA infections. It is also effective against staphylococci and streptococci isolates, vancomycin-resistant enterococci, other gram-negative infections, anaerobes, and atypical organisms. The bacteriostatic mechanism of action of tigecycline is inhibition of bacterial protein synthesis at the 30S ribosomal subunit. The drug is metabolized in the liver; therefore, dosing adjustments are needed in patients with severely impaired liver function (i.e., those with Child-Pugh classification C cirrhosis). No dosing adjustment is needed in patients with renal impairment, and tigecycline does not alter the effectiveness of warfarin (Coumadin) therapy.31
Tigecycline is administered intravenously. The most common adverse effects are nausea and vomiting. Disadvantages of tigecycline therapy include high cost and difficulty in maintaining therapeutic serum concentrations, which limits its use in the outpatient setting. As with tetracyclines, tigecycline is not recommended for use in children.31
DAPTOMYCIN
Daptomycin exhibits bactericidal activity against most gram-positive organisms, including MRSA and vancomycin-resistant enterococci. This unique medication disrupts the ionic electric potentials of the cell membrane by promoting rapid calcium-dependent efflux of potassium from the cell.32,33 In a prospective, open-label study comparing daptomycin with vancomycin for the treatment of complicated SSTIs with risk of MRSA, both groups had complete clinical resolution, with the daptomycin-treated group demonstrating a significantly reduced duration of intravenous therapy and reduced median number of days to achieve clinical cure.34
Daptomycin is administered intravenously. Potential adverse effects include myopathy and gastrointestinal effects, such as nausea, vomiting, and diarrhea. Daptomycin may elevate the prothrombin time and, therefore, should be used with caution in patients taking warfarin.33
RETAPAMULIN
Retapamulin (Altabax) is a topical antibiotic that has been investigated for use in the treatment of impetigo. Data from a randomized, double-blind multicenter study of 213 patients showed a clinically significant improvement in outcomes after seven days of retapamulin therapy, compared with placebo.35 Retapamulin has not been compared with topical mupirocin, however.
Prevention of SSTIs
The use of topical and intranasal mupirocin, antibacterial soaps, and body washes has been proposed to eradicate asymptomatic MRSA with the goal of reducing the incidence of symptomatic infection. However, studies have found insufficient data to support this strategy.36,37 Standard infection control precautions should be implemented and encouraged for all patients in ambulatory and inpatient settings, including proper and frequent handwashing, use of gloves when managing wounds, and contact precautions (e.g., use of gowns and gloves, grouping patients with similar infections) for patients with known or suspected MRSA infections.12 To prevent SSTIs, current consensus guidelines support proper foot care among patients with diabetes, tinea pedis, or pedal edema from venous insufficiency or lymphedema.5

