Am Fam Physician. 2010;81(10):1186-1188
Author disclosure: Nothing to disclose.
to the editor: Actinic keratoses are premalignant skin lesions resulting from chronic sun damage. For the average person, the 10-year probability of progression to squamous cell carcinoma is approximately 6 to 10 percent if lesions are not treated.1
There are multiple options for treating actinic keratoses. Isolated lesions are most commonly treated with a single course of liquid nitrogen cryotherapy. However, actinic keratoses can become numerous or confluent and cover such an extensive area that cryotherapy becomes impractical. In such cases, involved areas are commonly treated with topical fluorouracil, a pyrimidine analog antimetabolite. Fluorouracil is most effective for face or scalp lesions and is typically applied once or twice daily for several weeks, sometimes with short breaks in treatment as needed.
A major challenge of fluorouracil therapy is determining the duration of treatment. Evidence to support a definitive treatment duration is severely lacking.2 Optimal duration varies, typically ranging from two to six weeks. If duration is too short, lesions will persist. If duration is too long, patients experience prolonged and unnecessary physical discomfort and alterations in appearance, either of which could lead to decreased future compliance. Forearm lesions are relatively resistant to fluorouracil3 and often require a duration of six to eight weeks.
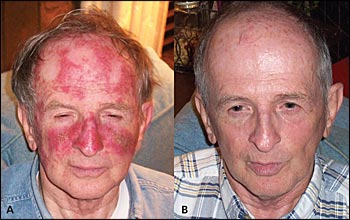
A 66-year-old man presented to our office with multiple facial actinic keratoses. Figure 1A shows this patient after twice-daily application of fluorouracil five times per week for four weeks. This demonstrates the desired appearance after an appropriate duration of usage. Erythema develops in several days. After continued application, the damaged skin becomes painful and inflamed with a beefy-red appearance with erosions and crusting. At this point, the medication should be stopped. Patients should be aware that this reaction is nearly inevitable,4,5 and they should not view it as an alarming adverse effect or allergic reaction. Non–sun-damaged skin shows little or no reaction.
Several months after this patient's treatment was stopped, his skin gradually returned to the appearance it had before treatment began, minus the actinic keratoses (Figure 1B). Complete healing of the inflammation generally takes one to two months. In addition to treating clinically visible lesions, fluorouracil can also treat subclinical lesions6 that may have become clinically visible in the future. Treatment is often prescribed in winter months because of an increased sensitivity to the sun. Because of the risk of future actinic keratoses and squamous cell carcinoma, close follow-up is recommended.