Background: For healthy newborns discharged before 72 hours of life, neonatal hyperbilirubinemia is the most common cause of readmission to the hospital. Predis-charge serum bilirubin levels require blood draws that can be invasive, time-consuming, and stressful. Hospitals are increasingly using transcutaneous bilirubin (TcB) values because the measurement devices are noninvasive and save time. However, there are limited data on the predictive value of TcB measurements. Varvarigou and colleagues developed a predictive nomogram using TcB measurements for the evaluation of neonatal hyperbilirubinemia.
The Study: This prospective study involved healthy infants born at 35 weeks of gestation and older, and weighing at least 2,000 g (4 lb, 7 oz), who were admitted to the well-infant nursery at a hospital in Greece from late 2005 through 2007. Exclusion criteria included admission to the intensive care unit, positive direct Coombs testing, jaundice requiring intervention within 24 hours of birth, and glucose-6-phosphate dehydrogenase deficiency.
Infants had TcB testing at 12, 18, 24, 36, 48, 60, and 72 hours of life (± 2 hours at each measurement), with a final measurement between 96 and 120 hours. All infants stayed for at least 72 hours, and had outpatient follow-up within 48 hours after discharge. Results were plotted against hour-specific bilirubin nomograms proposed by the American Academy of Pediatrics (AAP), and total serum bilirubin (TSB) testing was performed immediately if the TcB level was more than 15 mg per dL (256.56 μmol per L) or if the TcB level was 15 mg per dL or less but exceeded or was within 2 mg per dL (34.21 μmol per L) of the phototherapy guidelines of the AAP.
Results: Of the 2,745 infants born during the study, 2,039 met inclusion criteria and completed the follow-up process, resulting in 10,382 TcB measurements and 3,249 TSB measurements. Significant hyperbilirubinemia was documented in 122 infants (6 percent). The authors constructed a TcB nomogram (see accompanying figure) based on likelihood ratios (LR) because they are not dependent on disease prevalence and can predict disease probability more reliably than percentiles. Consequently, LRs were used to determine the high-, intermediate-, and low-risk cutoffs for each time point. A positive LR of greater than 10 indicates a high likelihood of disease, whereas a negative LR of less than 0.1 indicates a low likelihood. The high-risk zone of the nomogram had a positive LR for significant hyperbilirubinemia of 12.1 (73.9 percent sensitivity) at 24 hours of life, and a positive LR of 12.1 (90 percent sensitivity) at 48 hours of life. The TcB levels for the high-risk zone were 8 mg per dL (136.83 μmol per L) or higher at 24 hours of life and 11 mg per dL (188.14 μmol per L) or higher at 48 hours of life. The low-risk zone of the nomogram had a negative LR of 0.04 (97.7 percent sensitivity) at 24 hours of life, and a negative LR of 0.02 (98.8 percent sensitivity) at 48 hours of life. The TcB levels for the low-risk zone were 5.8 mg per dL (99.20 μmol per L) or lower at 24 hours of life and 9.5 mg per dL (162.49 μmol per L) or lower at 48 hours of life. Comparing the TcB and TSB results showed similar predictive value for the risk of significant hyperbilirubinemia. This study may be limited by the relatively homogeneous patient population, a cesarean delivery rate of 38 percent, and the hospital protocol requiring that nursery stays last a minimum of 72 hours.
Figure. Assessing the Risk of Hyperbilirubinemia in Newborns
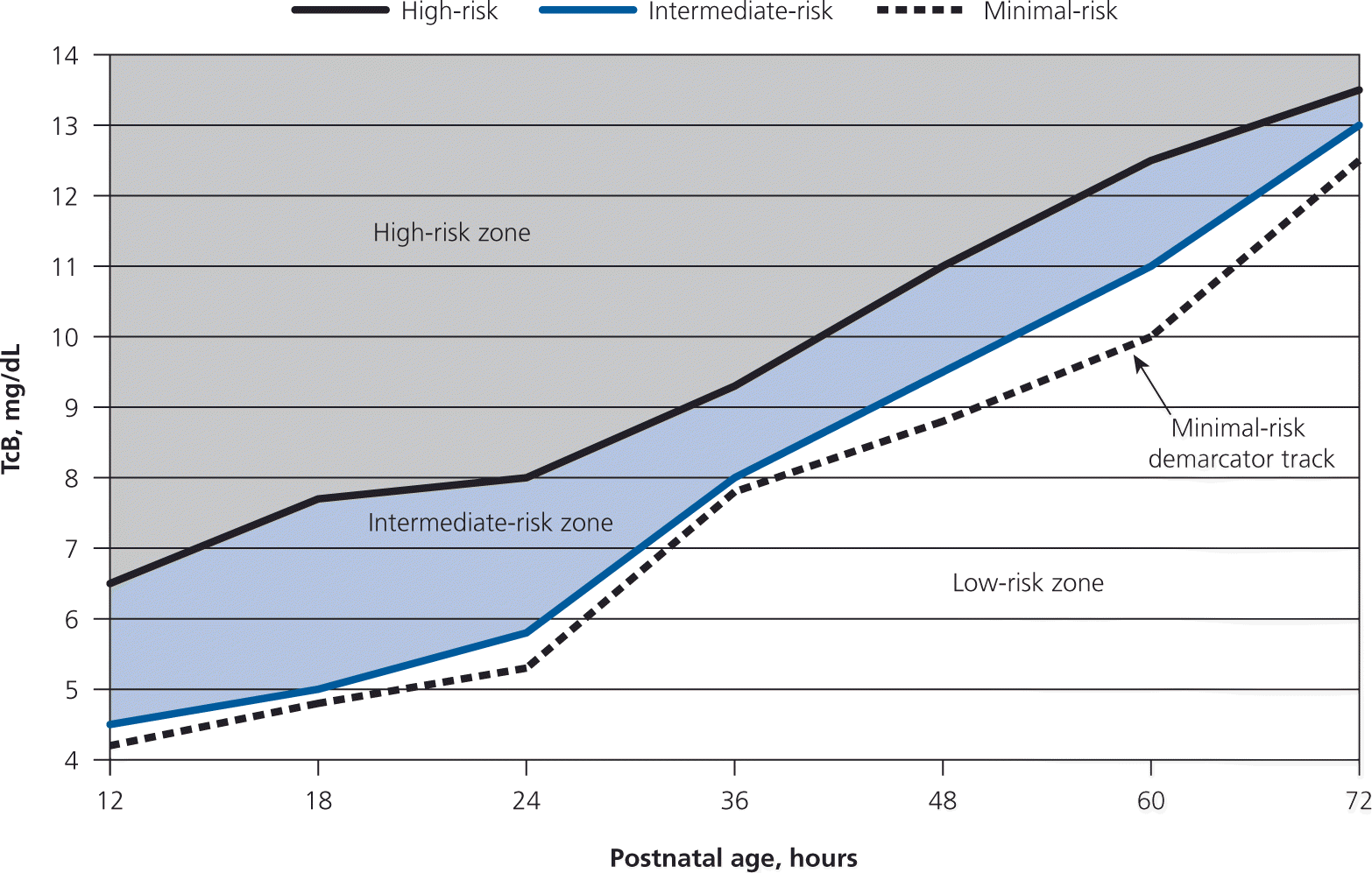
TcB nomogram for assessing the risk of subsequent significant hyperbilirubinemia in healthy term and near-term newborns. The high-risk zone is defined by the track of TcB values with a positive LR of greater than 10 and the low-risk zone by the track of TcB values with a negative LR of less than 0.1. The minimal-risk demarcator track (negative LR of 0) is also presented (dotted line). The nomogram was developed by using a total of 10,382 TcB measurements from 2,039 neonates with a gestational age of 35 weeks or older and a birth weight of 2,000 g (4 lb, 7 oz) or more. (LR = likelihood ratio; TcB = transcutaneous bilirubin.)
Reprinted with permission from Varvarigou A, Fouzas S, Skylogianni E, Mantagou L, Bougioukou D, Mantagos S. Transcutaneous bilirubin nomogram for prediction of significant neonatal hyperbilirubinemia. Pediatrics. 2009;124(4):1056.
Conclusion: The authors conclude that using TcB measurements between 12 and 72 hours of life can predict significant neonatal hyperbilirubinemia.

